(al-PROSS-tah-dill)
Prostaglandin E1, Prostin VR Pediatric
Prostaglandin (ductus arteriosus patency adjunct)
Usual dose
Pediatric patients:
Begin with 0.05 to 0.1 mcg/kg of body weight/min. When therapeutic response is achieved, reduce infusion rate in increments to the lowest dose that maintains the response (e.g., reduce from 0.1 to 0.05 or from 0.025 to 0.01 mcg/kg/min). If necessary, dose may be increased gradually to a maximum of 0.4 mcg/kg/min. Generally these higher rates do not produce greater effects. May be given through infusion in a large vein or, if necessary, through an umbilical artery catheter placed at the ductal opening.
Dilution
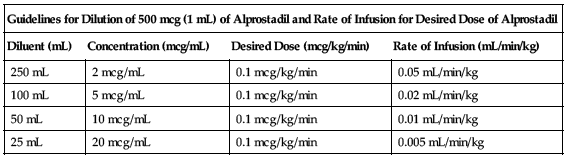
Each 500 mcg (1 mL) must be further diluted with NS or D5W. When using a volumetric infusion chamber, the appropriate amount of IV infusion solution should be added to the chamber first. The undiluted alprostadil should then be added to the IV infusion solution, avoiding direct contact of the undiluted solution with the wall of the volumetric infusion chamber; see Compatibility. Various volumes of infusion solution may be used depending on infusion pump capabilities and desired infusion rate.
| Guidelines for Dilution of 500 mcg (1 mL) of Alprostadil and Rate of Infusion for Desired Dose of Alprostadil | |||
| Diluent (mL) | Concentration (mcg/mL) | Desired Dose (mcg/kg/min) | Rate of Infusion (mL/min/kg) |
| 250 mL | 2 mcg/mL | 0.1 mcg/kg/min | 0.05 mL/min/kg |
| 100 mL | 5 mcg/mL | 0.1 mcg/kg/min | 0.02 mL/min/kg |
| 50 mL | 10 mcg/mL | 0.1 mcg/kg/min | 0.01 mL/min/kg |
| 25 mL | 20 mcg/mL | 0.1 mcg/kg/min | 0.005 mL/min/kg |
Storage:
Refrigerate until dilution. Prepare fresh solution for administration every 24 hours.
Compatibility
Consider any drug NOT listed as compatible to be INCOMPATIBLE until consulting a pharmacist; specific conditions may apply.
Manufacturer states that undiluted alprostadil may interact with the plastic sidewalls of the volumetric infusion chamber, causing a change in the appearance of the chamber and creating a hazy solution. Should this occur, the solution and the volumetric infusion chamber should be replaced; see Dilution.
One source suggests the following compatibilities:
Y-site:
Ampicillin, cefazolin (Ancef), cefotaxime (Claforan), chlorothiazide (Diuril), dobutamine, dopamine, fentanyl, gentamicin, methylprednisolone (Solu-Medrol), nitroprusside sodium, tobramycin, vancomycin, vecuronium.
Rate of administration
See Usual Dose. Infusion pump capable of delivering 0.005, 0.01, 0.02, or 0.05 mL/min/kg required. Use for the shortest time possible at the lowest rate therapeutically effective. Decrease rate of infusion stat if a significant fall in arterial pressure occurs.
Actions
A naturally occurring acidic lipid. A prostaglandin. Has various pharmacologic effects, including vasodilation, inhibition of platelet aggregation, and stimulation of the intestinal and uterine smooth muscle. Lowers blood pressure by decreasing peripheral resistance. A reflex increase in cardiac output and heart rate accompany the reduction in blood pressure. Smooth muscle of the ductus arteriosus is susceptible to alprostadil’s relaxing effect. Infants with congenital defects that restrict the pulmonary or systemic blood flow may benefit from alprostadil infusion. In some infants with restricted pulmonary blood flow, an increase in blood pO2 was observed. In some infants with restricted systemic blood flow, an increase in pH in those who were acidotic, an increase in SBP, and a decrease in the ratio of pulmonary artery pressure to aortic pressure were seen. Rapid metabolism by oxidation (80% in one pass through the lungs) necessitates administration by continuous infusion. Remainder excreted as metabolites in the urine.
Indications and uses
Temporarily maintain the patency of the ductus arteriosus until corrective or palliative surgery can be performed on neonates who have congenital heart defects and who depend on the patent ductus for survival. Such congenital heart defects include pulmonary atresia, pulmonary stenosis, tricuspid atresia, tetralogy of Fallot, interruption of the aortic arch, coarctation of the aorta, or transposition of the great vessels. ■ Used by intercavernosal injection to treat impotence.
Contraindications
None known.
Precautions
Usually administered by trained personnel in pediatric intensive care facilities. ■ Establish a diagnosis of cyanotic heart disease (restricted pulmonary blood flow). Not indicated for infant respiratory distress syndrome (hyaline membrane disease). ■ Response is poor in infants with Po2 values of 40 torr or more or those more than 4 days old. More effective with lower Po2. ■ Apnea has been experienced in 10% to 12% of treated neonates; see Monitor. ■ Use caution in neonates with bleeding tendencies; inhibits platelet aggregation. ■ Administration of alprostadil to neonates may result in gastric outlet obstruction secondary to antral hyperplasia. Appears to be related to duration of therapy and cumulative dose. Risk of long-term infusion should be weighed against the possible benefits in a critically ill neonate; see Monitor. ■ Cortical proliferation of the long bones has been observed in infants during long-term infusions. May regress after withdrawal of drug.
Monitor:
Monitor respiratory status continuously. Ventilatory assistance must be immediately available. May cause apnea, especially in infants under 2 kg. Apnea usually appears during the first hour of infusion. ■ Monitor arterial pressure intermittently by umbilical artery catheter, auscultation, or Doppler transducer. Decrease rate of infusion stat if a significant fall in arterial pressure occurs. ■ Decrease or stop infusion if infant develops increased respiratory distress; bleeding, bruising, or hematoma formation; or sudden changes in cardiac status (e.g., decreased BP, bradycardia, cardiac arrest, cyanosis). ■ Measure effectiveness with increase of Po2 in infants with restricted pulmonary blood flow and increase of BP and blood pH in infants with restricted systemic blood flow. ■ Monitor for evidence of antral hyperplasia and gastric outlet obstruction in neonates receiving more than 120 hours of therapy at recommended doses.
Drug/lab interactions
No drug interactions have been reported between alprostadil and the therapy that is standard in neonates with restricted pulmonary or systemic blood flow. Standard therapy includes antibiotics such as penicillin or gentamicin; vasopressors such as dopamine and isoproterenol; cardiac glycosides; and diuretics such as furosemide. ■ Inhibits platelet aggregation.
Side effects
Apnea, bradycardia, cardiac arrest, cerebral bleeding, cortical proliferation of long bones, diarrhea, DIC, edema, fever, flushing, hyperextension of the neck, hyperirritability, hypokalemia, hypotension, hypothermia, seizures, sepsis, tachycardia. Many other side effects have occurred in 1% or less of infants receiving alprostadil.
Overdose:
Antidote
Notify physician of all side effects. Discontinue immediately if apnea occurs. Institute emergency measures. If infusion is restarted, use extreme caution. Decrease or stop infusion if infant develops increased respiratory distress; bleeding, bruising, or hematoma formation; or sudden changes in cardiac status (e.g., decreased BP, bradycardia, cardiac arrest, cyanosis). Decrease rate if pyrexia, hypotension, or fall in arterial pressure occurs.
Alteplase
(AL-teh-playz)
Activase, rt-PA  , Tissue Plasminogen Activator, tPA ■ Cathflo Activase
, Tissue Plasminogen Activator, tPA ■ Cathflo Activase
Thrombolytic agent
pH 7.3
Usual dose
Selected indications (e.g., acute ischemic stroke, acute myocardial infarction [AMI] in patients weighing less than 65 or 67 kg) require exact weight-adjusted dosing. To deliver an accurate dose without possibility of overdose, calculate desired dose and withdraw any amount NOT needed from a 50- or 100-mg vial and discard. In all situations follow total dose with at least 30 mL of NS or D5W through the IV tubing to ensure administration of total dose.
Acute myocardial infarction
Administer as soon as possible after onset of symptoms. Total dose is based on patient weight and should not exceed 100 mg regardless of selected administration regimen (accelerated infusion and 3-hour infusion are outlined in the following sections).
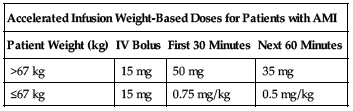
Accelerated infusion:
Recommended accelerated infusion dose consists of an IV bolus over 1 to 2 minutes followed by an IV infusion as shown in the following chart.
| Accelerated Infusion Weight-Based Doses for Patients with AMI | |||
| Patient Weight (kg) | IV Bolus | First 30 Minutes | Next 60 Minutes |
| >67 kg | 15 mg | 50 mg | 35 mg |
| ≤67 kg | 15 mg | 0.75 mg/kg | 0.5 mg/kg |
The safety and efficacy of accelerated infusion of alteplase have been investigated only with concomitant administration of heparin and aspirin. See comments under Usual Dose.
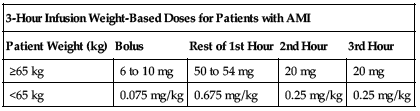
3-hour infusion:
For patients weighing 65 kg or more, administer a total dose of 100 mg titrated over 3 hours as an IV infusion. Initially, administer 60 mg over the first hour (with 6 to 10 mg of this administered as a bolus over 1 to 2 minutes). Follow with 20 mg/hr for 2 hours. For smaller patients (less than 65 kg), calculate total dose using 1.25 mg/kg of body weight administered over 3 hours (with 0.075 mg/kg administered as a bolus over 1 to 2 minutes). Follow with 0.675 mg/kg over the first hour and 0.25 mg/kg for 2 hours. Weight-based doses are shown in the following chart. See comments under Usual Dose.
| 3-Hour Infusion Weight-Based Doses for Patients with AMI | ||||
| Patient Weight (kg) | Bolus | Rest of 1st Hour | 2nd Hour | 3rd Hour |
| ≥65 kg | 6 to 10 mg | 50 to 54 mg | 20 mg | 20 mg |
| <65 kg | 0.075 mg/kg | 0.675 mg/kg | 0.25 mg/kg | 0.25 mg/kg |
Acute ischemic stroke
Administer as soon as possible but within 3 hours after onset of symptoms. Perform non–contrast-enhanced CT or MRI before administration to rule out intracranial bleed. Recommended dose is 0.9 mg/kg. Do not exceed the maximum dose of 90 mg. See comments in Usual Dose. Give a bolus of 10% of the calculated dose over 1 minute followed by balance of calculated dose (90%) as an infusion evenly distributed over 60 minutes. Do not give aspirin, heparin, or warfarin for 24 hours; see Precautions.
Pulmonary embolism
100 mg administered over 2 hours as an IV infusion. Begin parenteral anticoagulation near the end of or immediately following the alteplase infusion when the partial thromboplastin time (PTT) or thrombin time (TT) returns to twice normal or less.
Central venous access device (CVAD) occlusions, cathflo activase
See Rate of Administration, Precautions, Monitor, and Maternal/Child.
Patient weight 30 kg or greater:
Instill 2 mg in 2 mL into the occluded catheter.
Patient weight less than 30 kg:
Instill 110% of the internal lumen volume of the occluded catheter into the occluded catheter. Do not exceed 2 mg in 2 mL.
Attempt to aspirate blood from the catheter after 30 minutes of dwell time. If catheter function has been restored, aspirate 4 to 5 mL of blood in patients equal to or greater than 10 kg or 3 mL of blood in patients less than 10 kg to remove Cathflo Activase and residual clot, then gently irrigate the catheter with NS.
If catheter function has not been restored (unable to aspirate blood), allow the first dose to remain in the catheter for 90 additional minutes of dwell time and then attempt to aspirate again (total elapsed time is 120 minutes). If catheter function has been restored, aspirate and irrigate as above. If function has not been restored, a second dose may be instilled and the dwell time and aspiration process repeated.
Dilution
Myocardial infarction, stroke, and pulmonary embolism:
Must be reconstituted with SWFI without preservatives (provided by manufacturer). Available in 50- and 100-mg vials. May be administered as reconstituted solution at 1 mg/mL or further diluted immediately before administration in an equal volume of NS or D5W to yield a concentration of 0.5 mg/mL using either polyvinyl chloride bags or glass vials. Slight foaming is expected; let stand for several minutes to dissipate large bubbles. Do not shake. Mix by swirling or slow inversion; avoid agitation during dilution.
50-mg vial:
Use a large-bore (18-gauge) needle and syringe to direct the stream of provided diluent (SWFI) into the lyophilized cake. A vacuum must be present when the diluent is added to the powder for injection. Do not use if vacuum is not present.
Bolus dose can be given when indicated by IV injection through a med port or by IV pump. To obtain bolus dose, do not prime syringe used with air; insert needle into the alteplase vial stopper to withdraw dose. Administer balance of dose using either a polyvinyl chloride bag or glass vial and infusion set. Adjust pump rate of delivery as required. Complete by flushing tubing with IV solution.
100-mg vial:
Does not contain a vacuum. Diluent and transfer device provided. Insert one end of transfer device into upright vial of diluent (do not invert diluent vial yet). Hold alteplase vial upside down and push center of vial down onto piercing pin. Now invert vials and allow diluent to flow into alteplase. Small amount (0.5 mL) of diluent will not transfer. Swirl gently to dissolve. Do not shake. Process takes several minutes. Remove any quantity of drug in excess of that specified for patient treatment; see Usual Dose.
Bolus dose can be given when indicated by IV injection through a med port or by IV pump. When withdrawing the bolus dose from a 100-mg vial, the needle should be inserted away from the puncture mark made by the transfer device. Administer the balance of dose by inserting an infusion set into puncture site created by piercing pin. Hang by plastic capping on bottom of vial. Prime tubing with alteplase and administer. Adjust pump rate of delivery as required. Complete by flushing tubing with IV solution.
Cathflo activase for CVAD occlusion:
Supplied as a sterile lyophilized powder in 2-mg vials. Must be reconstituted with 2.2 mL of SWFI without preservatives. Direct diluent stream into the powder. Allow to stand undisturbed until large bubbles dissipate; may foam slightly. Mix by swirling gently; do not shake. Should be completely dissolved within 3 minutes. Final concentration is 1 mg/mL. If the 2-mg vials are not available, some pharmacies dilute 50 mg of alteplase with 50 mL of SWFI without preservatives (1 mg/mL). Withdraw 2.2 mL (2.2 mg) to 2- or 5-mL sterile disposable syringes. In studies these were transferred to sterile glass vials and frozen. Defrost and use as needed (22 prepared doses).
Filters:
Specific information not available.
Storage:
Systemic alteplase:
Protect from light in cartons. May be stored at CRT or refrigerated before and/or after reconstitution. Manufacturer recommends reconstitution immediately before use. Must be used within 8 hours of reconstitution. Discard unused solution. Stable as a 0.5 mg/mL solution in NS or D5W for 8 hours at CRT.
Cathflo activase:
Refrigerate unopened vials; protect from light during extended storage. Reconstitution immediately before use is recommended, but solution may be used up to 8 hours after reconstitution if refrigerated at 2° to 30° C (36° to 46° F). Do not use beyond expiration date on vial. Discard unused solution.
Compatibility
Consider any drug NOT listed as compatible to be INCOMPATIBLE until consulting a pharmacist; specific conditions may apply.
Manufacturer states, “No other medication should be added to infusion solutions containing alteplase; see Dilution.
