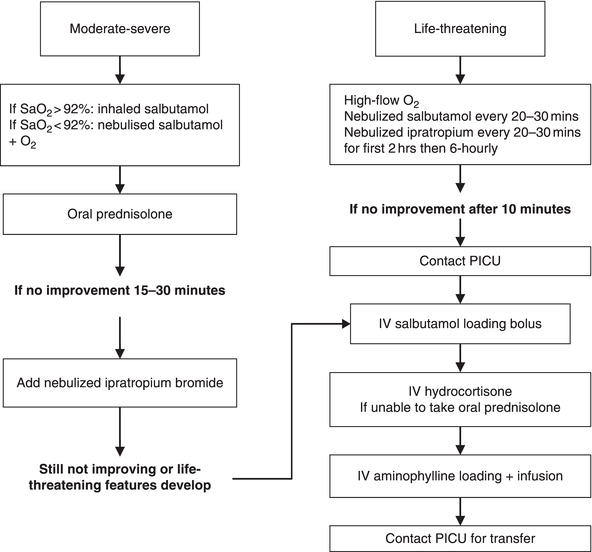
1Consultant Respiratory Paediatrician Asthma is one of the most common chronic conditions and 10–15% of children in western society suffer from it (Pearce et al. 1993), with boys twice as likely as girls to be affected. Asthma UK estimates (2009) that: The International Study of Asthma and Allergies in Childhood (ISAAC) has shown the rising prevalence of self-reported asthma symptoms and has allowed worldwide comparisons of the prevalence of asthma symptoms and changes over time (ISAAC 1998; Pearce et al. 2007). Despite the rise in overall prevalence, the number of acute asthma episodes in the community has shown a recent decline (Sunderland and Fleming 2004). There is a significant cost burden to the NHS due to asthma. The Office of Health Economics (2004) estimated that the costs to the NHS in 2001 totalled £889 million. The word asthma derives from Greek aazein, ‘to breath with open mouth or to pant’. ‘This disorder starts with a common cold, & the patient is forced to gasp for breath day & night, until the phlegm is expelled, the flow completed & the lung well cleared’, from Treatise on Asthma by Moses Maimonides (AD 1135–1204). The Global Initiative for Asthma (GINA) is an organisation which works to increase awareness of asthma among health professionals, public health authorities and the general public, with the objective of improving prevention and management through a concerted worldwide effort. The GINA (2008) definition of asthma is as follows. Asthma is a chronic inflammatory disorder of the airway in which many cells play a role, in particular mast cells, eosinophils, and T-lymphocytes. In susceptible individuals this inflammation causes recurrent episodes of wheezing, breathlessness, chest tightness and cough particularly at night and/or in the early morning. These symptoms are usually associated with widespread but variable airflow limitation that is at least partly reversible either spontaneously or with treatment. The inflammation also causes an associated increase in airway responsiveness to a variety of stimuli. This descriptive definition highlights the three underlying cardinal pathophysiological factors in asthma. Godfrey (2000) classifies the AHR challenges as allergen mediated, direct and indirect. Challenges with an inhaled allergen to which the susceptible individual has become sensitised could be potentially dangerous because of severe reactions; they are rarely used except for research. Chemicals such as methacholine or histamine will act directly on the airway smooth muscle. This test of AHR is commonly used as an endpoint in asthma clinical trials to determine the effectiveness of an intervention (Liem et al. 2008). These chemicals act directly on airway smooth muscle (ASM) to cause narrowing of the ASM in all individuals. In normal individuals, a much higher dose is required to do this while in those with AHR, this happens at a much lower dose. The difference in the asthmatic child is quantitative and not qualitative. With indirect challenges, the stimulus causes changes in the bronchial environment which are similar to what happens during an attack of asthma. The most common indirect challenge is the exercise test which under standardized environmental conditions in the laboratory assesses whether there is (greater than 12–15%) decline in lung function after a standardized level of exercise. When considering treatment, it is important to acknowledge that a diagnosis of asthma is not made on a single symptom. History taking is just as important as clinical features, investigations or tests and the whole process is often like putting several pieces of a jigsaw together to get the whole picture. The episodic nature of symptoms including breathlessness, cough, chest tightness and wheeze should raise the possibility of an asthma diagnosis. Parents often use the term ‘wheeze’ to describe any abnormal respiratory noise which on further investigation turns out to be a different respiratory noise (rattle or stridor) and not wheeze; therefore parents are often better at locating sounds than describing them (Elphick et al. 2001; Cane and McKenzie 2001). It is sometimes helpful to imitate various respiratory noses and clarify the wheeze as a whistling noise on breathing out (expiration). Wheeze on auscultation is a continuous, high-pitched musical sound coming from the chest. While taking the history, one should clarify that the child indeed has wheeze and not any other cause of noisy breathing. Once it is established that the child has a wheeze, one should try and ascertain if this is episodic or persistent as there are many different causes of wheeze in childhood and different clinical patterns of wheezing can be recognised in children. When wheezing is persistent or frequent and associated with symptoms in between episodes, the likelihood of these symptoms being due to asthma is greater. Children who have persisting or interval symptoms are most likely to benefit from therapeutic interventions. Enquiry should also be made about the presence or absence of atopy (inherited predisposition to produce IgE antibodies to various allergens). A personal history of atopic disorder (eczema, intermittent or persistent allergic rhinitis) or a family history of atopic disorder and/or asthma increases the probability of a diagnosis of asthma (BTS 2011). Atopic status can be established by performing skin rick tests or measuring blood-specific IgE to various inhaled or food allergens. A positive test is only indicative of sensitisation (the production of specific IgE antibody to the relevant allergen). Assessing atopy is also helpful in identifying triggers and in an individual may be related to the severity of current asthma and persistence through childhood. Presence of certain clinical features lowers the probability of an asthma diagnosis and alternative or additional diagnoses need to be considered. These features include symptoms associated with a cold only, with no interval symptoms. Others include isolated cough in the absence of wheeze or difficulty breathing, history of moist cough, prominent dizziness, light-headedness and peripheral tingling. Other elements would include repeatedly normal physical examination of chest when symptomatic, measurements such as normal peak expiratory flow (PEF) or spirometry when symptomatic, and medication provided for a trial period can demonstrate no response to a trial of asthma therapy when taken correctly through an appropriate inhaler device. Therefore the clinical features point to an alternative diagnosis, for example cystic fibrosis or vocal cord dysfunction. Normal oxygen saturation levels in association with sudden onset of wheezing, without any clear trigger, may indicate the presence of other conditions such as vocal cord dysfunction or hyperventilation. The BTS guidelines recommend that in children with a high probability of asthma, a trial of treatment should be started and the response reviewed within 2–3 months. In those children with a low probability of asthma and an additional or an alternative diagnosis, further investigations or specialist assessment would be appropriate. The GINA report (2008), based on a global strategy for asthma management, recommends specific goals for asthma treatment: minimal, and ideally no, symptoms during the day or at night; minimal, and ideally no, asthma episodes (exacerbations), including minimal use (less than daily) of a reliever medication (short-acting β2-agonist (SABA) such as salbutamol). Other variables include normal activities, normal lung function (PEFR/forced expiratory volume in 1 second (FEV1) ≥ 80% of the child’s personal best) and minimal, or ideally no, adverse effects from medications. In children and young adults, often it is better to set personal goals as well, for example the ability to complete a game of football without the need to take the reliever medication frequently. Treatment should be commenced at the appropriate level of severity for the individual patient. The BTS guidelines recommend a stepwise approach as described later in this chapter. When commencing inhaled treatment, an appropriate inhaler device should be prescribed after reviewing the technique and a spacer device should be prescribed for children for use with a pressurised metered dose inhaler. Children over 6 may be able to use a dry powder inhaler, and the device that the child demonstrates that they can use best and prefers should be prescribed. Regular review of inhaler technique should be carried out at each clinic as reinforcing the training is required to continue to maintain correct inhaler technique (Kamps et al. 2002). A more detailed discussion on use of inhaled devices is presented later in this chapter. Monitoring of treatment response and asthma control will be clinical and symptom based in the majority of patients. A structured symptomatic assessment is required to correctly assess the severity of asthma as unstructured assessments lead to underestimating the severity and could adversely influence the treatment decisions (Halterman et al. 2006). Several patient-based validated tools are available, including the Asthma Control Test (ACT) (Schatz et al. 2006) and are also included as recommendations by the BTS guidelines. The ACT helps to identify patients with poorly controlled asthma (Schatz et al. 2006). This tool consists of five pertinent questions, shown in Box 10.1, relating to symptoms and quality of life over a 4-week period. Essentially, the higher the ACT scores, the better the control. A score of 20 or more indicates good control and the goal should be an ideal score of 25 for complete control. Other things that need to be included in assessing control are lung function, number and severity of exacerbations requiring use of healthcare resources, oral corticosteroid use and time off school or work since last assessment. Treatment concordance should also be assessed, especially if the response to correctly prescribed treatment is not as expected. Adverse environmental influences, including exposure to environmental tobacco smoke, ongoing allergen exposure and psychosocial factors need to be reviewed in those with ‘difficult’ asthma (Ranganathan et al. 2001). Even though it has been shown that increased allergen exposure in sensitised individuals is associated with an increase in asthma symptoms, bronchial hyper-responsiveness and deterioration in lung function, the research evidence supporting reduction in allergen exposure to reduce morbidity and/or mortality in asthma is tenuous (BTS 2009, 2011). Measures to decrease house dust mites have been shown to reduce their numbers but have not been shown to have an effect on asthma severity. Cochrane reviews on house dust mite control measures in a normal domestic environment have concluded that chemical and physical methods aimed at reducing exposure to house dust mite allergens cannot be recommended (Gøtzsche et al. 2004). Some very committed families with evidence of house dust mite sensitisation may wish to try mite avoidance by considering the following: The literature on pet avoidance is very confusing and generally speaking, individual aeroallergen avoidance strategies have limited or no benefit. A multifaceted approach is more likely to be effective if it addresses all the indoor asthma triggers. Treatment and management need the co-operation of the child and parents. Advances in medicines and proficient guidelines have facilitated care based on best practice. The 2008 BTS Guideline on the Management of Asthma was based on review of the literature published up to March 2007. The 2011 revision includes updates on monitoring asthma, a whole new section on asthma in adolescents and pharmacological management. These revisions are clearly highlighted as 2011 changes and stress the importance of using closed questions when asking about asthma control and being aware that the best predictor for future exacerbations is level of current control. On an annual basis at least, the following should be monitored: It should be recognised that doses of inhaled steroids are referenced against beclomethasone dipropionate (BDP) with hydrofluroalkane (HFA) propellants and that these products are more potent and all should be prescribed by brand. In children (5–12 years) on inhaled corticosteroids (ICS) at a dose of 400 µg/day of BDP equivalent, addition of an inhaled long-acting β2-agonist (LABA) is the first choice. These agents improve lung function AND symptoms, and decrease exacerbations. This should be done before doubling the dose of ICS as the dose–response curve with ICS is flat beyond a certain dose so the likelihood of systemic side-effects increases. A flat dose–response curve means that increasing the dose is unlikely to give additional benefit and more likely to produce side-effects. LABAs should never be used on their own without ICS. A review by the Medicines and Healthcare products Regulatory Agency (MHRA) has concluded that the benefits of these medicines used in conjunction with ICS in the control of asthma symptoms outweigh any apparent risks. Leukotriene receptor antagonists may provide improvement in lung function, a decrease in exacerbations, and an improvement in symptoms. Omalizumab (humanised monoclonal antibody which binds to circulating IgE) has NICE approval for use in patients > 12 years old. The criteria for its use in this age group include the use of high-dose ICS and LABA, impaired lung function, symptomatic with frequent exacerbations and IgE levels 30–1300 kilounits (KU)/L. Omalizumab is given as a subcutaneous injection every 2–4 weeks and should only be initiated in specialist centres with experience of evaluation and management of patients with severe and difficult asthma. It is also licensed for use but not NICE approved for children 6–12 years of age and the IgE levels in this age range should be lower than 1500 KU/L. The guidelines recommend a stepwise approach to management and initial treatment should be at the step most appropriate to the initial severity of the patient’s asthma. The goals should be individualised (see GINA above) but with a stepwise approach, the aim should be to abolish symptoms as soon as possible and to optimise lung function. The aim is to achieve early control and to maintain control by stepping up treatment as necessary and stepping down when control is good. For paediatric patients, there are different recommendations for stepwise management across three age ranges including children > 12 years old (similar to adults), children 5–12 years, and children under 5 years. The evidence to support treatment recommendations is less clear in children under 2 and the threshold for seeking an expert opinion should be lowest in these children. An inhaled SABA (salbutamol or terbutaline) as short-term reliever therapy should be prescribed for all patients with symptomatic asthma. SABAs work more quickly and/or with fewer side-effects than the alternatives (inhaled ipratropium bromide, β2-agonist or theophylline tablets). This should be on an as-required basis and ideally there should be little or no need for SABA use. Patients who require inhaled SABA three times a week or more or are symptomatic three times a week or more or waking one night a week should move to step 2. Inhaled corticosteroids such as beclomethasone, used in recommended doses, are the most effective and safe preventer drugs for achieving symptom control, improvement in lung function, and prevention/reduction of exacerbations. ICS should be considered for children aged 5–12 and children under the age of 5 if they: Reasonable starting doses are, in children > 12 years old (similar to adults), 400 µg per day, and in children 5–12 years, 200 µg per day. In children less than 5 years of age, higher doses may be required if there are problems in obtaining consistent drug delivery. The dose should be titrated to the lowest dose at which effective control of asthma is maintained. Leukotriene receptor antagonists can be used as monotherapy in this age group if ICS cannot be used. Inhaled corticosteroids have been in use since the 1970s and are safe drugs when taken in recommended doses as discussed above. However, systemic side-effects secondary to ICS have come to light since the late 1990s and every clinician prescribing ICS should be vigilant to the possibility of side-effects, especially if there is additional steroid burden in the form of frequent courses of oral steroids for exacerbations as well as cumulative steroid burden, for example an atopic child also receiving steroid creams for eczema and nasal steroids for significant allergic rhinitis. Patients on higher dose ICS could be issued steroid warning cards but this may have an impact on compliance and the benefits of this suggestion are not clearly defined. Administration of ICS at or above 400 µg a day of BDP or equivalent may be associated with systemic side-effects, including growth failure and adrenal suppression. It has been known for some years that suppression of the hypothalamic-pituitary-adrenal (HPA) axis occurs in children exposed to ICS (Broide et al. 1995; Fitzgerald et al. 1998; Kannisto et al. 2000). More recently, clinical adrenal insufficiency at the time of intercurrent illness has been identified in a small number of children who have become acutely unwell (Patel et al. 2001). Most of these children had been treated with high doses of ICS, such as budesonide, fluticasone and beclomethasone, outside the recommended doses (Todd et al. 2002). The dose or duration of ICS treatment required to place a child at risk of clinical adrenal insufficiency is unknown but is likely to occur at ≥ 800 µg per day of BDP or equivalent. The low-dose ACTH test (short synacthen) is considered to provide a physiological stimulation of adrenal responsiveness but it is not known how useful such a sensitive test is at predicting clinically relevant adrenal insufficiency and also how often it should be repeated if abnormal. Regular review of ICS doses should be undertaken, stepping down on dose with good control, being vigilant about cumulative steroid exposure and prescribing the lowest dose of ICS required for disease control. Systemic side-effects of ICS should be kept in mind and specific written advice about steroid replacement in the event of a severe intercurrent illness should be part of the management plan. Although recommended in previous guidelines and as part of asthma action plans, doubling the dose at the time of an exacerbation is of unproven value. At each clinical review and when asthma control is not achieved, inhaler technique should be checked and reinforced. Treatment adherence needs to be explored, checked and encouraged and trigger factors eliminated as far as possible. Stepping up and using a trial of add-on therapy should be considered if control is not good. The options for add-on therapy are as follows. Long-acting β2-agonists are not licensed for use in children under 5 years old and step 3 for these children should be as follows. Guidance from NICE recommends that in children with chronic asthma in whom treatment with an ICS and LABA is considered appropriate, the following apply. There is no difference in efficacy in giving ICS and LABA in combination or in separate inhalers. However, once a patient is on stable therapy, combination inhalers have the advantage of guaranteeing that the LABA is not taken without ICS. In children under 5 years of age, if control is not achieved on step 3, indicating persistently poor control, early referral to a respiratory paediatrician should be made. If control remains inadequate on 800 µg daily (>12 years) or 400 µg daily (children) of an ICS plus a LABA, consider the following interventions. However, remember that this should be as a trial and if it is ineffective, stop the drug or reduce the dose back. Use daily steroid tablets at the lowest dose providing adequate control and refer to a respiratory paediatrician. Regular monitoring of systemic side-effects should be undertaken, including measuring blood pressure, growth, urine sugar, adrenal insufficiency, cataracts and bone health. The aim of treatment is to control the asthma using the lowest possible dose or, if possible, to stop long-term steroid tablets completely (BTS 2009). Treatment trials of steroid-sparing agents such as immunosuppressants (methotrexate, ciclosporin and oral gold), intravenous immunoglobulin and continuous subcutaneous terbutaline should only be undertaken in centres with relevant experience and facilities. Omalizumab (humanised monoclonal antibody which binds to circulating IgE) has NICE approval for use in patients > 12 years old on high-dose ICS and LABA who have impaired lung function, are symptomatic with frequent exacerbations, and have IgE levels 30–700 KU/L. It is given as a subcutaneous injection every 2–4 weeks and should only be initiated in specialist centres with experience of evaluation and management of patients with severe and difficult asthma (Kulis et al. 2010). Patients should be maintained at the lowest possible dose of ICS and the dose reduction should be slow and individualised but can be considered every 3 months, decreasing the dose by approximately 25–50% each time. Once asthma control is achieved, stepping down therapy should be undertaken. This should be done at regular clinical reviews and the lowest dose of ICS that achieves asthma control should be used as maintenance treatment. For those admitted to hospital with severe asthma, a different approach is required. Acute severe asthma is a major reason for admission to hospital and remains the most common medical emergency in children. Prevalence of asthma has increased by 50% since 1980. Severe asthma still remains a cause of death; it is tragic that one-third of these patients would have been previously diagnosed as being mild. The last few years have seen a steady increase in the number of patients admitted to paediatric intensive care units (PICU), although the majority of them stay for a relatively short period. Elements of respiratory system development have been discussed in Chapter 1. During an acute attack of asthma there is marked airway inflammation producing excessive mucus, leading to airway plugging. In addition, bronchospasm produces severe airflow limitation, leading to progressive respiratory failure. In a small group of children acute anaphylaxis will lead to intense bronchospasm without significant mucus plugging. In the majority of children there are no specific identifiable precipitating factors although occasionally a history of anaphylaxis may be present. A detailed history is essential for rapid and appropriate management. The following details are required. The most common precipitating factors are viral infections, allergens such as exposure to cats or grass pollen, and food such as eggs and peanuts. Occasionally emotional and social factors may bring on a severe attack (Halterman et al. 2006). It is always necessary to exclude other diagnoses such as lower respiratory tract infection, bronchiolitis and pneumonia. As well as infection, there could also be lower respiratory tract obstruction. Therefore in a child with no previous history, there is the need to rule out foreign body inhalation or mediastinal tumour, e.g. lymphoma. Another area to consider is upper airway obstruction (usually inspiratory stridor would be obvious). A variety of conditions such as epiglottitis, croup or bacterial tracheitis, foreign body, polyps/tumour, vascular ring or tracheomalacia can contribute towards upper airway obstruction. Other possibilities include: Some of the risks associated with a severe attack of asthma include a history of sudden and rapid deterioration, possibly due to acute anaphylaxis. With poor compliance in clinical practice, it is noted that these children will often have a severe attack. If there is evidence of previous attacks of severe asthma requiring PICU admission and mechanical ventilation, this can also be a contributing factor to the increased risk associated with severe asthma attacks. Again, the evidence suggests that excessive use of β-agonists also increases the risk, including a reluctance to recognise severity and poor perception of hypoxia. Note: PEFR measurement is not tolerated by acutely distressed or young children. Assessment of severity of the attack is shown in Table 10.1 and the initial management in the emergency department is summarised in Figure 10.1. Table 10.1 Assessment of severity bpm, beats per minute; PEFR, peak expiratory flow rate. Initial management as always involves emergency treatment to maintain the airway, such as providing high oxygen concentration using a reservoir bag. Nebulised salbutamol continuously with oxygen also assists in the process, giving consideration to the fact that poor response may be due to inadequate gaseous exchange. In such cases intravenous salbutamol should be administered. We have already established that the process involved in asthma includes inflammation so early steroid, either orally if tolerated or intravenously, is essential. Sitting the child up will minimise ventilation/perfusion mismatch but failure to improve with the initial treatment necessitates admission to the PICU. Regular medical and nursing observations are essential. It is important to assess, evaluate and monitor clinical signs regularly, for example an increasing heart rate is a sign of worsening asthma. Respiratory rate and the degree of breathlessness, including the use of accessory muscles, should be assessed and evidence of air entry on auscultation is significant. In relation to this element, wheezing may be biphasic and may disappear on worsening airway obstruction. Following regular checks and observation, any signs of progressive agitation or obtundation indicate end-stage respiratory failure and require urgent intubation.
2Consultant in Paediatric Respiratory Medicine
3Specialist Practitioner in Children’s Community Nursing, Nottingham Children’s Hospital
Introduction
Definition of asthma
Treatment of asthma
Diagnosis
Goals for treatment
Allergen avoidance
House dust mite
Other allergens
British Thoracic Society guidelines for the management of asthma
Step 1: mild intermittent asthma
Step 2: introduction of regular preventer therapy
Inhaled corticosteroids
Safety and side-effects of inhaled corticosteroids
Step 3: initial add-on therapy
Combination inhalers
Step 4: poor control on moderate dose of inhaled steroid plus add-on therapy: addition of fourth drug
Step 5: continuous or frequent use of oral steroids
Steroid tablet-sparing medication
Stepping down
Acute severe asthma
Pathophysiology
History
Differential diagnosis
Risk factors for severe asthma
General management
General points for all asthmatics referred
Age 0–5 years
Age 5–15 years
Moderate-severe
Too breathless to talk
Too breathless to feed
Respiratory rate > 50
Pulse > 130 bpm
Significant respiratory distress
Too breathless to talk
PEFR < 50% predicted
Respiratory rate >30
Pulse >120 bpm
Use of accessory muscles
Life-threatening (any of these features)
Management of acute asthma in the emergency department
Clinical evaluation and monitoring
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree