36 Care of the vascular surgical patient
Aneurysm: A localized abnormal dilation, distention, or sac in an artery. True aneurysm involves all arterial layers. Dissecting aneurysm allows blood to dissect between the vessel layers. False aneurysm (pseudoaneurysm) develops as a result of disruption of the vessel wall that allows blood to escape from the lumen into a contained sac.4
Angiography (Arteriography): The injection of radiopaque dye into the arteries followed by rapid sequential radiographs of the vascular tree for determination of abnormalities in a specific region.5
Atherectomy: Procedure performed with a special catheter that contains a shaver device at the distal tip. The rotating blade shaves the plaque from the inner lining and removes it from the vessel through a suction device.5
Bypass: Performed to reroute blood flow around an area of stenosis in a blood vessel.5
Cryoplasty: A technique that uses cooling and balloon angioplasty to open stenotic vessels.6
Embolectomy: Extraction of an embolus from an artery to restore blood flow.4
Endarterectomy: Surgical removal of atheromatous plaque from a stenotic vessel.5
Endograft: Device designed to exclude an area of a blood vessel and provide a new conduit through which blood flows. It is primarily used to exclude aneurysmal vessels.7
Fibrinolytic (Thrombolytic) Therapy: Technique that uses clot-dissolving agents to dissolve clot material in a blood vessel.7
Ischemia: Lack of adequate blood flow to an area to meet the needs of the tissues.8
Ligation: Transection and tying off of a blood vessel.8
Stent: Device made of metal or other material used to maintain patency of a blood vessel after angioplasty. The device may be balloon expandable or self expanding.7
Sympathectomy: Interruption of the sympathetic nerve chain performed to produce vasodilation of blood vessels distal to the surgical site.7,9
Thrombectomy: Surgical removal of a thrombus.7
Thrombus: Stationary blood clot or atheromatous plaque that partially or totally occludes a blood vessel.7
Transluminal Angioplasty: Use of a special catheter with a balloon at the distal tip that is passed through the vessel to area of stenosis and inflated to compress stenosis and widen the vessel lumen. The balloon is deflated before removal of the catheter from the vessel. The procedure may be done percutaneously, with only a puncture site, or open through an incision in the vessel, and may be done in conjunction with a stent, atherectomy, or cryoplasty.5
Peripheral arterial disease (PAD) is a “diverse group of disorders that lead to progressive stenosis or occlusion, or aneurysmal dilatation of the aorta and its noncoronary branch arteries.”1,2 PAD is the preferred clinical term, and the definition includes the carotid arteries, upper extremity, visceral and lower extremity arterial branches.1,2 It is estimated that PAD affects 8 million people in the United States.2 Men are more affected than women, and the disease is age-related. As many as 20% of the population older than 70 years may have PAD.2 The consequences of PAD include a decrease in quality of life with reduction in everyday activities and a greater risk of cardiovascular morbidity and mortality.2 Treatment is aimed at prevention or reduction of long-term complications.
Venous insufficiency is estimated to affect as many as 25% of women and 15% of men and is a chronic, debilitating disease that consumes much of the health care budget.3 The treatment of venous disease is aimed at preventing long-term complications such as chronic edema and ulceration.
General considerations
Persons with PAD commonly have other underlying disease processes, some of which may contribute to the development of disease and increase the morbidity and mortality rates associated with surgery. Among the underlying conditions are chronic tobacco use with chronic obstructive pulmonary disease, diabetes, peripheral neuropathy, hypertension, dyslipidemia, obesity, and advancing age.2
The progression of atherosclerosis, which leads to most vascular surgical procedures, is a systemic disease that affects all arterial beds, including those in the extremities, heart, kidneys, and brain.2 A vital part of treatment of arterial disease is risk factor modification to include smoking cessation and control of underlying comorbidities such as diabetes, hypertension, and dyslipidemia.10 Venous surgical procedures have also increased in number as a result of improvements in technology and the use of minimally invasive techniques.3 This chapter is limited to care of the patient undergoing surgery on blood vessels outside the heart.
Diagnostic procedures
Noninvasive diagnostics, such as the ankle-brachial index (ABI), ultrasonography, computed tomography angiogram, magnetic resonance angiography and imaging, have contributed greatly to the early treatment of vascular disease.5,7 These procedures do not usually require sedation.
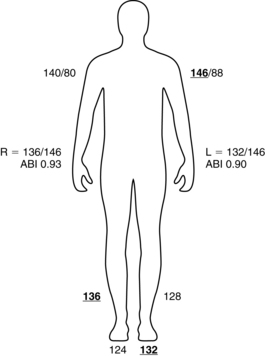
The ABI is a ratio of ankle to brachial blood pressure. PAD is present if the ABI is 0.90 or less.2 Box 36-1 describes the calculation of an ABI. This test may also be used postoperatively to assess patency of vessels following stent placement, percutaneous transluminal angioplasty (PTA), bypass graft placement, or endarterectomy. In patients with diabetes who have developed calcification of the large vessels, an ABI might not be obtainable because of the inability to fully compress the vessels to obtain a pressure. In these patients, toe-brachial index is a useful tool for assessing distal blood flow.8
BOX 36-1 Obtaining an Ankle Brachial Index
1. Obtain the blood pressure in both arms with the patient lying supine.
2. Obtain the blood pressure with a Doppler scan at the dorsalis pedis and posterior tibial pulses on each ankle.
3. Calculate the ABI with the highest ankle pressure divided by the highest arm pressure. (Example: Right BP 140/80 mm Hg; left BP 146/88 mm Hg; right DP 136 mm Hg; right PT 124 mm Hg; left DP 128 mm Hg; left PT 132 mm Hg; right ABI = 136 mm Hg/146 mm Hg [0.93]; left ABI = 132 mm Hg/146 mm Hg [0.90].)
Arteriography continues to be the gold standard for invasive diagnostic testing and usually requires intravenous (IV) sedation. This test requires direct injection of contrast media into the arterial bed and is used to examine the arterial supply of a specific region. Arteriography can also be done in conjunction with various treatment methods, including angioplasty, cryoplasty, atherectomy, and stenting.5 Computed tomographic angiography (CTA) and magnetic resonance angiography (MAR) may also be used in the diagnosis of vascular disease.8
Interventional procedures
Interventional treatments for peripheral vascular disease include PTA, PTA with stent placement, atherectomy, cryoplasty, and fibrinolytic therapy.5 These endovascular procedures are performed by a variety of specialists, including interventional radiologists, vascular surgeons, and cardiologists. These procedures may require only local anesthetic and IV sedation depending on the patient’s condition and physician’s preferences. The procedures can also be performed alone or in conjunction with other vascular surgical procedures in the operating room and may be done with local plus IV sedation, epidural, spinal, or general anesthesia.7
Percutaneous transluminal angioplasty may be performed on carotid, aortic, mesenteric, renal, iliac, femoral, popliteal, and tibial vessel stenosis. This procedure may be used alone or in conjunction with stent placement. Major complications after PTA or stenting include bleeding, hematoma, thrombus formation, and intimal tears (disruption of the inner lining of the vessel).5 Other complications may occur specific to the vascular bed being treated, such as transient ischemic attack or stroke for carotid stenting11 and worsening renal failure in treatment of renal artery stenosis.5 Stents are used to compress and hold the plaque against the vessel wall and are associated with longer patency rates of the vessel.5 They can also be used to treat an intimal tear in the vessel wall. Stents and balloons with drug coating, such as sirolimus and paclitaxel, are being evaluated for their potential to reduce in-stent stenosis as a result of their effects on smooth muscle cell proliferation. The goal is to increase long-term patency rates. Long-term data will help to determine whether these will be an effective tool for PAD.12
Atherectomy is a technique designed for removing plaque from the vessel wall with a special rotating blade and suction apparatus. Angioplasty or stenting may follow atherectomy.13,14 Cryoplasty uses a freezing technique with nitrous oxide inside a balloon for the opening of occluded vessels and theoretically carries the advantage of less risk of intimal hyperplasia, vessel recoil, and dissection.6
After these procedures, patients are monitored for recovery from IV sedation and for bleeding and hematoma formation at the puncture site. Distal pulses are assessed bilaterally to detect any change in blood flow that may be related to formation of an embolism or thrombus for procedures that involve the abdominal vessels or extremities.7 These pulses should be compared with the baseline pulses documented before the procedure. Intake and output should be monitored closely and adequate hydration should be maintained after any procedure with IV contrast. IV contrast can be toxic to the renal system leading to contrast-induced nephropathy; this is characterized by an increase in creatinine of 25% from baseline within 48 hours of contrast administration. This is of greater concern in patients with preexisting diabetic nephropathy.15 Treatments such as additional IV fluids, N-acetylcysteine, diuretics, sodium bicarbonate, and fenoldopam can be used to provide additional protection against contrast-induced nephropathy.15
Bed rest is maintained for 6 to 8 hours after the procedure with the extremity in a straight position to prevent bleeding at the puncture site. If a closure device is used at the puncture site, the patient may be allowed out of bed sooner.5,16 Any patient who undergoes an arteriogram, angioplasty, or stenting that involves the carotid or cranial circulation should undergo frequent neurologic assessment after the procedure. Special protection devices are used during angioplasty and stenting to trap any free-floating particles of plaque or thrombus that may be dislodged during the procedure. These devices serve to minimize postprocedural complications such as stroke.11
Fibrinolytic therapy is used when an embolus or thrombus has occluded a vessel. Special catheters are placed in the area of the thrombus, and agents such as alteplase, tenecteplase, or reteplase are used to lyse the clot.5 This process can be done by initial bolus and then completed via infusion and may take hours for complete lysis of the thrombus. These patients need close observation throughout the infusion for signs of bleeding, bruising, anaphylaxis, hematoma at the puncture site, and hematuria. Blood pressure should be monitored closely to decrease the risk of cerebral hemorrhage.17 Assessing for signs of cerebral bleeding is critical. If treatment is needed, aminocaproic acid (Amicar) can be used to inhibit the fibrinolytic process.5 Frequent assessment of the limb is also needed as reperfusion occurs. As the limb reperfuses, pain may actually worsen initially as microemboli break away from the thrombus and move distally to smaller vessels. As the infusion continues, pain improves as these emboli are dissolved. Frequent laboratory work includes serial monitoring of complete blood count, fibrinogen, prothrombin time and international normalized ratio (PT/INR), and partial thromboplastin time.18 Periodic assessment in radiology is done to follow the progress of the lytic agent. The infusion is discontinued when lysis is complete, fibrinogen levels drop to less than 100, bleeding occurs that necessitates transfusion, or no response to the agent is found.19
Medications used in vascular surgery
Anticoagulants are among the most commonly used medications in the treatment of the patient for vascular surgery. Unfractionated heparin can be administered before, during, and after surgery. Its actions occur at multiple points in the coagulation cascade to ultimately inactivate thrombin and prevent conversion of fibrinogen to fibrin. Heparin has a short, 60- to 90-minute half-life and may be administered IV or subcutaneously. The response to heparin is measured with the activated partial thromboplastin time and is targeted at 1.5- to 2.5-fold greater than normal to obtain a therapeutic response and prevent thromboembolism.20 Complications associated with the use of heparin include increased risk of bleeding and heparin-induced thrombocytopenia (HIT). Platelet counts should be monitored for decrease of 40% to 50% from baseline or any decrease to less than 100,000. If HIT develops, heparin must be discontinued and alternative anticoagulants should be used.20 Protamine is the antidote for heparin; its action occurs within 5 minutes of administration. Care must be taken to avoid overly rapid administration of protamine. When administration is too rapid, side effects can include hypotension, pulmonary hypertension, shortness of breath, and flushing. The usual target dose for reversal is 1 mg of protamine for every 90 units of heparin.20
Low-molecular-weight heparins (LMWHs), such as enoxaparin (Lovenox), dalteparin (Fragmin), and tinzaparin (Innohep), may also be used in the care of the patient for vascular surgery. These drugs are administered subcutaneously and have a significantly lower molecular weight than unfractionated heparin, which gives them improved predictability in the dose response and a longer half-life. This advantage greatly reduces the need for laboratory monitoring. If testing is needed, anti-factor Xa level is the test of choice for monitoring. The LMWHs are administered subcutaneously and have a significantly lower incidence rate of HIT associated with their use; they are primarily used to prevent thromboembolism after surgery, but are also approved in the treatment of deep vein thrombosis (DVT) and pulmonary embolism (PE). They may also be used to bridge patients before and after surgery who require long-term anticoagulation with warfarin (Coumadin). The decision to bridge is based on diagnosis and thromboembolic risk. Patients with renal disease may need a dose reduction depending on the severity of their disease. Complications are similar to those of unfractionated heparin. Laboratory testing should be monitored for signs of HIT, although it occurs much less frequently with the use of LMWH.20
Warfarin is an oral anticoagulant that inhibits vitamin K–dependent coagulation factors and the anticoagulant proteins C and S; it has a half-life of 36 to 42 hours. Monitoring of warfarin is done with the PT and the international normalized ratio.20 The PT/INR is laboratory dependent, and specific methods vary among institutions. Caregivers should be familiar with institutional methods. Warfarin is used to treat a variety of thromboembolic disorders, and it can be used to promote long-term patency of infrainguinal bypass grafts, particularly following thrombosis of previously placed grafts. Complications include increased risk of hemorrhage and skin necrosis. Patients receiving warfarin must be counseled to discontinue the drug several days before any invasive procedure to allow time for the PT/INR levels to decrease to normal. Reversal of warfarin is achieved with vitamin K or fresh-frozen plasma.20
Two additional oral anticoagulants are currently available. Dabigatran (Pradaxa), a direct thrombin inhibitor, and rivaroxaban (Xarelto), a direct factor Xa inhibitor, are currently approved for the prevention of stroke in nonvalvular atrial fibrillation. Rivaroxaban is also approved for short-term thrombophyphylaxis after elective hip or knee surgery.20 Dabigatran has a shorter half-life than warfarin and requires twice daily dosing. Patients receiving dabigatran should be counseled to discontinue its use the day before surgery if renal function is normal. Earlier discontinuation may be needed in patients with creatinine clearance less than 30 mL/min.21 Rivaroxaban is administered 10 mg daily. There are no reversal agents for dabigatran and rivaroxaban.20
IV direct thrombin inhibitors act at the active site of thrombin. These drugs provide an alternative to heparin in the patient with HIT. Current drugs available in this category are lepirudin (Refludan), bivalirudin (Angiomax), and argatroban (Acova).5,20 Desirudin (Iprivask) is a direct thrombin inhibitor that is administered subcutaneously. It may be used in patients who cannot use fondaparinux (Aristra) or LMWH.22
Parenteral factor X inhibitors currently include only fondaparinux (Arixtra). Fondaparinux activates antithrombin III, leading to inactivation of factor X. It is administered subcutaneously and requires no laboratory monitoring. Its primary use is preventing DVT and treating acute coronary syndromes. There is no reversal agent for fondaparinux.20
Antiplatelet agents, such as aspirin, clopidogrel (Plavix), prasugrel (Effient), dipyridamole (Persantine), dipyridamole ER/aspirin (Aggrenox), ticlopidine (Ticlid), and cilostazol (Pletal), may also be used in the patient with vascular disease as a preventative measure for myocardial infarction and stroke or as part of medical management for patients following placement of infrainguinal bypass grafts, carotid endarterectomy, and peripheral and carotid stenting.5,20,23 These drugs exhibit an irreversible permanent effect on the platelet for its lifespan and produce a qualitative effect on the platelet that is measured with the bleeding time. Platelet counts are not affected by these agents. Cilostazol also has some vasodilatory effects and is contraindicated in patients with heart failure. Patients should be counseled regarding the discontinuation of these drugs 7 to 10 days before invasive procedures to decrease the risk of bleeding.20 Patients at moderate to high risk for cardiovascular events may continue their antiplatelet agents to the time of surgery.20
Glycoprotein IIb/IIIa inhibitors are parenteral agents that also interfere with platelet aggregation and include abciximab (Reopro), eptifibatide (Integrilin), and tirofiban (Aggrastat). These agents are administered by IV infusion. Patients who have had drug-eluting coronary stent placement within the previous 12 months of a surgical procedure may need to be bridged with a GP IIb/IIIa inhibitor to discontinue their antiplatelet drugs for surgery or may be instructed to continue their aspirin and clopidogrel (Plavix) up to surgery.20
Perioperative use of beta blockers has been associated with decreased morbidity and mortality in the high-risk vascular surgery patient, particularly those undergoing lower extremity bypass or open abdominal procedures.24,25 There is evidence that, although cardiac events are decreased, there may be an increased risk of stroke.25,26 There is also some indication that atenolol may be associated with reduced mortality compared with metoprolol.27 Incidence of preoperative myocardial infarction ranges from 14% to 17.8% for open abdominal procedures and 5% to 15% for lower extremity bypass procedures.26,28 Preoperative stress testing has assisted in determining a plan of care before surgery, including coronary intervention or beta blockade, and may help to lower the risk of cardiac events postoperatively.29 Women are at particular risk for complications after vascular surgery and also have lower survival rates. Their responses to beta blockade may also be less beneficial than in men.29
Arterial surgical procedures
Extremity vessels and extraanatomic procedures
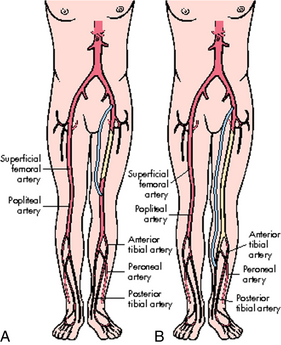
Arterial surgical procedures that involve the extremities include bypass, endarterectomy, embolectomy, and thrombectomy.7 A bypass is performed to reroute blood flow around an area of stenosis and is named for the vessels it arises from and connects into. Examples of these in the lower extremities are femoral-popliteal, femoral-tibial, and femoral-peroneal. The distal anastomosis for femoral-popliteal bypass procedures may be above the knee or below the knee depending on the location of the stenosis (Fig. 36-1). Distal bypasses may also connect into the anterior tibial or posterior tibial artery.7 Bypass procedures may also be performed in the upper extremities, but are much less common and may include bypass to circumvent lesions of the axillary artery, such as carotid-axillary bypass. Extraanatomic bypass procedures route blood in a more unusual fashion and may be done based on a patient’s condition or inability to tolerate a more major procedure. Examples of these types of bypass include axillofemoral, axillary-axillary, and femoral-femoral (Fig. 36-2).7 Endarterectomy can be done alone or in conjunction with a bypass and involves removal of plaque from a stenotic vessel. The most commonly endarterectomized vessels are the carotid, subclavian, iliac, and femoral arteries. Embolectomy and thrombectomy can also be performed to remove a clot from a vessel.7
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree