On completion of this chapter the reader will be able to: • Describe the characteristics of infants that affect their ability to adapt to fluid loss or gain. • Formulate a care plan for the infant with acute diarrhea. • Compare and contrast the inflammatory diseases of the gastrointestinal tract. • Describe the nursing care of the child with hepatitis. • Formulate a plan for teaching parents preoperative and postoperative care of the child with a cleft lip or palate. • Formulate a care plan for the child with an obstructive disorder. • Identify nutritional therapies for the child with a malabsorption syndrome. http://evolve.elsevier.com/wong/essentials Animations—Appendicitis; Digestive Tract, Passage of Food; Intestines; Intussusception; Volvulus, Pediatric Case Studies—Acute Diarrhea; Appendicitis; Cleft Lip and Palate; Dehydration; Dehydration and Diarrhea; Gastroenteritis; Gastrointestinal Problems; Hepatitis Nursing Care Plans—The Child with Acute Diarrhea (Gastroenteritis); The Child with Appendicitis; The Child with Cleft Lip and/or Cleft Palate; The Child with Fluid and Electrolyte Disturbances Nurses should be alert for altered fluid requirements in various conditions: Basal maintenance calculations for required body water are based on the body’s requirements for water in a normometabolic state at rest; estimated fluid requirements are then increased or decreased from these parameters based on increased or decreased water losses, such as with elevated body temperature (increased) or heart failure (decreased). Daily maintenance fluid requirements for infants, toddlers, and older children are listed in Table 24-1.These requirements are not appropriate for neonates. TABLE 24-1 DAILY MAINTENANCE FLUID REQUIREMENTS* Infants ingest and excrete a greater amount of fluid per kilogram of body weight than do older children. Because electrolytes are excreted with water and infants have a limited ability for conservation, maintenance requirements include both water and electrolytes. The daily exchange of ECF in infants is greatly increased over that of older children, leaving infants with little fluid volume reserve in dehydrated states. Fluid requirements depend on hydration status, size, environmental factors, and underlying disease. Daily maintenance fluid requirements are shown in Table 24-1. Depletion of ECF, usually caused by gastroenteritis, is one of the most common problems encountered in infants and children. Until modern techniques for fluid replacement were perfected, gastroenteritis was one of the chief causes of infant mortality. Fluid and electrolyte problems related to specific diseases and their management are discussed throughout the book where appropriate. The major fluid disturbances, their usual causes, and clinical manifestations are listed in Table 24-2. Problems of fluid and electrolyte disturbance always involve both water and electrolytes; therefore, replacement includes administration of both calculated on the basis of ongoing processes and laboratory serum electrolyte values. TABLE 24-2 DISTURBANCES OF SELECT FLUID AND ELECTROLYTE BALANCE A number of clinicians have reported water intoxication in children after swimming lessons. Although they hold their breath, some children apparently swallow a large amount of water during repeated submersion. Anticipatory guidance to parents should include a discussion of swimming instruction and advice to stop a lesson if the child swallows unusual amounts of water or exhibits any symptoms of hyponatremia (see Table 24-2). Diagnosis of the type and degree of dehydration is necessary to develop an effective plan of therapy. The degree of dehydration has been described as a percentage of body weight dehydrated: mild—less than 3% in older children or less than 5% in infants; moderate—5% to 10% in infants and 3% to 6% in older children; and severe—more than 10% in infants and more than 6% in older children (Greenbaum, 2011). Water constitutes only 60% to 70% of an infant’s weight. However, adipose tissue contains little water and is highly variable in individual infants and children. A more accurate means of describing dehydration is to reflect acute loss (time frame of ≤48 hours) in milliliters per kilogram of body weight. For example, a loss of 50 ml/kg is considered to be a mild fluid loss, but a loss of 100 ml/kg produces severe dehydration. Weight is the most important determinant of the percent of total body fluid loss in infants and younger children. However, often the pre-illness weight is unknown. Other predictors of fluid loss include a changing level of consciousness (irritability to lethargy), altered response to stimuli, decreased skin elasticity and turgor, prolonged capillary refill (>2 sec), increased heart rate, and sunken eyes and fontanels. Clinical signs provide clues to the extent of dehydration (Table 24-3). The earliest detectable sign is usually tachycardia followed by dry skin and mucous membranes, sunken fontanels, signs of circulatory failure (coolness and mottling of extremities), loss of skin elasticity, and prolonged capillary filling time (Table 24-4). TABLE 24-3 EVALUATING EXTENT OF DEHYDRATION *These signs are less prominent in patients who have hypernatremia. Data from Jospe N, Forbes G: Fluids and electrolytes—clinical aspects, Pediatr Rev 17(11):395–403, 1996 and Steiner MJ, DeWalt DA, Byerly JS: Is this child dehydrated? JAMA 291(22):2746–2754, 2004. TABLE 24-4 CLINICAL MANIFESTATIONS OF DEHYDRATION • The degree of dehydration based on physical assessment • The type of dehydration based on the pathophysiology of the specific illness responsible for the dehydrated state • Specific physical signs other than general signs • Initial plasma sodium concentrations • Serum bicarbonate concentration (CO2) • Any associated electrolyte (especially serum potassium) and acid–base imbalances (as indicated) Laboratory data are said to be useful only when results are significantly abnormal (Emond, 2009). Urine specific gravity, urine ketones, and urinary output during rehydration are reportedly unreliable assessments for determining dehydration in children (Steiner, Nager, and Wang, 2007). Shock, tachycardia, and very low blood pressure are common features of severe depletion of ECF volume (see Shock, Chapter 25). Medical management is directed at correcting the fluid loss or deficit and treating the underlying cause. When the child is alert, awake, and not in danger, correction of dehydration may be attempted with oral fluid administration. Mild cases of dehydration can be managed at home by this method. Several commercial rehydration fluids are available for use (Table 24-5). Oral rehydration management consists of replacement of fluid loss over 4 to 6 hours, replacement of continuing losses, and provision for maintenance fluid requirements. In general, a mildly dehydrated child may be given 50 ml/kg of oral rehydration solution (ORS), and a child with moderate dehydration may be given 100 ml/kg of ORS. A child with fluid losses from diarrhea may be given 10 ml/kg for each stool. Amounts and rates are determined from body weight and the severity of dehydration and are increased if rehydration is incomplete or if excess losses continue until the child is well hydrated and the basic problem is under control. TABLE 24-5 COMPOSITION OF SOME ORAL REHYDRATION SOLUTIONS Cl, Chloride; K, potassium; Na, sodium. *Note that many generic products are available with compositions identical to Pedialyte. The child may not be thirsty even though dehydrated and may refuse oral fluids initially for fear of continued emesis (if occurring) or because of decreased strength, oral stomatitis, or thrush. In such children, rehydration may proceed by administering 2 to 5 ml of ORS by a syringe or small medication cup every 2 to 3 minutes until the child is able to tolerate larger amounts; if the child has emesis, administering small amounts (5–10 ml) of ORS every 5 minutes or so may help overcome fluid deficit, and the emesis will often lessen over time. Oral administration of ondansetron (Zofran) to children with acute gastroenteritis and vomiting may reduce emesis and increase time to oral rehydration, thus preventing IV therapy. Oral rehydration therapy (ORT) is effective for treating mild or moderate dehydration in children, is less expensive, and involves fewer complications than parenteral therapy (American Academy of Pediatrics [AAP], Committee on Infectious Diseases and Pickering, 2009). Parenteral rehydration therapy has three phases. The initial therapy is used to expand ECF volume quickly and to improve circulatory and renal function. During initial therapy, an isotonic solution is used at a rate of 20 ml/kg, given as an IV bolus over 20 minutes, and repeated as necessary after assessment of the child’s response to therapy (Ford, 2009; Friedman, 2010). Subsequent therapy is used to replace deficits, meet maintenance water and electrolyte requirements, and catch up with ongoing losses. Water and sodium requirements for the deficit, maintenance, and ongoing losses are calculated at 8-hour intervals, taking into consideration the amount of fluids given with the initial boluses and the amount administered during the first 24-hour period. With improved circulation during this phase, water and electrolyte deficits can be evaluated, and acid–base status can be corrected either directly through the administration of fluids or indirectly through improved renal function. Potassium is withheld until kidney function is restored and assessed and circulation has improved (see Evidence-Based Practice box).
The Child with Gastrointestinal Dysfunction
Distribution of Body Fluids
BODY WEIGHT (kg)
AMOUNT OF FLUID PER DAY
1–10
100 ml/kg
11–20
1000 ml plus 50 ml/kg for each kg >10 kg
>20
1500 ml plus 20 ml/kg for each kg >20 kg
Changes in Fluid Volume Related to Growth
Water Balance in Infants
Fluid Requirements
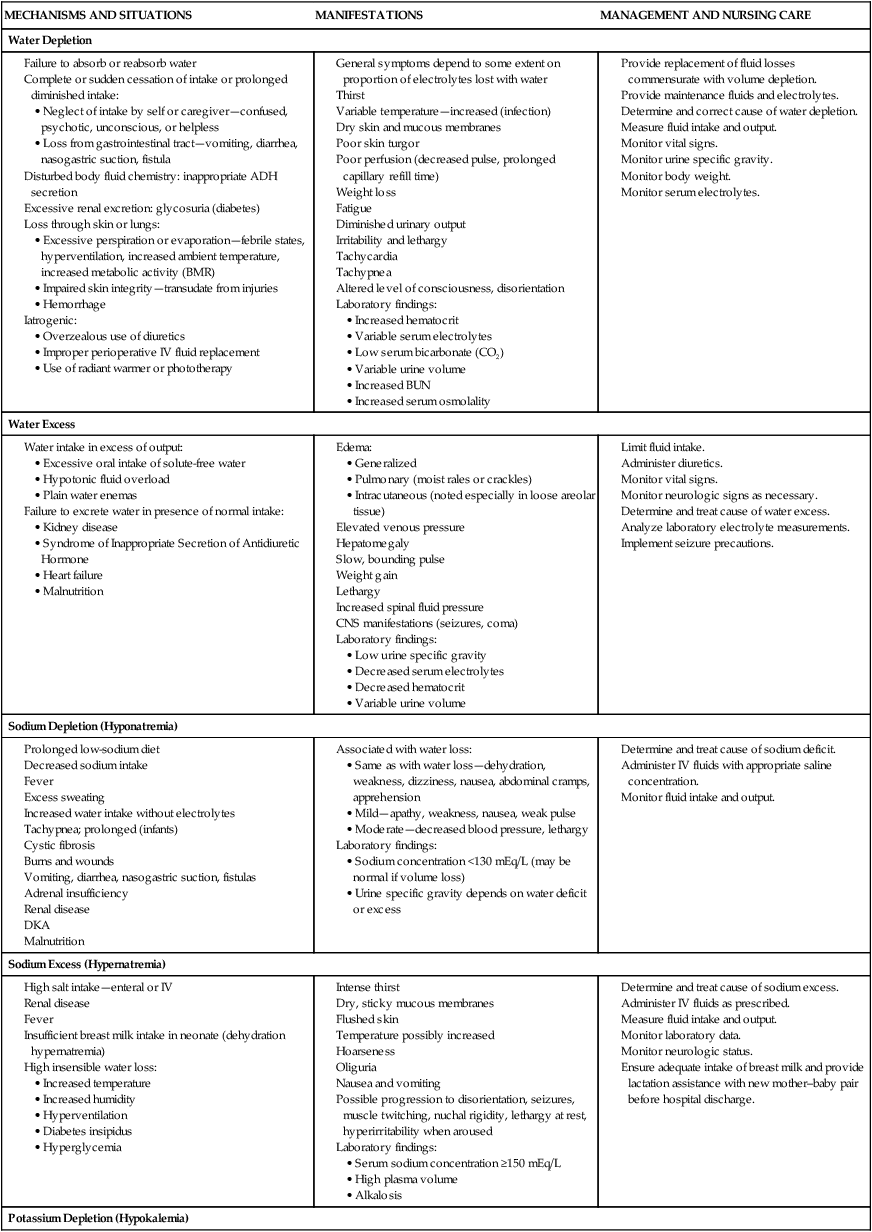
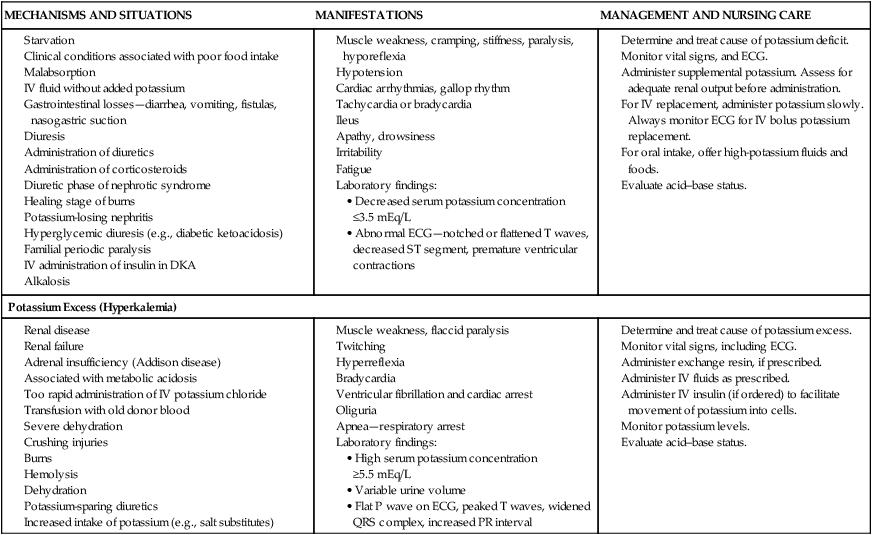
Disturbances of Fluid and Electrolyte Balance
![]() Nursing Care Plan—The Child with Fluid and Electrolyte Disturbances
Nursing Care Plan—The Child with Fluid and Electrolyte Disturbances
Water Intoxication
Dehydration
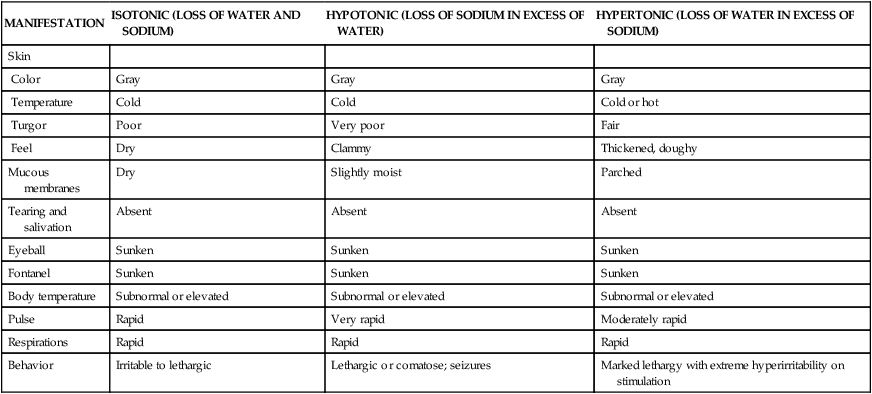
Types of Dehydration
![]() The pathophysiology of dehydration is understood by recognizing that the distribution of water between the ECF and ICF spaces depends on active transport of potassium into and sodium out of cells by energy-requiring processes. Sodium is the chief solute in ECF and is the primary determinant of ECF volume. Sodium is considered a unique electrolyte in that water balance determines sodium concentration; when water is lost and sodium concentration becomes elevated compensatory mechanisms in the kidney stop ADH secretion so water is retained. The thirst mechanism (not fully functional in infants) is also stimulated so water is replaced, thus increasing the total body water content and returning sodium to a normal level (Greenbaum, 2011). Potassium is primarily found inside the cell (intracellular) but small amounts are also found in extracellular fluid. Sodium depletion in diarrhea occurs in two ways: out of the body in stool and into the ICF compartment to replace potassium to maintain electrical equilibrium.
The pathophysiology of dehydration is understood by recognizing that the distribution of water between the ECF and ICF spaces depends on active transport of potassium into and sodium out of cells by energy-requiring processes. Sodium is the chief solute in ECF and is the primary determinant of ECF volume. Sodium is considered a unique electrolyte in that water balance determines sodium concentration; when water is lost and sodium concentration becomes elevated compensatory mechanisms in the kidney stop ADH secretion so water is retained. The thirst mechanism (not fully functional in infants) is also stimulated so water is replaced, thus increasing the total body water content and returning sodium to a normal level (Greenbaum, 2011). Potassium is primarily found inside the cell (intracellular) but small amounts are also found in extracellular fluid. Sodium depletion in diarrhea occurs in two ways: out of the body in stool and into the ICF compartment to replace potassium to maintain electrical equilibrium.
![]() Case Study—Dehydration and Diarrhea
Case Study—Dehydration and Diarrhea
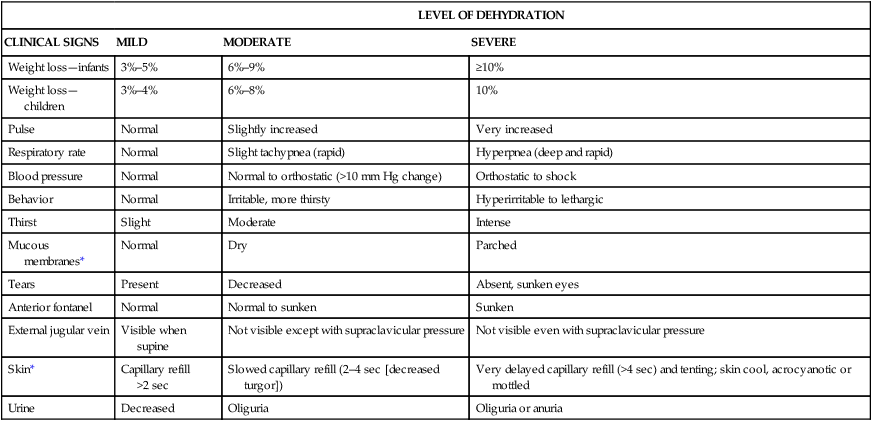
Degree of Dehydration
LEVEL OF DEHYDRATION
CLINICAL SIGNS
MILD
MODERATE
SEVERE
Weight loss—infants
3%–5%
6%–9%
≥10%
Weight loss—children
3%–4%
6%–8%
10%
Pulse
Normal
Slightly increased
Very increased
Respiratory rate
Normal
Slight tachypnea (rapid)
Hyperpnea (deep and rapid)
Blood pressure
Normal
Normal to orthostatic (>10 mm Hg change)
Orthostatic to shock
Behavior
Normal
Irritable, more thirsty
Hyperirritable to lethargic
Thirst
Slight
Moderate
Intense
Mucous membranes*
Normal
Dry
Parched
Tears
Present
Decreased
Absent, sunken eyes
Anterior fontanel
Normal
Normal to sunken
Sunken
External jugular vein
Visible when supine
Not visible except with supraclavicular pressure
Not visible even with supraclavicular pressure
Skin*
Capillary refill >2 sec
Slowed capillary refill (2–4 sec [decreased turgor])
Very delayed capillary refill (>4 sec) and tenting; skin cool, acrocyanotic or mottled
Urine
Decreased
Oliguria
Oliguria or anuria
MANIFESTATION
ISOTONIC (LOSS OF WATER AND SODIUM)
HYPOTONIC (LOSS OF SODIUM IN EXCESS OF WATER)
HYPERTONIC (LOSS OF WATER IN EXCESS OF SODIUM)
Skin
Color
Gray
Gray
Gray
Temperature
Cold
Cold
Cold or hot
Turgor
Poor
Very poor
Fair
Feel
Dry
Clammy
Thickened, doughy
Mucous membranes
Dry
Slightly moist
Parched
Tearing and salivation
Absent
Absent
Absent
Eyeball
Sunken
Sunken
Sunken
Fontanel
Sunken
Sunken
Sunken
Body temperature
Subnormal or elevated
Subnormal or elevated
Subnormal or elevated
Pulse
Rapid
Very rapid
Moderately rapid
Respirations
Rapid
Rapid
Rapid
Behavior
Irritable to lethargic
Lethargic or comatose; seizures
Marked lethargy with extreme hyperirritability on stimulation
Diagnostic Evaluation
![]() To initiate a therapeutic plan, several factors must be determined:
To initiate a therapeutic plan, several factors must be determined:
Therapeutic Management
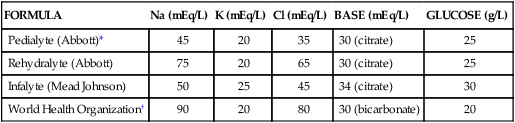
FORMULA
Na (mEq/L)
K (mEq/L)
Cl (mEq/L)
BASE (mEq/L)
GLUCOSE (g/L)
Pedialyte (Abbott)*
45
20
35
30 (citrate)
25
Rehydralyte (Abbott)
75
20
65
30 (citrate)
25
Infalyte (Mead Johnson)
50
25
45
34 (citrate)
30
World Health Organization†
90
20
80
30 (bicarbonate)
20
Parenteral Fluid Therapy.

The Child with Gastrointestinal Dysfunction
Get Clinical Tree app for offline access


