Concurrent Disorders During Pregnancy
Learning Objectives
After studying this chapter, you should be able to:
• Describe the effects of pregnancy on glucose metabolism.
• Discuss the effects and management of preexisting diabetes mellitus during pregnancy.
• Explain the effects and management of gestational diabetes mellitus.
• Describe management of the pregnant and postpartum woman who has heart disease.
![]()
http://evolve.elsevier.com/McKinney/mat-ch/
Pregnancy may alter the course of a concurrent disease, or a disease and its treatment may have unwanted effects on the pregnancy. As a result, the usual antepartum care must be adapted to include increased surveillance of the mother and the fetus. Moreover, some disorders that are mild or even subclinical in the pregnant woman can cause massive damage to a fetus.
Diabetes Mellitus
Pathophysiology
Etiology
Preexisting, or type 1, diabetes mellitus is a complex disorder of carbohydrate metabolism caused primarily by a partial or complete lack of insulin secretion by the beta cells of the pancreas. Some cells, such as those in skeletal and cardiac muscles and in adipose tissue, require insulin to carry glucose across the cell membranes. Without insulin, glucose accumulates in the blood, resulting in hyperglycemia. The body attempts to dilute the glucose load by any means possible. The first strategy is to increase thirst (polydipsia), one of the classic symptoms of diabetes mellitus. Next, fluid from the intracellular spaces is drawn into the vascular bed, resulting in dehydration at the cellular level but fluid volume excess in the vascular compartment. The kidneys attempt to excrete large volumes of this fluid plus the heavy solute load of glucose (osmotic diuresis). This excretion produces the second sign of diabetes, polyuria, as well as glycosuria (glucose in the urine). Without glucose, the cells starve, so weight loss occurs even though the person ingests large amounts of food (polyphagia).
If the body cannot metabolize glucose, it begins to metabolize protein and fat (lipogenesis) to meet energy needs. Metabolism of protein produces a negative nitrogen balance, and the metabolism of fat results in the buildup of ketone bodies (e.g., acetone, acetoacetic acid, or beta-hydroxybutyric acid) or ketosis (accumulation of ketone bodies or acids) in the body.
If the disease is not well controlled, serious complications may occur. Hypoglycemia or hyperglycemia can result if the amount of insulin does not match the diet. Moreover, fluctuating periods of hyperglycemia and hypoglycemia damage small blood vessels throughout the body. This damage can cause serious impairment, especially in the kidneys, eyes, and heart.
Effect of Pregnancy on Fuel Metabolism
To comprehend the relationship of diabetes mellitus and pregnancy, it is necessary to understand how pregnancy and diabetes alter the metabolism of food.
Early Pregnancy
Metabolic changes can be divided into those that occur early in pregnancy (from 1 to 20 weeks of gestation) and those that occur late in pregnancy (from the end of 20 weeks of gestation until birth). During early pregnancy, maternal metabolic rates and energy needs change little. During this time, however, insulin release in response to serum glucose levels increases. As a result, significant hypoglycemia may occur, particularly in women who experience the nausea, vomiting, and anorexia that often occur during the first weeks of pregnancy.
In an uncomplicated pregnancy, the availability of glucose and insulin favors the development and storage of fat during the first half of pregnancy. Accumulation of fat prepares the mother for the rise in energy use by the growing fetus during the second half of pregnancy.
Late Pregnancy
During the second half of pregnancy, when fetal growth accelerates, levels of placental hormones rise sharply. These hormones, particularly estrogen, progesterone, and human placental lactogen, create resistance to insulin in maternal cells to provide an abundant supply of glucose for the fetus. The hormones have a diabetogenic effect, or a condition that produces the effects of diabetes mellitus. These effects may leave the woman with insufficient insulin and episodes of hyperglycemia.
For most women, insulin resistance is not a problem. The pancreas responds by simply increasing the production of insulin. If the pancreas is unable to respond, however, the woman will experience periods of hyperglycemia.
During late pregnancy, the fetus continuously withdraws nutrients, such as glucose and amino acids, from maternal blood. The result is an earlier-than-normal switch from carbohydrate metabolism to gluconeogenesis (formation of glycogen from noncarbohydrate sources such as proteins and fat). Because the fetus uses many of the amino acids, the process becomes predominantly one of fat utilization. This process produces high levels of free fatty acids that further inhibit the uptake and oxidation of glucose and thus preserve glucose for use by the central nervous system (CNS) and the fetus. These metabolic changes are similar to those that occur during “accelerated starvation,” when fat is metabolized to meet the body’s energy needs.
Classification
Diabetes is classified as type 1 (insulin deficient) or type 2 (insulin resistant, with a relative deficiency of insulin to metabolize carbohydrate) according to whether the person requires the administration of insulin to prevent ketoacidosis. A third type, gestational diabetes mellitus (GDM), is one in which any degree of glucose intolerance has its onset or first recognition during pregnancy (Box 26-1).
An additional classification of diabetes may be used for descriptive purposes. The White classification describes the age at onset of diabetes, its duration based on the woman’s current age, and vascular complications, such as retinopathy, that are present. GDM descriptions in White’s classification also include A1 (diet controlled) or A2 (diet and insulin controlled) (Castro & Ogunyemi, 2010; Cunningham, Leveno, Bloom, et al., 2010; Fraser & Farrell, 2011; Moore & Catalano, 2009).
Incidence
Diabetes mellitus is a medical condition that can adversely affect pregnancy, and its frequency is increasing along with obesity and abnormal lipid profiles. About 90% to 95% of diagnoses in the total population are type 2, whereas type 1 accounts for only 5% to 10% of those diagnosed. The pregnant woman may have preexisting diabetes (type 1 or type 2), or she may develop GDM during the course of pregnancy. The pregnant woman may have had undiagnosed type 2 diabetes that is discovered during pregnancy screening for GDM or her postpartum visit at 6 to 12 weeks (American College of Obstetricians and Gynecologists [ACOG], 2010d; Centers for Disease Control and Prevention [CDC], 2011b).
About 7% of all pregnancies are affected by GDM, but the range varies from 1% to 14% among different ethnic groups, with higher rates in African-Americans, Latinas, American Indians, some Asian-Americans, and Pacific Islanders. Women who have GDM in pregnancy have a 35% to 60% likelihood of developing diabetes in the next 10 to 20 years (ACOG, 2010d; American Diabetes Association [ADA], 2011a; CDC, 2011b).
Pathology
The root cause for type 2 diabetes is insulin resistance, in which body cells do not use glucose properly. The need for insulin rises, and the pancreas gradually loses the ability to supply enough of the hormone needed to metabolize glucose. Type 2 diabetes may be controlled by diet, exercise, and weight reduction, or it may require oral agents or insulin to control high glucose levels.
Preexisting Diabetes Mellitus
Maternal Effects
Preeclampsia occurs more often in the woman with diabetes than in the unaffected population (ACOG, 2010c; Cunningham et al., 2010). The development of ketoacidosis is a threat to women who require insulin to properly control their diabetes. Ketoacidosis is often precipitated by infection or missed insulin doses, particularly in the woman with type 1 diabetes. Moreover, ketoacidosis may develop during pregnancy at lower thresholds of hyperglycemia than when the woman is not pregnant. Untreated ketoacidosis can progress to fetal and maternal death.
Urinary tract infections are more common, possibly because glucose-rich urine provides a good medium for bacterial growth. Other effects include hydramnios (excess volume of amniotic fluid), which may result from fetal hyperglycemia and consequent fetal diuresis, and premature rupture of membranes, which may be caused by overdistention of the uterus by hydramnios or a large fetus. A difficult labor, shoulder dystocia (delayed or difficult birth of fetal shoulders after the head is born), and injury to the birth canal are more likely if the fetus is large. Large fetal size also increases the likelihood that a cesarean birth will be necessary and increases the risk for postpartum hemorrhage.
Production of excess amniotic fluid (hydramnios) may occur if maternal insulin control is not optimal. The excess fluid distends the uterus, possibly leading to early rupture of membranes, prolapsed cord (see Chapter 27), abnormal labor, and postpartum hemorrhage caused by failure of the uterus to contract effectively.
Fetal Effects
Fetal and neonatal effects of preexisting diabetes depend on the timing and severity of maternal hyperglycemia and the degree of maternal vascular impairment. During the first trimester, when major fetal organ development is occurring, the effects of the abnormal metabolic environment, such as hypoglycemia, hyperglycemia, and ketosis, may lead to an increased incidence of spontaneous abortion or major fetal malformations.
Congenital Malformation
The most common major congenital malformations associated with preexisting diabetes are neural tube defects, caudal regression syndrome (malformation that results when the sacrum, lumbar spine, and lower extremities fail to develop), and cardiac defects. Women who are hyperglycemic during the first trimester have a risk that is three to four times higher than for women with normal serum glucose of having an infant with a structural anomaly. Fewer malformations occur in women with good glycemic control during formation of major body structures. Recent data indicate that control of GDM reduces newborn obesity and later improved control of childhood obesity (Fleming & Corbett, 2010; Moore & Catalano, 2009; Moore, 2010).
The occurrence of maternal and fetal-neonatal complications can be greatly diminished if the mother maintains normal and stable blood glucose levels before and throughout pregnancy. The objective of the team providing treatment is to devise a plan that allows the woman to maintain a blood glucose level as close to normal as possible (see Nursing Care Plan).
Variations in Fetal Size
Fetal growth is related to maternal vascular integrity. In women without vascular impairment, glucose and oxygen are easily transported to the fetus; if the woman is hyperglycemic, so is the fetus. Although maternal insulin does not cross the placental barrier, the fetus produces insulin by the 10th week of gestation. Fetal macrosomia (large fetal size, ≥4000 g at term) results when elevated levels of blood glucose stimulate excessive production of fetal insulin, which acts as a powerful growth hormone.
Conversely, placental perfusion may be decreased with vascular impairment. Vascular impairment may be caused by complications of the diabetes or by vasoconstriction that occurs in preeclampsia, a common added complication for all women with diabetes. When placental perfusion is impaired, the supply of glucose as well as oxygen will be decreased. If placental perfusion is impaired for a prolonged period, the infant is likely to experience fetal growth restriction ( FGR).
Neonatal Effects
The four major complications of maternal diabetes for the newborn are hypoglycemia, hypocalcemia, hyperbilirubinemia, and respiratory distress syndrome. All can be minimized by stabilizing and maintaining maternal glucose levels near normal, particularly in the last weeks of pregnancy and during labor.
Newborns of a woman with poorly controlled diabetes and no vascular impairment are more likely to be very large, often weighing well over 4500 g (9 lb, 15 oz). About 30% of these newborns may have an enlarged heart (cardiomegaly), and 5% to 10% may require care for congestive heart failure (Moore & Catalano, 2009).
Hypoglycemia
The neonate is at higher risk for hypoglycemia because fetal insulin production was accelerated during pregnancy to metabolize excessive glucose received from the expectant mother. The constant stimulation of hyperglycemia leads to hyperplasia and hypertrophy of the islets of Langerhans in the pancreas. At birth, when the maternal glucose supply is withdrawn, the level of neonatal insulin exceeds the available glucose and hypoglycemia develops rapidly.
Hypocalcemia
Hypocalcemia, defined as a calcium concentration of less than 7 mg/dL, usually occurs within 72 hours of birth. The risk for hypocalcemia is less if the maternal glucose level is controlled (Moore & Catalano, 2009).
Hyperbilirubinemia
The fetus who experiences recurrent hypoxia caused by maternal vascular impairment compensates by producing additional erythrocytes to carry oxygen supplied by the mother, resulting in polycythemia. After birth, the excess in erythrocytes is broken down, releasing large amounts of bilirubin into the neonate’s circulation.
Respiratory Distress Syndrome
Fetal hyperinsulinemia retards cortisol production, which is necessary for synthesis of surfactant that increases the risk that the newborn will experience respiratory distress syndrome. Reduced lung fluid clearance and delayed thinning of lung connective tissue may also play a part, although other authorities believe that gestational age is the primary determinant of whether an infant will have respiratory distress syndrome (Cunningham et al., 2010; Fraser & Farrell, 2011). (See Chapter 29 for additional information about neonatal complications.)
Maternal Assessment
The initial prenatal assessment for the woman with preexisting diabetes includes a history, physical examination, and laboratory tests.
History
A detailed history should include the onset and management of the diabetic condition. How long has the woman had the disease? How does she maintain normal blood glucose levels? Can she monitor her blood glucose level and self-administer insulin? The degree of glycemic control before pregnancy is of particular interest. Effective management depends on the woman’s adherence to a plan of care. Therefore her knowledge of how diabetes and pregnancy interact must be determined. Her support person’s knowledge also must be assessed, and specific learning needs should be identified. In addition, the woman’s emotional status should be assessed to determine how she is coping with pregnancy superimposed on preexisting diabetes.
All women with diabetes should be seen by a qualified nurse educator for an individualized assessment to ensure that they can monitor blood glucose accurately. Accurate readings depend on performing the test correctly and as often as recommended by the health care team. Most pregnant women who need a hypoglycemic agent take insulin rather than an oral agent, although an occasional woman refuses to take injectable medication for blood glucose control. The nurse, who is often a diabetic educator, must validate the woman’s skill in mixing and administering insulin, using a sliding scale for added insulin, or using an insulin pump if the drug will be given that way. A woman who has taken insulin before pregnancy often needs teaching about why the very tight control is needed during pregnancy and why the need for progressively more insulin is common in the later weeks of pregnancy.
Physical Examination
In addition to routine prenatal examination (see Chapter 13), specific efforts should be made to assess the effects of diabetes. A baseline electrocardiogram (ECG) determines cardiovascular status. Evaluation for retinopathy should be performed, with referral to an ophthalmologist if necessary. The woman’s weight and blood pressure must be monitored carefully because of the increased risk for the development of preeclampsia. Fundal height should be measured, noting any abnormal increase in size that may indicate macrosomia or hydramnios. Reduced growth in fundal height suggests FGR associated with maternal vascular impairment. Ultrasonography is indicated to determine true gestational age and to identify any abnormal fetal growth or amount of fluid (ACOG 2010c).
Laboratory Tests
In addition to routine prenatal laboratory examinations, baseline renal function should be assessed with a 24-hour urine collection for total protein excretion and creatinine clearance. A random midstream urine sample should be checked at each prenatal visit for possible urinary tract infections and for the presence of protein, glucose, and ketones. Thyroid function tests should be performed because of the risk for coexisting thyroid disease.
Glycemic control should be evaluated on the basis of glycosylated hemoglobin, or HbA1c. Prolonged hyperglycemia causes some of the hemoglobin in erythrocytes to remain saturated with glucose for the life of the red blood cell. Unlike tests that reflect the amount of glucose in the plasma at that moment, the HbA1c assay result is not affected by recent intake or restriction of food.
Fetal Surveillance
Because of greater risk for congenital anomalies or fetal death, surveillance should begin early for women with preexisting diabetes. Testing for anomalies includes multiple marker screening to identify possible neural tube or other open defects and for possible chromosome abnormalities. Testing also includes performing ultrasonography and fetal echocardiography at 20 to 22 weeks to determine the integrity of the fetal body and cardiac structure (Cunningham et al., 2010; Moore & Catalano, 2009).
During the third trimester, the care goal is to identify markers that suggest a worsening intrauterine environment with a higher probability of fetal death. Surveillance may include maternal perception of fetal movement, biophysical profiles, and nonstress or contraction stress tests. Ultrasound is also used to document fetal growth rates and estimate amniotic fluid volume. Doppler velocimetry may identify if vascular complications exist or if hypertension develops. See Chapter 15 for a description of fetal surveillance methods.
Therapeutic Management
The goals of therapeutic management for a pregnant woman with diabetes are to (1) normalize and maintain maternal blood glucose levels as near normal as possible, (2) increase the likelihood that the baby will be born healthy, and (3) avoid accelerated impairment of maternal blood vessels and other major organs. Pregnant women with diabetes are cared for by a team, which may include a diabetologist, who assists in regulation of maternal blood glucose; an obstetrician, who monitors the mother and fetus and determines the optimal time for birth; a registered dietitian (RD) or registered dietary technician (DTR), who provides a balanced meal plan that considers the woman’s individual needs; and a diabetes educator, often a nurse, who provides ongoing education and support as the therapy changes during pregnancy. The team is completed by a neonatologist, who will care for the newborn, and by the family physician and the pediatrician, who will provide ongoing care for the infant and mother after birth. A maternal-fetal medicine specialist and support staff may be added if multiple fetal evaluation procedures are needed.
Preconception Care
Ideally, care should begin before conception. Both prospective parents should participate in care sessions to learn more about the following issues if diabetes exists before pregnancy:
Diet
Diet recommendations are individualized during a diabetic pregnancy. The average recommended caloric intake for the pregnant diabetic woman of normal weight is 30 kcal/kg/day. Approximately 40% to 45% of the calories should be from carbohydrates, 12% to 20% from protein (approximately 60 g), and up to 40% from fat. Caloric intake should be distributed among three meals and two or more snacks. The bedtime snack should include a complex carbohydrate and protein. Women who are overweight or underweight usually have lower or higher caloric goals.
Self-Monitoring of Blood Glucose (SMBG)
The best frequency for SMBG has not yet been established. One common testing regimen requires obtaining fasting and 2-hour postprandial levels. Another includes testing six times per day: a fasting capillary glucose, 1 to 2 hours after breakfast, before and after lunch, before dinner, and at bedtime. One study found that the postprandial levels were most effective at predicting fetal macrosomia and other adverse outcomes (Moore & Catalano, 2009). In addition to regular monitoring, the woman should also perform a glucose test whenever she experiences symptoms of hypoglycemia. She should record all test results to a log for review by the health care provider at each visit. Most glucometers for SMBG have a memory to provide accurate recall of times and glucose levels and to verify accuracy of log book entries.
Insulin Therapy
Maintaining rigorous control of maternal metabolism during pregnancy requires more frequent doses of insulin than usual. Most treatment regimens rely on three daily injections, with a combination of short-acting (regular) insulin and intermediate-acting (neutral protein Hagedorn [NPH]) insulin given before breakfast, regular insulin before dinner, and NPH insulin at bedtime. Lispro and aspart (Humalog and NovoLog, respectively) insulins act rapidly and should be injected just before a meal. The rapid-acting insulins have been shown to control postprandial hyperglycemia with less between-meal hypoglycemia (Moore & Catalano, 2009). Because placental hormones cause insulin needs to change throughout pregnancy, insulin coverage will need to be adjusted as pregnancy progresses.
First Trimester
Insulin needs generally decline during the first trimester because the secretion of placental hormones that are antagonistic to insulin remains low during this time. The woman may also experience nausea, vomiting, and anorexia, resulting in reduced food intake, and thus may need less insulin. Moreover, the fetus receives its share of glucose, which reduces maternal plasma glucose levels and decreases the need for maternal insulin.
Second and Third Trimesters
Insulin needs increase markedly during the second and third trimesters, when placental hormones reach their peak and result in greater maternal resistance to the effects of insulin. The nausea of early pregnancy usually resolves, and the woman needs additional calories per day to meet the increased metabolic demands of pregnancy.
During Labor
Maintenance of tight maternal glucose control during birth is desirable to reduce neonatal hypoglycemia. Continuous infusion of a regular insulin solution combined with a separate intravenous solution containing glucose, such as 5% dextrose in Ringer’s lactate, allows titration to maintain blood glucose levels between 80 and 110 mg/dL. The insulin infusion rate is raised, lowered, or discontinued to maintain euglycemia based on hourly capillary blood glucose levels. If blood glucose levels remain too high, the insulin infusion is adjusted, and the primary infusion is changed to one without glucose.
Women with type 2 or GDM that has been controlled by diet during pregnancy can usually maintain normal glucose levels during labor if intravenous solutions containing glucose are avoided (Cunningham et al., 2010; Fraser & Farrell, 2011; Moore & Catalano, 2009).
Postpartum
Insulin needs should fall rapidly after delivery of the placenta and the abrupt cessation of placental hormones. Blood glucose levels should be monitored at least four times daily, however, so that the insulin dose can be adjusted to meet individual needs. However up to one third of women who had GDM will have diabetes or impaired glucose metabolism that need either lifestyle modifications or medication. Postpartum screening at 6 to 12 weeks is recommended to identify long-term health promotion needs for these women. Women who had GDM have a 35% to 60% chance of developing diabetes in the next 10 to 20 years after birth (ACOG, 2009b; CDC, 2011b).
Timing of Delivery
If possible, the pregnancy should be allowed to progress to 39 weeks or later to allow fetal lungs to mature, reducing the risk for neonatal respiratory distress syndrome. With evidence of fetal compromise, such as nonreassuring biophysical profile or reduced amniotic fluid, prompt delivery may be needed. Amniocentesis is usually done if delivery is considered for non-emergency reasons before completion of 38 weeks because fetal lung maturation may be slower than in nondiabetic pregnancies.
Gestational Diabetes Mellitus
Risk Factors
GDM is a carbohydrate intolerance of variable severity that develops or is first recognized during pregnancy. Some women diagnosed with GDM may actually have unrecognized type 2 diabetes. Factors associated with a higher risk for GDM are similar to those for type 2 diabetes (ACOG, 2010d; ADA, 2011a).
Identifying Gestational Diabetes Mellitus
All pregnant women should be screened by identification of a history or risk factors that are consistent for GDM or by blood glucose testing. A common prenatal screening test is the glucose challenge test (GCT) administered between 24 and 28 weeks. An oral glucose tolerance test may be used as the initial test if a woman is at high risk for GDM but is more likely to be used as a diagnostic test when abnormally high GCT results occur. Women with a fasting glucose level greater than 126 mg/dL or a nonfasting level of more than 200 mg/dL meet the criteria for GDM, and no added testing is needed (ACOG, 2010d; ADA, 2011a; Cunningham et al., 2010; Moore & Catalano, 2009).
Glucose Challenge Test
Fasting is not necessary for a GCT, and the woman is not required to follow any pretest dietary instructions. The woman should ingest 50 g of oral glucose solution; 1 hour later a blood sample is taken. If the blood glucose concentration is 140 mg/dL or greater, a 3-hour oral glucose tolerance test (OGTT) is recommended. Some practitioners use a lower cutoff of 130 or 135 mg/dL to identify more women at risk (ACOG, 2010d; ADA, 2011a; Cunningham et al., 2010; Moore & Catalano, 2009).
Oral Glucose Tolerance Test
The OGTT is the gold standard for diagnosing diabetes, but it is a more complicated test. After a fasting plasma glucose level is determined, the woman should ingest 100 g of oral glucose solution. Plasma glucose levels are then determined at 1, 2, and 3 hours. GDM is the diagnosis if the fasting blood glucose level is abnormal or if two or more of the following values occur on the OGTT (ACOG, 2010d; ADA, 2011a):
Maternal, Fetal, and Neonatal Effects
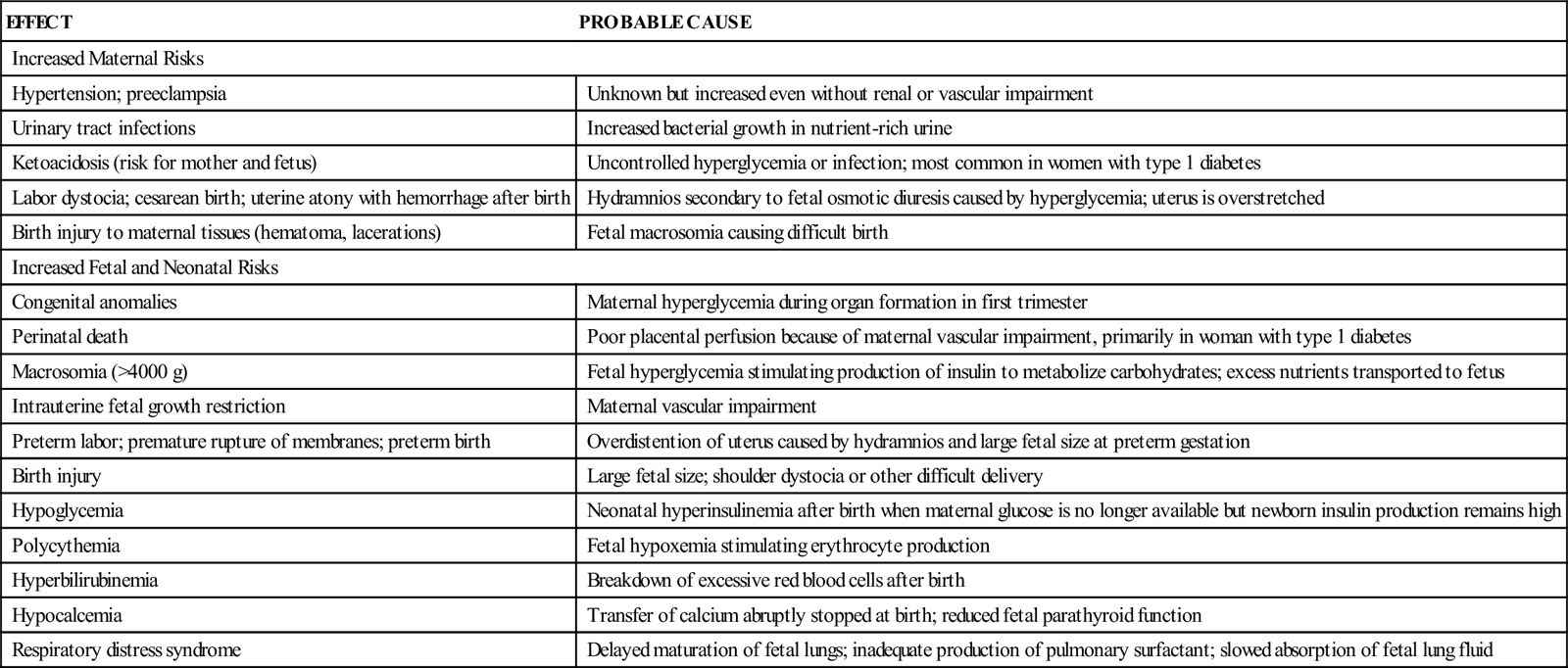
With a few important exceptions, the effects of GDM are similar to those associated with preexisting type 2 diabetes. Because GDM develops after the first trimester, which is the critical period of major fetal organ development (organogenesis), it is not usually associated with an increase in the incidence of major congenital abnormalities. Nevertheless, poorly controlled GDM, with maternal hyperglycemia during the third trimester, is associated with fetal macrosomia and neonatal hypoglycemia. Hypocalcemia, hyperbilirubinemia, and respiratory distress also may occur. Table 26-1 summarizes maternal, fetal, and neonatal effects of diabetes mellitus and their probable causes.
TABLE 26-1
MAJOR EFFECTS OF DIABETES MELLITUS ON PREGNANCY
| EFFECT | PROBABLE CAUSE |
| Increased Maternal Risks | |
| Hypertension; preeclampsia | Unknown but increased even without renal or vascular impairment |
| Urinary tract infections | Increased bacterial growth in nutrient-rich urine |
| Ketoacidosis (risk for mother and fetus) | Uncontrolled hyperglycemia or infection; most common in women with type 1 diabetes |
| Labor dystocia; cesarean birth; uterine atony with hemorrhage after birth | Hydramnios secondary to fetal osmotic diuresis caused by hyperglycemia; uterus is overstretched |
| Birth injury to maternal tissues (hematoma, lacerations) | Fetal macrosomia causing difficult birth |
| Increased Fetal and Neonatal Risks | |
| Congenital anomalies | Maternal hyperglycemia during organ formation in first trimester |
| Perinatal death | Poor placental perfusion because of maternal vascular impairment, primarily in woman with type 1 diabetes |
| Macrosomia (>4000 g) | Fetal hyperglycemia stimulating production of insulin to metabolize carbohydrates; excess nutrients transported to fetus |
| Intrauterine fetal growth restriction | Maternal vascular impairment |
| Preterm labor; premature rupture of membranes; preterm birth | Overdistention of uterus caused by hydramnios and large fetal size at preterm gestation |
| Birth injury | Large fetal size; shoulder dystocia or other difficult delivery |
| Hypoglycemia | Neonatal hyperinsulinemia after birth when maternal glucose is no longer available but newborn insulin production remains high |
| Polycythemia | Fetal hypoxemia stimulating erythrocyte production |
| Hyperbilirubinemia | Breakdown of excessive red blood cells after birth |
| Hypocalcemia | Transfer of calcium abruptly stopped at birth; reduced fetal parathyroid function |
| Respiratory distress syndrome | Delayed maturation of fetal lungs; inadequate production of pulmonary surfactant; slowed absorption of fetal lung fluid |
Therapeutic Management
Diet
Ideally, an RD, RDT, or diabetes educator determines the dietary needs for the woman with GDM. The diet should provide the calories and nutrients needed for maternal and fetal health, result in euglycemia, avoid ketosis, and promote appropriate weight gain. Calories should be distributed in a way similar to that for preexisting diabetes. Simple sugars found in concentrated sweets should be eliminated from the diet. The obese woman may be prescribed a diet with a smaller percentage of carbohydrates than the woman of normal weight. Carbohydrates should be adequate to prevent ketosis in all women. Calories should be divided among three meals and at least three snacks (ACOG, 2010d; Bowers, Curran, Freda, et al., 2008; Moore & Catalano, 2009).
Exercise
Exercise plays a significant role in managing blood glucose levels in women who develop GDM and in women with type 2 diabetes who become pregnant. The exercise regimen should be recommended by a physician who takes into account each woman’s risk factors and risks to the fetus. Regular exercise improves glucose metabolism, offers cardiorespiratory benefits, and aids in weight control. See www.diabetes.org for information about diabetes and its control (Bowers et al., 2008; Franz, 2012).
Glucose Level Monitoring
As in care of the woman with preexisting diabetes, SMBG levels help guide diet and insulin therapy (see p. 615). A common method is measurement of fasting glucose (no food for the previous 4 hours) and 2-hour postprandial blood glucose levels (2 hours after each meal). If blood glucose levels repeatedly exceed 95 mg/dL or postprandial values exceed 120 mg/dL, insulin is started. Additional checks for glucose levels may be performed if needed.
Fetal Surveillance
Antepartum surveillance to identify fetal compromise (see Chapter 15) may begin as early as 28 weeks of gestation if the woman has poor glycemic control or by 34 weeks in lower-risk women with GDM. Surveillance testing may include maternal “kick counts,” ultrasound assessment of fetal growth and amniotic fluid volume, biophysical profile, nonstress or contraction stress tests, or amniocentesis for fetal lung maturity.
Nursing Considerations
The care of a pregnant woman with diabetes mellitus focuses primarily on helping her maintain normal blood glucose levels and optimum fetal condition. Some women respond calmly to the intense medical supervision. Other women may respond with anxiety, fear, denial, or anger and may feel unable to control the diabetes to the degree expected by the health care team. Still others fear for their own or their baby’s health, especially when the diagnosis is a new one.
Increasing Effective Communication
Nurses must ask specifically about the feelings and concerns the woman and her family have about the pregnancy to help the woman avoid unnecessary guilt, anxiety, or frustration and thus promote her active participation in her plan of care.
The nurse must be an active listener and allow time for the woman and her family to express concerns and feelings. The nurse must convey acceptance of feelings that are expressed, whether they are negative or positive. Sharing emotions may help promote positive feelings about her ability to participate successfully in her plan of care.
Providing Opportunities for Control
Providing ways for the woman to make decisions increases her sense of control. For example, she can select foods from the exchange list that provide the necessary nutrients but still give choices. A dietitian should be consulted if the list does not include food the woman likes or that suits her ethnic or cultural preferences. The woman should be assisted to develop a regular schedule of exercise and sleep that helps maintain good blood glucose control. Nurses should allow as much flexibility as possible when scheduling stressful events, such as fetal monitoring tests and amniocentesis.
Providing Normal Pregnancy Care
A woman with diabetes also needs to know about the normal aspects of pregnancy. The nurse caring for a woman with diabetes should provide education and counseling regarding normal pregnancy changes and discomforts.
Nursing Care
The Pregnant Woman with Diabetes Mellitus
Assessment
Determine how well the woman understands the prescribed management and how she plans to carry out the recommended regimen. She may be newly diagnosed and may have no experience in the necessary skills and procedures, or she may be skilled in monitoring glucose levels and administering insulin but may have no knowledge of the effects of diabetes on pregnancy or the effects of pregnancy on diabetes management. If she has used an oral hypoglycemic to manage her glucose, she may be unfamiliar with the use of insulin for that purpose.
To determine whether her techniques are accurate, ask the pregnant woman to demonstrate how she monitors her blood glucose level and observe how she mixes and injects insulins. Verify that she and her family are aware of the need to select appropriate sites and injection techniques.
Although diet is often prescribed individually, it is necessary to assess how well the family understands the diet. Determine if special problems with food preferences or availability of recommended foods exist. Diet recommendations include a target number of calories, plus targets for grams of carbohydrate, protein, and fat to meet calorie needs. Any of several methods to count and exchange foods may be used. One method uses exchange lists, in which the listed foods all have about the same grams of carbohydrate, protein, and fat. Thus one food from the list may be substituted, or exchanged, for another in the same list. Another method uses carbohydrate counting, in which foods on the starch, fruit, or milk list supply about 15 g of carbohydrate, or one carbohydrate choice. The diet plan would prescribe the number of carbohydrate choices for each meal and snack. Insulin is often adjusted according to the carbohydrate count for each meal or snack.
Identify the woman’s knowledge of potential complications, such as hypoglycemia and hyperglycemia, so that she and her family can be provided with pertinent information to avoid and treat it.
Explain why greater fetal surveillance is often ordered. Some women are highly motivated to continue the treatment regimen when test results indicate the fetus is thriving. Other women find the frequent testing stressful and inconvenient.
Nursing Diagnosis and Planning
One of the most common nursing diagnoses is either:
Expected Outcomes
• Demonstrate competence in SMBG and administration of insulin before home management is initiated.
• Describe a plan for meeting dietary recommendations that fits family lifestyle and food preferences.
• Identify signs and symptoms of hypoglycemia and hyperglycemia and the management required for each.
• Verbalize knowledge of fetal surveillance procedures and keep scheduled appointments for testing.
Interventions
Although management of diabetes mellitus during pregnancy is a team effort, a nursing responsibility is to provide accurate information about the recommended therapeutic regimen and to offer consistent support for the woman’s efforts to comply with the recommendations. It may be necessary to demonstrate specific skills that the woman and her support person must master and to review and reinforce information that comes from other members of the health care team.
Teaching Self-Care Skills
Demonstration and return demonstration are effective ways to teach and evaluate psychomotor skills. The woman (and her family) must learn to use a meter and obtain a small sample of blood to determine glucose levels and to correctly mix and inject insulin. Both procedures are invasive and cause mild discomfort, which may make the woman reluctant to start. Mixing insulins accurately or using a sliding scale may be intimidating at first. Using food exchanges is often unfamiliar to the woman who is newly diagnosed, but it is critical to glucose control. Acknowledge these feelings before teaching begins. As needed, reinforce skills taught. Also give positive reinforcement when the woman demonstrates these skills successfully.
Self-Monitoring of Blood Glucose
Spring-loaded lancets make home blood glucose monitoring easier. The side of the fingertip is less sensitive than the pad and may be less uncomfortable. Teach her to cleanse the area with warm water before obtaining a sample to prevent infection. The first drop of blood is wiped away, and the second drop is used to place blood on the meter’s strip. Each home monitoring kit contains specific instructions for use of the meter. Teach her how to record glucose values in a handwritten log. Teach her that glucometers have a memory option for retrieval of previous glucose readings.
Insulin Administration
The woman is often prescribed a combination of short- and intermediate-acting insulins. Teach the woman the difference in onset, peak, and duration of action of each type of insulin. She also needs to learn how to mix the two insulins in the same syringe. If she will use a sliding scale to keep glucose levels close to normal, she will need teaching about how to determine the additional dose of insulin if she has never used sliding scale insulin administration.
Insulin is administered subcutaneously. Common sites include the upper thighs, abdomen, and upper arms. Because the pregnant woman is injecting insulin frequently, emphasize these precautions:
• The needle should be inserted quickly to minimize discomfort.
• Aspirating when injecting into subcutaneous tissue is not necessary.
Emphasize the importance of administering the correct dosage at the correct time. Teach the woman and her family the function of insulin and the importance of following the directions of her physician in regard to coordinating meals with the administration of insulin.
Continuous Subcutaneous Insulin Infusion
Many women who have preexisting diabetes use continuous subcutaneous insulin infusion and wish to continue this method during pregnancy. The programmable insulin infusion pump allows tailoring of insulin administration to the woman’s individual lifestyle. Prompt emergency counseling and assistance must be available 24/7 to deal with unexpected problems such as pump malfunction.
Teaching Dietary Management
A dietitian prescribes the recommended diet, and the nurse must be aware of the general requirements and sensitive to the expectant mother’s dietary habits and preferences. Often, reviewing and clarifying how exchange lists are used to plan meals and snacks are needed. Encourage the woman to avoid simple sugars (candy, cake, cookies, juice), which raise the blood glucose levels quickly but may result in wide swings between high and low levels.
It may be necessary to help the woman select foods high in nutrients but low in cost or to meet cultural or religious constraints. Animal protein is especially expensive, and alternative sources of protein (beans, peas, corn, grains) can be substituted to meet some of the protein needs as well as provide high-quality carbohydrate and fiber.
Recognizing and Correcting Hypoglycemia and Hyperglycemia
Every woman and her family must be aware of the signs and symptoms that indicate abnormal blood glucose levels. If they are not identified and corrected quickly, hypoglycemia and hyperglycemia pose a threat to mother and fetus.
Hypoglycemia
Treat hypoglycemia at once to prevent damage to the brain, which depends on glucose. The woman should take 15 g of carbohydrate if she can swallow. Retest after 15 minutes. If the level is less than 70 mg/dL, repeat carbohydrate intake and retest every 15 minutes until the blood glucose level returns to normal (Franz, 2012). Examples of foods containing 15 g of carbohydrate include:
• Three glucose tablets, depending on their carbohydrate content; oral glucose gel
• 4 to 6 ounces fruit juice or regular soft drink
Family members should read instructions on commercial glucose preparations. Teach family members to inject glucagon if the woman cannot retain oral glucose or food and to notify the physician at once. Intravenous glucose will be given if the woman is hospitalized. If untreated, hypoglycemia can progress to seizures and death.
To prevent episodes of hypoglycemia, instruct the woman to have meals at a fixed time each day and to plan snacks at the recommended times. Suggest that she carry glucose tablets or dry crackers whenever possible.
Hyperglycemia
Because infection is the most common cause of hyperglycemia in a woman with preexisting diabetes, pregnant women must be instructed to notify the physician whenever they have an infection of any type.
Untreated hyperglycemia can lead to ketoacidosis, coma, and maternal and fetal death. If signs and symptoms occur, notify the physician at once so that treatment can be initiated for hyperglycemia and any underlying infection. Hospitalization is necessary for monitoring blood glucose levels and intravenous administration of insulin and antibiotics if needed.
Explaining Procedures, Tests, and Plan of Care
Explain the schedule and the reasons for frequent checkups and tests. Encourage the woman and her family to ask questions if any part of the schedule is confusing. Pregnant women and their families need to know why frequent antepartum surveillance tests are necessary. They should know that their diabetic care will take more time and effort than it did before pregnancy but that this care greatly improves the likelihood that they will have healthy infants.
Evaluation
After the procedures, tests, and plan of care have been explained, the family should be evaluated.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


