Traumatic Peripheral Nerve Injuries
Andrea L. Strayer
Joanne V. Hickey
Traumatic peripheral nerve injuries result from a variety of mechanisms with variable clinical presentation and degree of disability. The most common injury is traction placed on a nerve subsequent to a vehicular, predominately motorcycle, crash. Peripheral nerve regeneration, whether occurring spontaneously because the nerve is in continuity or following surgical intervention, is a slow, arduous process that can last years. Mechanisms of injury, nerve anatomy, classification, nerve response to injury, diagnosis, treatment options, and nursing care considerations will be described.
MECHANISMS OF INJURY
Acute traumatic events by which peripheral nerves are injured include: traction and stretching; partial or complete nerve transection;
contusion; compression and ischemia; and less common mechanisms such as electric shock and drug injection. The following describes the injuries.
contusion; compression and ischemia; and less common mechanisms such as electric shock and drug injection. The following describes the injuries.
Stretch injuries result from traction and stretching exerted on the nerve related to incidents such as trauma (i.e., vehicular accidents), falls, sports injuries, or improper positioning in the operating room. In traumatic upper extremity injuries, the head and neck are forcibly moved in the opposite direction of the shoulder and arm, stretching peripheral nerve(s). The peripheral nerve has elastic qualities; however, if stretched too far, injury will occur. With sufficient force, the nerve root(s) can avulse from the spinal cord. In most circumstances, some nerve continuity is maintained. Peroneal nerve injury, an example of lower extremity injury, can occur with excessive application of orthopedic traction weight or knee injury.
Avulsion is a tearing away from a structure or part of a structure by severe traction. This is a particular consideration in brachial plexus injuries where nerve roots can be torn away from the spinal cord. Most root avulsions are irreparable.
A partial or complete transection of a nerve is a structural loss of integrity of some or all of the fascicles of a nerve, usually the result of a severe traumatic laceration nerve injury, such as from glass, knife, metal; equipment such as a saw; bullet; or long bone fracture.
Contusive injuries bruise the nerve, producing a relatively focal lesion. The nerve remains structurally intact, and there may be spontaneous recovery, dependent upon the degree of axonal injury. Forces from gunshot, shrapnel, or other artillery are examples of concussive peripheral nerve injury.
With compression or ischemia, the nerve remains structurally intact, but with undetermined axonal injury. The injury occurs from pressure on the nerve. For example, compression on the radial nerve by falling asleep (often in an intoxicated state) with the upper limb draped over the back or arm of a chair, placing direct pressure on the radial nerve, is commonly referred to as “Saturday Night Palsy.” Total loss of both motor and sensory function may occur, although the nerve is maintained in complete continuity. Both mechanical compression as well as ischemia are believed to contribute to this type of injury. A closed compartment syndrome is an emergency and prompt attention to decompression of the affected nerve to prevent irreversible damage to the nerve and the muscle is warranted.
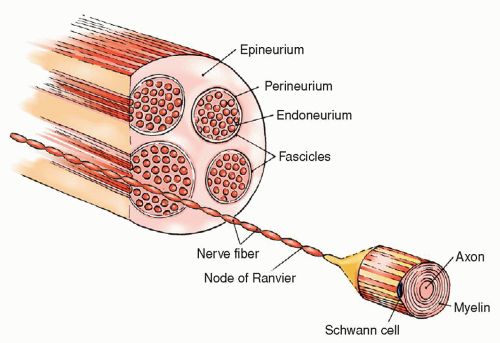
Figure 19-1 ▪ Anatomic components of the peripheral nerve. (From: Benanoch, C., et al. [1998]. Medical neurosciences [4th ed.]. Philadelphia: Lippincott-Raven.)
Electrical or thermal injuries results from a current passing through the peripheral nerve when contact is made with electrical wires. The resulting injuries produce severe muscle and nerve coagulation, in addition to burns of the skin and destruction of bone. Prognosis for muscle reinnervation in these instances is generally poor. Thermal nerve injury causes similar types of local responses, with major damage resulting from burning and necrosis of tissue.
Iatrogenic injuries occur following medical interventions such as lymph node biopsy, mass resections (involvement of the nerve is not appreciated), joint surgeries, venipuncture, drug injections. Drug injections most commonly affect the sciatic and the radial nerves. Injections given directly into the nerve or proximally to it can cause scarring or a neuropathy.
NERVE ANATOMY
Peripheral nerves are composed of many types of nerve fibers and connective tissue. The anatomy of a peripheral nerve includes the endoneurium, which is the connective tissue that surrounds both myelinated and unmyelinated axons. Each unit is grouped with many other axons into bundles of fascicles by the perineurium.
Groups of fascicles are bound together by an outer covering of connective tissue called the epineurium (Fig. 19-1). Nerves are composed of afferent and efferent fibers. These axons can be differentiated on the basis of their size and presence or absence of myelin (i.e., myelinated or unmyelinated fibers). Each nerve fiber is composed of an axon encased in a series of Schwann cells that cover the length of the axon. The junctional points between Schwann cells are called the nodes of Ranvier. As nerves develop, one of the two events can occur. Many unmyelinated axons become embedded in the Schwann cell, or the Schwann cell wraps around one axon in concentric circles to fuse together to form the myelin of the myelinated nerve fiber. The electrical activity of each nerve fiber is independent of the activity in all the other fibers in the nerve even though the fibers are close to each other. The endoneurium and the myelin isolate the action potentials of each fiber from other adjacent fibers.1, 2
Axonal transport is the continuous and regulated flow of material from the cell body to the axons and synaptic terminals as well as in the reverse direction (anterograde and retrograde transport). This action is critical for neuronal function through the transport
of material synthesized in the cell body and dendrites to reach the axon terminals, and for the material in the axon terminals to reach the cell body of the nerve. The resting and action potentials of axons are discussed in Chapter 5.
of material synthesized in the cell body and dendrites to reach the axon terminals, and for the material in the axon terminals to reach the cell body of the nerve. The resting and action potentials of axons are discussed in Chapter 5.
CLASSIFICATION OF PERIPHERAL NERVE INJURIES
Nerve injuries are classified using the Seddon3 or Sunderland4 classification systems. For purposes of this chapter, peripheral nerve injury is based on the simpler Seddon system, which classifies injury according to whether nerve or connective tissue is disrupted. Classification of injury is important to facilitate interpretation of clinical findings, provide guidance for treatment decisions, and suggest prognosis. If neither the nerve nor its connective tissue covering is disrupted, the injury is called a neuropraxia. The mildest injury is a physiologic rather than an actual transection, but the axonal transport is impaired. In addition, no Wallerian degeneration occurs. Recovery may occur in hours to months, with an average time of 6 to 8 weeks. If the neuronal components are disrupted, but the connective tissue through which the nerve travels remains intact, the injury is called an axonotmesis. As a result of stretch and crush injuries, the axon is disrupted and Wallerian degeneration occurs (Fig. 19-2). Regeneration is possible, but it is dependent on the distance to the muscle innervated and the degree of injury to the nerve. If the neuronal and connective tissue elements are completely disrupted, the injury is called a neurotmesis, and any functional recovery depends on appropriate surgical intervention. Neurotmesis is seen with severe traction injuries where there is avulsion or rupture of the nerves or with a sharp, severe laceration injury.5
Nerve Response to Injury
Following injury to a peripheral nerve, a highly regulated sequence of events takes place to remove damaged tissue and begin the process of regeneration. Important distinctions are the changes occurring proximal to the lesion, or injury site, and those distal. The proximal portion of the axon is in continuity with the cell body and begins the regeneration process once a short distance of axon degenerates and associated debris are removed by macrophages. The distal portion is physically separated from the proximal axon and degenerates, termed Wallerian degeneration. The distal stump is unable to regenerate without growth from the proximal segment. (Fig. 19-2) Changes in the cell body, chromatolysis, appears within 24 hours after the axon is injured and progresses until a maximum is reached between the second and third weeks. Chromatolysis usually indicates increased RNA and protein synthesis, an example of metabolic activity necessary for the regeneration of severed axonal fibers. Concurrently, the distal axonal stump undergoes Wallerian degeneration (secondary degeneration). Phagocytic cells remove debris and the only remaining evidence of the severed axonal segment is the Schwann (neurolemma) cells. Muscle degeneration also occurs when innervation is lost and accompanies Wallerian degeneration. In lesions with a partial transection, not all axons are destroyed and some function may remain intact. If a nerve is completely severed, reinnervation is poor because the axonal buds or sprouts have no pathway to follow. They may grow haphazardly, form large abnormal tips, or form a painful neuroma.5 Conversely, if the nerve experiences minor injury, it remains in continuity and no degeneration occurs usually leading to full recovery.
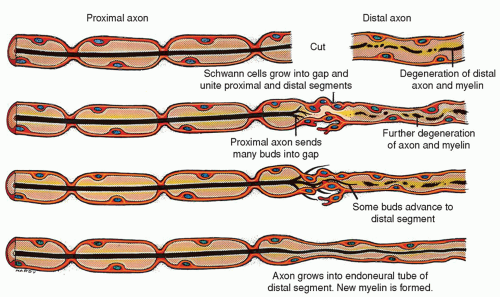 Figure 19-2 ▪ Wallerian degeneration: diagram of changes that occur in a nerve fiber that has been cut and then regenerates. |
If a peripheral nerve has been completely severed, there is still a possibility for regeneration after surgical reapproximation of the severed nerve ends (Fig. 19-1). After surgical repair, the regeneration process begins with the proliferation of Schwann cells in the proximal stump near the transection and in the distal stump. These cells divide by mitosis to form continuous cords of Schwann cells, covering an area that encompasses the proximal stump, the gap across the transected area, the distal stump, and the area up to the sites of the sensory receptors and motor endings. The neurolemma cords act as guidelines for the regenerating axon.
Meanwhile, the cell body is directing synthesized protein and metabolites distally to provide the nutritional machinery for axonal regeneration. The axis cylinder of the proximal axon at the transection begins to generate tiny, unmyelinated sprouts that
grow longitudinally. There may be as many as 50 sprouts. The random growth of buds or sprouts, which is accompanied by connective tissue proliferation, forms an enlargement called a neuroma; this can often be a source of intractable pain. Some sprouts will be misdirected and stray, but some will be successful in crossing the transected gap through the guidance of the neurolemma, finding their way to the distal stump. The rate of growth of a regenerating sprout is estimated at 1 mm/day or 1 in (2.5 cm)/mo. If the union is well aligned so that the axon will grow back into its former channel, functional return will be possible.
grow longitudinally. There may be as many as 50 sprouts. The random growth of buds or sprouts, which is accompanied by connective tissue proliferation, forms an enlargement called a neuroma; this can often be a source of intractable pain. Some sprouts will be misdirected and stray, but some will be successful in crossing the transected gap through the guidance of the neurolemma, finding their way to the distal stump. The rate of growth of a regenerating sprout is estimated at 1 mm/day or 1 in (2.5 cm)/mo. If the union is well aligned so that the axon will grow back into its former channel, functional return will be possible.
Successful regeneration is intricate and dependent on many factors. Obstacles include the patient’s general health and medical comorbidities and the distance to travel to the distal stump. Axons that do not reach the distal stump wander into tissue and become encased in scar tissue. The end organ muscle must receive the beginning of axonal sprouting within 12 to 18 months to be able to accept innervation, and thus experience functional recovery.5 If the nerve realignments are mismatched, functional weakness or no return of any strength, unintentional movements of muscles, and poor sensory discrimination and localization of stimuli may result. Sprouts unsuccessful in making connections degenerate. Nerves proximal to the injured neurons are stimulated to produce collateral innervation to denervated areas. This process provides innervation long before the axon has regenerated to provide innervation. Therefore, some sensory return may occur before regeneration can realistically occur.
DIAGNOSIS OF PERIPHERAL NERVE INJURIES
Diagnosis of plexus and peripheral nerve injury is based on a history of injury or presence of an irritative or injurious lesion and a complete neurological examination. The clinical examination is integral in establishing the type, degree, and site of injury. A motor and sensory examination is conducted, and pain characteristics evaluated. For upper extremity nerve injuries, Tinel’s sign and evidence of Horner’s sign are important. Plain x-rays of the chest (for upper extremity problems), computed tomography (CT) with myelography or magnetic resonance imaging (MRI), electromyography (EMG), and nerve conduction velocity (NCV) studies are diagnostic studies that may be conducted. EMG/NCV studies are not performed until 3 to 4 weeks after injury to allow for completion of Wallerian degeneration, confirming diagnosis, localizing injury site, and elucidating the severity of the injury. Physical examination in conjunction with EMG/NCV demonstrates if reinnervation is taking place or if there is persistent denervation.6
General Signs and Symptoms of Peripheral Nerve Trauma
Motor, sensory, and autonomic deficits and trophic signs and symptoms are the usual changes noted with peripheral nerve injuries. The degree of deficit in any area depends on the type and extent of injury. The general signs and symptoms include the following.
Flaccid paralysis of the muscle or muscle groups supplied by the nerve. A paresis results if some, but not all, of the lower motor neurons innervating the muscle are functional.
Diminished or complete loss of deep tendon reflexes (DTRs). If some neurons are still functional, DTRs are weak. If all neurons are affected, there is absence of DTRs in the affected area.
Atonic or hypotonic muscles.
Progressive muscle atrophy that begins early (reaches peak in several weeks).
Fibrillations and fasciculations that peak 2 to 3 weeks after the muscle is denervated. (Fibrillations are transitory muscle contractions caused by spontaneous stimulation of a single muscle fiber and can only be detected during EMG studies. Fasciculations are spontaneous contractions of several muscle fibers innervated by a single motor nerve filament and can be observed during the physical examination).
Diminished or complete sensory loss.
Changes in sensory perception and possibly dysesthetic pain.
Warm or dry skin anhidrosis (does not perspire) caused by transection of the postganglionic sympathetic fibers.
Trophic skin changes, which can be separated into a warm phase followed by a cold phase, are due to autonomic postinjury dysfunction. The warm phase lasts about 3 weeks, during which time the skin in the affected areas is dry, warm, and flushed. The cold phase is characterized by cold, cyanotic skin; brittle fingernails; loss of hair; dryness and ulceration of skin; and lysis of bones and joints. The digits are affected most frequently. In some incomplete lesions of the median, ulnar, or sciatic nerve with causalgia (see below), the warm phase may persist and may be accompanied by sweating.
Types of Peripheral Nerve Trauma
Common Traumatic Syndromes
The peripheral nerves are the major nerve trunks in the extremities and are derived from the plexuses. Each nerve has a welldefined anatomic course in an extremity and innervates specific areas of the skin and muscles. A plexus is a complex network of axons that come together to form new combinations. The three major plexuses are the brachial, lumbar, and sacral plexuses. Specific traumatic syndromes commonly seen include brachial plexus injuries, upper extremity injuries (medial, ulnar, and radial nerves), and lower extremity injuries (femoral, sciatic, and common peroneal nerves).6
Brachial Plexus Injuries
Anatomic Considerations. The brachial plexus (Fig. 19-3) is created from spinal nerves C-5, C-6, C-7, C-8, and T-1. By a series of division and recombination, three major trunks result: the upper trunk (C-5 and C-6), the middle trunk (C-7), and the lower trunk (C-8 and T-1). Again, these trunks divide and recombine to create three cords that give rise to the following nerves.
Lateral cord (chiefly derived from C-5 and C-6): musculocutaneous and the lateral half of the median nerve
Median cord (chiefly derived from C-8 and T-1): ulnar nerve and the medial half of the median nerve
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Get Clinical Tree app for offline access
