The High-Risk Newborn
Acquired and Congenital Conditions
Learning Objectives
After studying this chapter, you should be able to:
• Describe the steps involved in neonatal resuscitation.
• Explain common respiratory problems in the newborn.
• Explain the causes and significance of nonphysiologic jaundice.
• Describe the nursing care of the infant with nonphysiologic jaundice.
• Describe causes of neonatal infections and nursing care for infants with infections.
• Explain the effect of maternal diabetes on the newborn and implications for nursing care.
• Explain hypocalcemia and phenylketonuria and the nursing considerations of each.
• Describe the effect of maternal substance abuse on the newborn and the nursing care.
![]()
http://evolve.elsevier.com/McKinney/mat-ch/
In addition to the high-risk conditions related to gestational age discussed in Chapter 29, the newborn at risk may have acquired or congenital complications. Acquired conditions may be associated with prenatal complications or may occur at birth or shortly thereafter.
Respiratory Complications
Respiratory distress is one of the most common problems of the neonate. It may be caused by asphyxia before or during birth, disease of the respiratory system, and other conditions that affect the infant’s ability to breathe. The nurse is responsible for evaluation of respiratory status at birth and throughout the hospital stay.
Asphyxia
Asphyxia is a lack of oxygen and increase of carbon dioxide in the blood. It may occur in utero, at birth, or later. When asphyxia occurs at birth, it may be a continuation of asphyxia that began before birth or the result of other factors, such as preterm lungs with insufficient surfactant to function adequately.
Lack of oxygen transported to the cells leads to anaerobic metabolism and the production of lactic acid. Metabolic acidosis develops when not enough bicarbonate is available to buffer the accumulating acids. Respiratory acidosis occurs as carbon dioxide accumulates. A high partial pressure of carbon dioxide occurs in arterial blood (PaCO2) and the partial pressure of oxygen (PO2), the pH, and bicarbonate levels are low.
Vasoconstriction caused by low oxygen decreases blood flow to all organs except the brain, myocardium, and adrenal glands. The ductus arteriosus and foramen ovale may remain open because of the low oxygen in the blood, high resistance to blood flow through constricted pulmonary vessels, and elevated pressure on the right side of the heart. Therefore, even circulating blood remains low in oxygen. Progression toward brain injury and death is rapid unless intervention is prompt.
Manifestations
If asphyxia occurs after birth, rapid respirations are followed by cessation of respirations (primary apnea) and a rapid fall in heart rate. Stimulation alone or with oxygen may restart respirations. If asphyxia continues without intervention, gasping respirations may resume weakly until the infant enters a period of secondary apnea. In secondary apnea, the oxygen levels in the blood continue to decrease, the infant loses consciousness, and stimulation is ineffective. Resuscitative measures must be initiated immediately to prevent permanent injury to the brain or death. Asphyxia seen at birth may be a continuation of asphyxia that began before or during birth. Therefore it is essential to begin resuscitation without delay.
Infants at Risk
Complications during pregnancy, labor, or birth increase the infant’s risk for asphyxia. In addition, if the expectant mother receives narcotics shortly before birth, the infant may be too depressed at birth to breathe well spontaneously. Naloxone (Narcan) (see Drug Guide) may be given to these infants if they have a normal color and heart rate with depressed respirations and the mother received opiates within 4 hours of the birth.
Neonatal Resuscitation
Although 90% of newborns have no difficulty with breathing at birth, approximately 10% require some help to begin respirations, and 1% require extensive resuscitative measures (Kattwinkel, 2011). Therefore all personnel involved in deliveries should know how to perform resuscitative measures (see Procedure: Performing Resuscitation in Newborns). Equipment should be readily available and functioning properly at all times so there is no delay in starting resuscitation. Nurses begin resuscitation as necessary and assist the physician or nurse practitioner with intubation, insertion of umbilical vein catheters, and administration of medications.
Maintaining thermoregulation is very important throughout care. A warming pad placed under linens in the radiant warmer may be used to provide extra heat. Infants less than 29 weeks of gestation can be placed in a polyethylene bag up to the neck before drying to reduce heat loss from evaporation. The bag also reduces stress from handling during drying. It is also important to prevent hyperthermia (Kattwinkel, 2011).
Some infants develop hypoxic-ischemic encephalopathy after asphyxia. Therapeutic hypothermia has been used to improve neurologic outcomes for these infants. Infants must be 36 or more weeks of gestation, have evidence of an acute perinatal hypoxic-ischemic event, and be in a facility where the treatment can be initiated within 6 hours of birth (Kattwinkel, 2011).
Once the infant is stabilized, the nurse continues to assess for changes. Infants with asphyxia often have other complications such as hypoglycemia, feeding and thermoregulation problems, seizures, hypotension, pulmonary hypertension, metabolic acidosis, renal problems, and fluid and electrolyte imbalances. They need close monitoring and often need intensive nursing care. Communication with the parents is a vital nursing function. Parents need explanations, realistic reassurance, and continued support after the crisis.
Transient Tachypnea of the Newborn
Infants with transient tachypnea of the newborn (TTN) develop rapid respirations soon after birth when inadequate absorption of fetal lung fluid occurs. The condition resolves within 24 to 72 hours. Risk factors include cesarean birth without labor, precipitous delivery, male gender, perinatal asphyxia, and maternal diabetes or asthma. Infants are usually term or late preterm, although some may be preterm.
Etiology
Although the exact cause of TTN is unknown, it is thought to result from a delay in absorption of fetal lung fluid by the pulmonary capillaries and lymph vessels. This causes decreased lung compliance and air trapping and produces signs similar to respiratory distress syndrome (RDS).
Manifestations
In TTN, respirations of 60 to 120 breaths per minute develop within hours of birth. Retractions, nasal flaring, grunting, and mild cyanosis are present. Chest radiography shows hyperinflation, perihilar streaking showing interstitial fluid along the bronchovascular spaces, and fluid in the fissures between the lobes of the lungs.
Therapeutic Management
Treatment is supportive and may include oxygen for cyanosis and gavage feedings while the respiratory rate is high to prevent aspiration and conserve energy. Because the signs are similar to those of RDS and sepsis, the infant is observed for those complications. Antibiotics may be given until sepsis is ruled out.
Nursing Considerations
After identifying signs, the nurse notifies the provider and carries out treatment. General nursing care is similar to that of the respiratory care of the preterm infant (see Chapter 29).
Meconium Aspiration Syndrome
Meconium-stained amniotic fluid occurs in 10% to 15% of births. Meconium aspiration syndrome (MAS), a condition in which there is obstruction, air trapping, and chemical pneumonitis caused by meconium in the infant’s lungs, develops in 5% of those infants (Ambalavanan & Carlo 2011). The condition occurs most often in infants who are postterm, small for gestational age (SGA), and compromised before birth by placental insufficiency or cord compression (Abu-Shaweesh, 2011).
Etiology
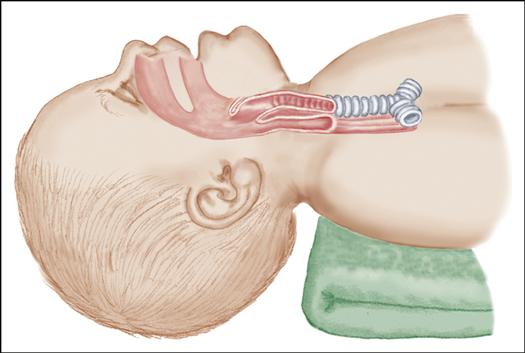
Although the normal fetus may pass meconium, MAS most often occurs when hypoxia causes increased peristalsis of the intestines and relaxation of the anal sphincter before or during labor. MAS develops when meconium in the amniotic fluid enters the lungs during fetal life or at birth. It may be drawn into the lungs if gasping movements occur in utero as a result of asphyxia and acidosis, or the meconium in the upper airways may be pulled deep into the respiratory passages when the infant takes the first breaths after birth.
Obstruction of the airways may be complete or partial. Atelectasis may result if small airways are completely obstructed. In partial obstruction, air can enter but not escape from the alveoli. During inhalation, the bronchioles expand slightly as air flows into them past the meconium. During exhalation, the passages constrict, and meconium blocks movement of air out of the lungs.
This ball-valve mechanism results in air trapping. The overdistended alveoli may develop an air leak, with escape of air into the pleural cavity (pneumothorax) or mediastinum (pneumomediastinum). Surfactant production may be inhibited, increasing the respiratory distress. In addition, meconium is irritating to lung tissue and causes an inflammatory reaction and chemical pneumonitis.
Severe MAS develops in only a small number of newborns with meconium below the vocal cords. The addition of meconium to lungs damaged by asphyxia may increase the severity of the condition. Injury from asphyxia interferes with clearing of lung fluid and production of surfactant and causes pulmonary vasoconstriction that can result in return to fetal circulation patterns. Persistent pulmonary hypertension of the newborn occurs in one third of infants with MAS (Burris, 2012).
Manifestations
Signs of mild to severe respiratory distress are present at birth, with tachypnea, cyanosis, retractions, nasal flaring, grunting, rales, and in severe cases, a barrel-shaped chest from hyperinflation. The infant’s nails, skin, and cord may be stained with meconium. Radiography shows atelectasis, consolidation, and hyperexpansion from air trapping.
Therapeutic Management
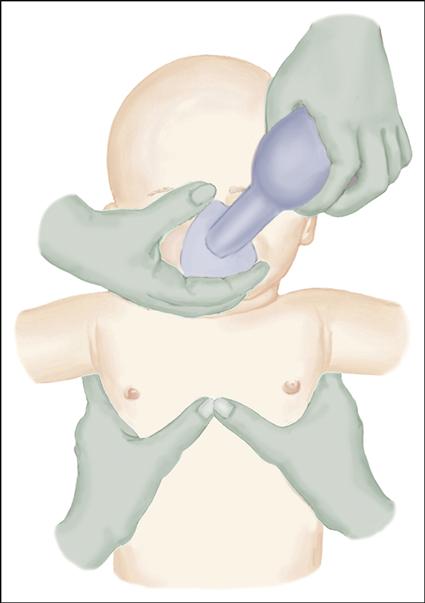
Suctioning the infant as soon as the head is born has not been found to reduce the incidence of MAS. The vigorous infant (good respirations and muscle tone and heart rate above 100 bpm) does not need special suctioning at birth and receives routine care. In infants with depressed respirations and muscle tone or a heart rate below 100 beats per minute (bpm), an endotracheal tube is used to remove as much meconium as possible (Kattwinkel, 2011).
Infants may need only warmed, humidified oxygen, or extensive respiratory support with mechanical ventilation may be required. High-frequency ventilation may be used. Surfactant lavage has been used in severe cases but is controversial (Abu-Shaweesh, 2011). Supportive care is given to meet the problems presented. Infants with severe MAS who do not respond to conventional treatment may benefit from extracorporeal membrane oxygenation (ECMO). ECMO, which is available in some hospitals, oxygenates the blood while bypassing the lungs to allow the infant’s lungs to rest temporarily and recover.
Nursing Considerations
When meconium is noted in the amniotic fluid during labor, the nurse notifies the primary caregiver so that delivery care can be adapted as necessary. Nurses from the neonatal intensive care unit (NICU) and a neonatologist may be present for the delivery. The nurse ensures that equipment, such as oxygen and suction, is functioning properly and assists with care at delivery. After the infant’s birth, nursing care is adapted as needed. Although meconium is sterile, lung injury promotes the growth of bacteria, and infants should be closely observed for infection. Nursing attention to thermoregulation and decreased stimulation is important.
Persistent Pulmonary Hypertension of the Newborn
Persistent pulmonary hypertension of the newborn (PPHN) is a condition in which vasoconstriction of pulmonary vessels prevents decrease of vascular resistance of the lungs after birth and normal changes to neonatal circulation are impaired. For this reason, the condition is also called persistent fetal circulation.
Etiology
PPHN occurs in infants who are late preterm, preterm, or term. The cause may be abnormal lung development, maternal use of nonsteroidal antiinflammatory drugs or selective serotonin reuptake inhibitors, or unknown. It is often associated with hypoxemia and acidosis from conditions such as asphyxia, meconium aspiration, sepsis, polycythemia, diaphragmatic hernia, and RDS (Ambalavanan & Carlo, 2011).
Inadequate oxygenation results in vasoconstriction, instead of the normal dilation, of the pulmonary artery and small pulmonary vessels, which causes increased resistance in the lungs. The elevated pulmonary vascular resistance causes a rise in pressure on the right side of the heart. This results in a right-to-left shunt of unoxygenated blood that flows through the foramen ovale. In addition, unoxygenated blood from the pulmonary artery flows through the ductus arteriosus to the aorta. Thus, blood bypasses the lungs, as occurs during fetal circulation. Metabolic acidosis causes more pulmonary vasoconstriction, making the condition even worse.
Manifestations
Infants with PPHN develop signs within the first 24 hours after birth. Tachypnea, respiratory distress, and progressive cyanosis often become worse with handling. Oxygen saturation and partial pressure of oxygen in arterial blood (PaO2) are decreased, PaCO2 is increased, and acidosis is present. Other signs may result from associated conditions. An echocardiogram demonstrates shunting.
Therapeutic Management
Management involves treating the underlying cause and relieving pulmonary vasoconstriction. Arterial pH may be increased with respiratory and drug therapy to cause pulmonary vasodilation. Sedation, high-frequency ventilation, surfactant therapy, and inhaled nitric oxide (which dilates pulmonary vessels) may be necessary. If other therapies fail, ECMO may be the only therapy that helps.
Nursing Considerations
Nursing care is similar to care of other infants with severe respiratory disease. Because infants with PPHN become hypoxic with activity and other stimuli, handling and noise are kept to a minimum. Attention to thermoregulation and assessment for hypoglycemia, hypocalcemia, anemia, and metabolic acidosis is important.
Hyperbilirubinemia
Jaundice is a common concern in caring for neonates. Conjugation of bilirubin and physiologic jaundice are discussed in Chapter 21. Nonphysiologic or pathologic jaundice is discussed here.
Jaundice becomes visible when the total serum bilirubin (TSB) reaches 5 to 6 mg/dL (Blackburn, 2013). Jaundice is considered abnormal or nonphysiologic when TSB rises more rapidly and to higher levels than is expected or stays elevated for longer than normal. Charts showing the expected rise and fall of bilirubin according to the age of the infant in hours are used to determine infants who need treatment for rising TSB.
Nonphysiologic jaundice may be seen in the first 24 hours of life. It is a concern because it may lead to bilirubin encephalopathy, a condition resulting from bilirubin toxicity. This may lead to kernicterus, the chronic and permanent result of bilirubin toxicity. In kernicterus, bilirubin deposits cause yellowish staining of the brain, especially the basal ganglia, cerebellum, hippocampus, and brainstem.
Although bilirubin encephalopathy and kernicterus are rare today because of improved treatment measures, the mortality and morbidity rate of affected infants is high. Those who survive may have cerebral palsy, intellectual impairment, hearing loss, or more subtle long-term neurologic and developmental problems. The exact level at which bilirubin encephalopathy develops is unknown. The toxic level may not be the same for all infants. It occurs at lower TSB levels and is more severe in infants who have complications or are preterm, late preterm, or low birth weight than in healthy, full-term infants.
Etiology
The most common cause of pathologic jaundice is hemolytic disease of the newborn from incompatibility between the blood of the mother and that of the fetus. The best known cause is Rh incompatibility, in which the Rh-negative mother forms antibodies when Rh-positive blood from the fetus enters her circulation (see Chapter 25). Antibodies may have developed during a previous pregnancy or after injury, abortion, amniocentesis, or a transfusion of Rh-positive blood. The antibodies cross the placenta and destroy fetal red blood cells. Excessive hemolysis causes erythroblastosis fetalis, agglutination and hemolysis of fetal erythrocytes from incompatibility between fetal and maternal blood types.
Infants with erythroblastosis fetalis are anemic from destruction of red blood cells. Severely affected infants may develop hydrops fetalis, a severe anemia that results in heart failure and generalized edema. Intrauterine fetal transfusions may be given. After birth, phototherapy and exchange transfusions are used to prevent kernicterus (Gruslin & Moore, 2011). Use of Rho(D) immune globulin, such as RhoGAM, to prevent the mother from forming antibodies against Rh-positive blood has greatly decreased the incidence of erythroblastosis fetalis.
ABO incompatibility also causes pathologic jaundice. Mothers with type O blood have natural antibodies to types A and B blood. The antibodies cross the placenta and cause hemolysis of fetal red blood cells. However, the destruction is much less severe than with Rh incompatibility and causes milder signs.
Other causes of nonphysiologic jaundice include infection, hypothyroidism, glucuronyl transferase deficiency, polycythemia, glucose-6-phosphate dehydrogenase deficiency, and biliary atresia. Infants of diabetic mothers are more likely to develop nonphysiologic jaundice, especially if they have macrosomia. Any condition that causes destruction of erythrocytes or impairment of the liver may result in elevated bilirubin levels.
Therapeutic Management
The focus of therapeutic management is prevention of bilirubin encephalopathy and kernicterus. The cause is determined by history and diagnostic tests to identify infections or blood abnormalities. During pregnancy, an Rh-negative expectant mother will have an indirect Coombs test to identify the presence of antibodies against fetal blood. If the test is positive, amniocentesis may be performed to determine the fetal Rh factor and the degree of hyperbilirubinemia (see Chapter 25).
When infants are jaundiced, the cord blood is used for a direct Coombs test to determine the infant’s blood type. A positive Coombs test indicates that antibodies from the mother have attached to the infant’s red blood cells. TSB levels are followed closely to detect changes that indicate treatment should be initiated or changed.
Nurses often assess infants for changes in jaundice. However visual inspection for jaundice is not an accurate way to determine the true bilirubin level. Other tests may be used to reduce the number of blood draws the infant must have. Transcutaneous bilirubinometers are hand-held devices that measure skin color to determine transcutaneous bilirubin (TcB). These noninvasive tests allow frequent checks of jaundice with no discomfort to the infant. However, they may not be accurate in preterm infants, infants receiving phototherapy, or if TSB levels are above 15 mg/dL (Bradshaw, 2010).
Phototherapy
Phototherapy is the most common treatment of jaundice and involves placing the infant under special lights. During phototherapy, bilirubin in the skin absorbs the light and changes into water-soluble products, the most important of which is lumirubin. These products do not require conjugation by the liver and can be excreted in the bile and urine. Because preterm infants are more vulnerable to bilirubin toxicity, phototherapy is begun at lower TSB levels than for full-term infants.
Phototherapy can be delivered in several ways. A bank of fluorescent lamps or “bili lights” can be placed over the infant, who is in an incubator or under a radiant warmer to maintain heat or in an open crib. The infant wears only a diaper to ensure maximal exposure of the skin. The diaper is removed if the TSB is becoming dangerously high. The eyes are closed and patches placed over them to prevent injury. More than one bank of lights may be used if the bilirubin level is high.
Other options for phototherapy include light-emitting diodes (LEDs), halogen lamps, and fiberoptic phototherapy blankets. The LED device is placed over the infant like fluorescent lights. The LED is long lasting and does not generate excessive heat. The halogen spotlight is used alone or with other lamps. The infant can be swaddled with the fiberoptic phototherapy blanket against the skin and does not require patches over the eyes. With the blanket, the mother may hold the infant without interfering with therapy. The blanket may be combined with phototherapy lights.
Side effects of phototherapy include frequent loose green stools, resulting from increased bile flow and peristalsis. The stools may injure the skin and cause fluid loss. Insensible water loss is increased too. A 25% increase in fluid intake is needed during phototherapy (Kaplan, Wong, Sibley, et al., 2011). Bronze baby syndrome, a grayish brown discoloration of the skin and urine, occurs in some infants with cholestatic jaundice. A macular skin rash may occur. The color changes and rash disappear gradually when phototherapy is completed.
TSB determinations are performed frequently to show the effectiveness of treatment and when it can be discontinued. When phototherapy is discontinued, TSB should be monitored for 24 hours to ensure further phototherapy is not necessary (Kamath, Thilo, & Hernandez, 2010). Explain to parents that the infants often have an elevation in bilirubin after phototherapy ends and that the health care provider may order additional blood tests after discharge.
Exchange Transfusions
Exchange transfusions are seldom necessary but are performed when phototherapy cannot reduce dangerously high bilirubin levels quickly enough. This treatment removes sensitized red blood cells, maternal antibodies, and unconjugated bilirubin and corrects severe anemia.
Procedure
During the exchange transfusion, blood in small portions is removed and replaced with an equal amount of donor blood. Twice the infant’s blood volume is administered. When an immediate transfusion is needed for Rh incompatibility, type O, Rh-negative blood cross-matched against the mother and infant is used. In ABO incompatibility, type O, Rh-negative (or Rh-compatible with the mother and infant) packed red blood cells with type AB plasma are used so that there are no anti-A or anti-B antibodies present (Gregory, Martin, & Cloherty, 2012).
At the end of the transfusion, about 85% of the infant’s red blood cells have been replaced, and the bilirubin level is reduced by 50% (Kaplan et al., 2011). When the level in the blood decreases, bilirubin from the tissues moves into the plasma. This rebound elevation of bilirubin may necessitate repeat transfusions, but phototherapy is generally adequate to resolve it.
Complications
Complications of exchange transfusions include electrolyte and acid-base imbalance, hypocalcemia, infection, hypoglycemia, acid-base imbalance, necrotizing enterocolitis, cardiac dysrhythmias, hemorrhage, thrombosis, and thrombocytopenia. Samples of the blood are analyzed for complete blood count (CBC), bilirubin and calcium levels, and other tests as needed.
Role of the Nurse
The nurse’s role during exchange transfusion is to prepare equipment, assess the infant during and after the procedure, and keep accurate records. A cardiac monitor is attached to the infant, and a radiant heater provides warmth. The nurse must clarify any misunderstandings that the parents may have about the treatment and help allay their anxiety.
Nursing Care
The Infant with Hyperbilirubinemia
Although collaborative care of the infant with jaundice is an important part of the nurse’s role, several nursing diagnoses are appropriate. Risk for Injury is discussed in this section. The nursing diagnosis Risk for Deficient Fluid Volume is discussed in the Nursing Care Plan: The Infant with Jaundice.
Assessment
Assess the level of jaundice at least every 8 hours by pressing the skin over a bony prominence and noting the color in the area before the blood returns. Assess the skin with phototherapy lights turned off because they distort the skin color. In infants with dark skin, assess the color of the conjunctivae of the eyes, palate, and oral mucous membranes. Determine the areas of the body affected by the jaundice, and document carefully for comparison during future assessments. Jaundice begins at the head and moves down the body as the bilirubin levels rise. Keep in mind that visual assessment is not an accurate method of assessing true bilirubin levels. Monitor TcB and laboratory TSB levels for change.
Assess for risk factors that might further increase bilirubin levels. Note temperature fluctuations, hypoglycemia, and infection. Determine the infant’s oral intake and number of stools.
Nursing Diagnosis and Planning
Nurses can do many things to prevent situations that might cause further rises in bilirubin. They must also protect the infant from injury from the light during phototherapy. The nursing diagnosis is:
Expected Outcomes
The infant will avoid injury resulting from increased bilirubin or exposure of the skin or eyes secondary to phototherapy lights.
Interventions
Maintaining a Neutral Thermal Environment
Prevent situations such as cold stress or hypoglycemia that could result in increased fatty acids in the blood caused by acidosis. Increased fatty acids decrease the availability of albumin-binding sites for unconjugated bilirubin. Prevent cold stress at birth and during all care by maintaining the infant in a neutral thermal environment. Check the infant’s axillary temperature every 2 to 4 hours to identify an early decrease before it becomes a problem. Dress the infant in warmed clothes and blankets on removal from phototherapy lights.
Prevent elevation of the infant’s temperature from exposure to the heat of the “bili lights.” Position the lights according to the manufacturer’s guidelines to prevent overheating the infant. Use a skin probe when the infant is in an incubator or radiant warmer to maintain the appropriate settings for the infant’s needs.