On completion of this chapter, the reader will be able to: • Differentiate the types of diabetes mellitus and their respective risk factors in pregnancy. • Compare insulin requirements during pregnancy, during the postpartum period, and with lactation. • Identify maternal and fetal risks or complications associated with diabetes in pregnancy. • Develop a plan of care for the pregnant woman with pregestational or gestational diabetes. • Compare the management of a pregnant woman with hyperthyroidism with one who has hypothyroidism. • Differentiate the management of various cardiovascular disorders in pregnant women. • Discuss the different types of anemia and their effects during pregnancy. • Explain the care of pregnant women with pulmonary disorders. • Discuss the effects of neurologic disorders on pregnancy. • Describe the care of women whose pregnancies are complicated by autoimmune disorders. • Discuss the care of pregnant women who use, abuse, or are dependent on alcohol or illicit or prescription drugs. Worldwide, the incidence of diabetes mellitus is increasing at a rapid rate. In 2011 an estimated 25.8 million people in the United States (8.3% of the total population) had diabetes. Of these, 7 million were undiagnosed. If current trends continue, by 2050 one in three U.S. adults will have diabetes (National Center for Chronic Disease Prevention and Health Promotion, 2011). In the United States experts predict a marked increase in the number of women with preexisting diabetes who will become pregnant (Moore and Catalano, 2009). Diabetes mellitus is currently the most common endocrine disorder associated with pregnancy, occurring in approximately 4% to 14% of pregnant women (Gilbert, 2011). The perinatal mortality rate for well-managed diabetic pregnancies, excluding major congenital malformations, is approximately the same as for any other pregnancy (Landon, Catalano, and Gabbe, 2012). The key to an optimal pregnancy outcome is strict maternal glucose control before conception and throughout the gestational period. Consequently for women with diabetes, much emphasis is placed on preconception counseling. Diabetes mellitus refers to a group of metabolic diseases characterized by hyperglycemia resulting from defects in insulin secretion, insulin action, or both (ADA, 2009). Insulin, produced by the beta cells in the islets of Langerhans in the pancreas, regulates blood glucose levels by enabling glucose to enter adipose and muscle cells, where it is used for energy. When insulin is insufficient or ineffective in promoting glucose uptake by the muscle and adipose cells, glucose accumulates in the bloodstream, and hyperglycemia results. Hyperglycemia causes hyperosmolarity of the blood, which attracts intracellular fluid into the vascular system, resulting in cellular dehydration and expanded blood volume. Consequently the kidneys function to excrete large volumes of urine (polyuria) in an attempt to regulate excess vascular volume and excrete the unusable glucose (glycosuria). Polyuria, along with cellular dehydration, causes excessive thirst (polydipsia). Diabetes may be caused either by impaired insulin secretion, when the beta cells of the pancreas are destroyed by an autoimmune process, or by inadequate insulin action in target tissues at one or more points along the metabolic pathway. Both of these conditions are commonly present in the same person; and determining which, if either, abnormality is the primary cause of the disease is difficult (ADA, 2009). For additional information on diabetes, visit the ADA website at www.diabetes.org. The current classification system includes four groups: type 1 diabetes, type 2 diabetes, other specific types (e.g., diabetes caused by genetic defects in beta cell function or insulin action, disease or injury of the pancreas, or drug-induced diabetes), and gestational diabetes mellitus (GDM) (ADA, 2009; Moore and Catalano, 2009). Type 1 diabetes includes cases that are caused primarily by pancreatic islet beta cell destruction and that are prone to ketoacidosis. People with type 1 diabetes usually have an abrupt onset of illness at a young age and an absolute insulin deficiency. Type 1 diabetes includes cases thought to be caused by an autoimmune process and those for which the cause is unknown (ADA, 2009; Landon, Catalano, and Gabbe, 2012). Type 2 diabetes is the most prevalent form of the disease and includes individuals who have insulin resistance and usually relative (rather than absolute) insulin deficiency. Specific causes of type 2 diabetes are unknown at this time. It often goes undiagnosed for years because hyperglycemia develops gradually and is often not severe enough for the person to recognize the classic signs of polyuria, polydipsia, and polyphagia. Most people who develop type 2 diabetes are obese or have an increased amount of body fat distributed primarily in the abdominal area. Other risk factors for the development of type 2 diabetes include aging, a sedentary lifestyle, family history and genetics, puberty, hypertension, and prior gestational diabetes. Type 2 diabetes often has a strong genetic predisposition (ADA, 2009; Moore and Catalano, 2009). Gestational diabetes mellitus (GDM) is any degree of glucose intolerance with the onset or first recognition occurring during pregnancy. This definition is appropriate whether or not medication is used for treatment or the diabetes persists after pregnancy. It does not exclude the possibility that the glucose intolerance preceded the pregnancy or that medication might be required for optimal glucose control. Women diagnosed with gestational diabetes should be retested 6 to 12 weeks after the pregnancy ends (Landon, Catalano, and Gabbe, 2012). Dr. Priscilla White, a physician who worked with pregnant women with diabetes during the 1940s, developed a classification system specifically for use with this group of women (Table 11-1). White’s system was based on age at diagnosis; duration of illness; and presence of end-organ involvement, especially eye and kidney (Landon, Catalano, and Gabbe, 2012; Moore and Catalano, 2009). Her classification system has been modified through the years but is still used frequently to assess both maternal and fetal risk. Women in classes A through C generally have positive pregnancy outcomes as long as their blood glucose levels are well controlled. However, women in classes D through T usually have poorer pregnancy outcomes because they have already developed the vascular damage that often accompanies long-standing diabetes. TABLE 11-1 WHITE’S CLASSIFICATION OF DIABETES IN PREGNANCY (MODIFIED) During the first trimester of pregnancy the pregnant woman’s metabolic status is significantly influenced by the rising levels of estrogen and progesterone. These hormones stimulate the beta cells in the pancreas to increase insulin production, which promotes increased peripheral use of glucose and decreased blood glucose, with fasting levels being reduced by approximately 10% (Fig. 11-1, A). At the same time an increase in tissue glycogen stores and a decrease in hepatic glucose production occur, which further encourage lower fasting glucose levels. As a result of these normal metabolic changes of pregnancy, women with insulin-dependent diabetes are prone to hypoglycemia during the first trimester. During the second and third trimesters pregnancy exerts a “diabetogenic” effect on the maternal metabolic status. Because of the major hormonal changes, decreased tolerance to glucose, increased insulin resistance, decreased hepatic glycogen stores, and increased hepatic production of glucose occur. Rising levels of human chorionic somatomammotropin, estrogen, progesterone, prolactin, cortisol, and insulinase increase insulin resistance through their actions as insulin antagonists. Insulin resistance is a glucose-sparing mechanism that ensures an abundant supply of glucose for the fetus. Maternal insulin requirements gradually increase from approximately 18 to 24 weeks of gestation to approximately 36 weeks of gestation. Maternal insulin requirements may double or quadruple by the end of the pregnancy (see Fig. 11-1, B and C). At birth expulsion of the placenta prompts an abrupt drop in levels of circulating placental hormones, cortisol, and insulinase (see Fig. 11-1, D). Maternal tissues quickly regain their prepregnancy sensitivity to insulin. For the nonbreastfeeding mother the prepregnancy insulin-carbohydrate balance usually returns in approximately 7 to 10 days (see Fig. 11-1, E). Lactation uses maternal glucose; therefore the breastfeeding mother’s insulin requirements remain low during lactation. On completion of weaning the mother’s prepregnancy insulin requirement is reestablished (see Fig. 11-1, F). Only about 10% of pregnancies complicated by diabetes occur in women who have preexisting disease (Landon, Catalano, and Gabbe, 2012). Women who have pregestational diabetes mellitus may have either type 1 or 2 diabetes, which may be complicated by vascular disease, retinopathy, nephropathy, or other diabetic complications. Type 2 is a more common diagnosis than type 1. Almost all women with pregestational diabetes are insulin dependent during pregnancy. According to White’s classification system, these women fall into classes B through T (see Table 11-1). Preconception counseling is recommended for all women of reproductive age who have diabetes because it is associated with less perinatal mortality and fewer congenital anomalies (Moore and Catalano, 2009). Under ideal circumstances women with pregestational diabetes are counseled before the time of conception to plan the optimal time for pregnancy, establish glycemic control before conception, and diagnose any vascular complications of diabetes. However, estimates indicate that less than 20% of women with diabetes in the United States participate in preconception counseling (Landon, Catalano, and Gabbe, 2012). Although maternal morbidity and mortality rates have improved significantly, the pregnant woman with diabetes remains at risk for the development of complications during pregnancy. Poor glycemic control around the time of conception and in the early weeks of pregnancy is associated with an increased incidence of miscarriage. Women with good glycemic control before conception and in the first trimester are no more likely to miscarry than women who do not have diabetes (Moore and Catalano, 2009) (see Evidence-Based Practice box). Poor glycemic control later in pregnancy, particularly in women without vascular disease, increases the rate of fetal macrosomia. Macrosomia has been defined as a birth weight more than 4000 to 4500 g or greater than the 90th percentile. It occurs in approximately 40% of pregestational diabetic pregnancies and up to 50% of pregnancies complicated by GDM (Landon, Catalano, and Gabbe, 2012). Infants born to women with diabetes tend to have a disproportionate increase in shoulder, trunk, and chest size. Because of this tendency the risk of shoulder dystocia is greater in these babies than in other macrosomic infants. Therefore women with diabetes face an increased likelihood of cesarean birth because of failure of fetal descent or labor progress or of operative vaginal birth (birth involving the use of episiotomy, forceps, or vacuum extractor) (Moore and Catalano, 2009). Women with preexisting diabetes are at risk for several obstetric and medical complications. In general the risk of developing these complications increases with the duration and severity of the woman’s diabetes. In one study the rates of preeclampsia, preterm birth, cesarean birth, and maternal mortality were much higher in women with preexisting diabetes than in women who did not have this disease. For example, approximately a third of women who have had diabetes for more than 20 years develop preeclampsia. Women with nephropathy and hypertension in addition to diabetes are also increasingly likely to develop preeclampsia. The rate of hypertensive disorders in all types of pregnancies complicated by diabetes is 15% to 30%. Chronic hypertension occurs in 10% to 20% of all pregnant women with diabetes and in up to 40% of women who have preexisting renal or retinal vascular disease (Moore and Catalano, 2009). Hydramnios (polyhydramnios) frequently develops during the third trimester of pregnancy in women with diabetes. Its cause is unknown. One theory is that hydramnios in women with diabetes is caused by an increased glucose concentration in amniotic fluid resulting from maternal and fetal hyperglycemia. The complications most frequently associated with hydramnios (usually defined as an amniotic fluid index [AFI] greater than 24 to 25 cm) are abruptio placentae (placental abruption), uterine dysfunction, and postpartum hemorrhage (Cunningham, Leveno, Bloom, et al., 2010). Ketoacidosis (accumulation of ketones in the blood resulting from hyperglycemia and leading to metabolic acidosis) occurs most often during the second and third trimesters, when the diabetogenic effect of pregnancy is the greatest. When the maternal metabolism is stressed by illness or infection, the woman is at increased risk for diabetic ketoacidosis (DKA). DKA can also be caused by poor compliance with treatment or the onset of previously undiagnosed diabetes (Moore and Catalano, 2009). The use of beta-mimetic drugs such as terbutaline (Brethine) for tocolysis to stop preterm labor or corticosteroids given to enhance fetal lung maturation may also contribute to the risk for hyperglycemia and subsequent DKA (Cunningham, Leveno, Bloom, et al., 2010; Iams, Romero, and Creasy, 2009). DKA may occur with blood glucose levels barely exceeding 200 mg/dL, compared with 300 to 350 mg/dL in the nonpregnant state. In response to stress factors such as infection or illness, hyperglycemia occurs as a result of increased hepatic glucose production and decreased peripheral glucose use. Stress hormones, which act to impair insulin action and further contribute to insulin deficiency, are released. Fatty acids are mobilized from fat stores to enter the circulation. As they are oxidized, ketone bodies are released into the peripheral circulation. The woman’s buffering system is unable to compensate, and metabolic acidosis develops. The excessive blood glucose and ketone bodies result in osmotic diuresis with subsequent loss of fluid and electrolytes, volume depletion, and cellular dehydration. DKA is a medical emergency. Prompt treatment is necessary to prevent maternal coma or death. Ketoacidosis occurring at any time during pregnancy can lead to intrauterine fetal death. The incidence of DKA has decreased in recent years because of advances in clinical management and blood glucose monitoring (Inturrisi, Lintner, and Sorem, 2013). Currently it affects only about 1% of pregnant women with diabetes (Cunningham, Leveno, Bloom, et al., 2010). The rate of intrauterine fetal demise (IUFD) with DKA, formerly approximately 35%, is 10% or less (Moore and Catalano, 2009) (Table 11-2). TABLE 11-2 DIFFERENTIATION OF HYPOGLYCEMIA (INSULIN SHOCK) AND HYPERGLYCEMIA (DIABETIC KETOACIDOSIS)
High Risk Perinatal Care
Preexisting Conditions
Diabetes Mellitus
Pathogenesis
Classification
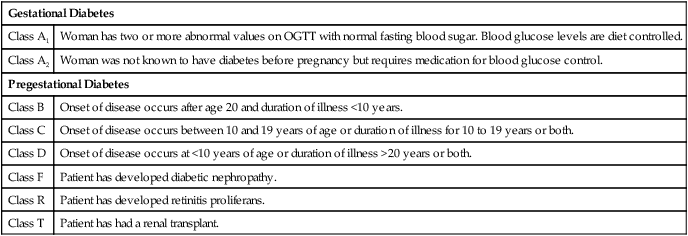
White’s Classification of Diabetes in Pregnancy
Gestational Diabetes
Class A1
Woman has two or more abnormal values on OGTT with normal fasting blood sugar. Blood glucose levels are diet controlled.
Class A2
Woman was not known to have diabetes before pregnancy but requires medication for blood glucose control.
Pregestational Diabetes
Class B
Onset of disease occurs after age 20 and duration of illness <10 years.
Class C
Onset of disease occurs between 10 and 19 years of age or duration of illness for 10 to 19 years or both.
Class D
Onset of disease occurs at <10 years of age or duration of illness >20 years or both.
Class F
Patient has developed diabetic nephropathy.
Class R
Patient has developed retinitis proliferans.
Class T
Patient has had a renal transplant.
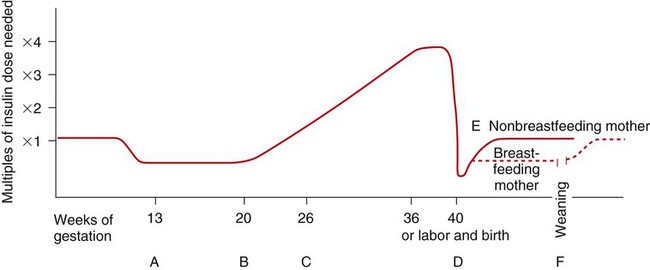
Metabolic Changes Associated with Pregnancy
Pregestational Diabetes Mellitus
Preconception Counseling
Maternal Risks and Complications