One source suggests the following compatibilities:
Y-site:
Lidocaine, metoprolol (Lopressor), and propranolol through Y-site of free-flowing alteplase infusion.
Rate of administration
Systemic alteplase:
See specific rates for each diagnosis under Usual Dose. In all situations use an infusion pump (preferred) or a metriset with microdrip (60 gtt/mL) and IV tubing to facilitate accurate administration. Flushing of IV line required. NS preferred for flushing of IV line.
Cathflo activase:
Avoid excessive pressure or force while attempting to clear catheters; see Usual Dose, Precautions, and Monitor.
Actions
A tissue plasminogen activator and enzyme produced by recombinant DNA. It binds to fibrin in a thrombus and converts plasminogen to plasmin. Plasmin digests fibrin and dissolves the clot. With therapeutic doses, a decrease in circulating fibrinogen makes the patient susceptible to bleeding. Onset of action is prompt, effecting patency of the vessel within 1 to 2 hours in most patients. Prompt opening of arteries increases probability of improved function. Cleared from the plasma by the liver within 5 (50%) to 10 (80%) minutes after the infusion is discontinued. Some effects may linger for 45 minutes to several hours.
Indications and uses
Systemic alteplase:
Use in acute myocardial infarction (AMI) for the reduction of the incidence of congestive heart failure and the reduction of mortality associated with AMI. ■ Treatment of acute ischemic stroke. Exclude intracranial hemorrhage as the primary cause of stroke S/S before initiation of treatment. ■ Lysis of acute massive pulmonary embolism defined as (1) acute pulmonary emboli obstructing blood flow to a lobe or to multiple lung segments, or (2) acute pulmonary emboli accompanied by unstable hemodynamics (e.g., failure to maintain BP without supportive measures).
Limitation of use:
In patients with AMI, the risk of stroke may outweigh the benefit produced by thrombolytic therapy in patients whose AMI puts them at low risk for death or heart failure.
Cathflo activase:
Restoration of function to CVADs as assessed by the ability to withdraw blood.
Unlabeled uses:
Systemic alteplase:
Treatment of acute ischemic stroke 3 to 4.5 hours after symptom onset. Has been shown to restore blood flow to frostbitten limbs. Has been used in peripheral arterial occlusion, prosthetic valve thrombosis, and submassive pulmonary embolism.
Contraindications
All indications:
Known hypersensitivity to alteplase or its components.
Acute myocardial infarction/pulmonary embolism:
Do not administer for the treatment of AMI or pulmonary embolism in the following situations in which the risk of bleeding is greater than the potential benefit: active internal bleeding, bleeding diathesis, history of recent cerebrovascular accident, recent (within 3 months) intracranial or intraspinal surgery or serious head trauma, current severe uncontrolled hypertension, presence of intracranial conditions that may increase the risk of bleeding (e.g., some neoplasms, arteriovenous malformation, or aneurysm).
Acute ischemic stroke:
Do not administer to treat acute ischemic stroke in the following situations in which the risk of bleeding is greater than the potential benefit: current intracranial hemorrhage (on pretreatment evaluation), subarachnoid hemorrhage, active internal bleeding, presence of intracranial conditions that may increase the risk of bleeding (e.g., some neoplasms, arteriovenous malformation, or aneurysm), recent (within 3 months) intracranial or intraspinal surgery or serious head trauma, bleeding diathesis, or current severe uncontrolled hypertension.
Precautions
All systemic indications:
Administered under the direction of a physician knowledgeable in its use and with appropriate emergency drugs and diagnostic and laboratory facilities available. ■ Bleeding is the most common complication. May be internal bleeding (involving intracranial or retroperitoneal sites or the GI, GU, or respiratory tracts) or external bleeding, especially at arterial and venous puncture sites. Use extreme care with the patient; avoid IM injections and any trauma to the patient who is taking alteplase. Avoid invasive procedures (e.g., arterial puncture and venipuncture). If these procedures are absolutely necessary, use extreme precautionary methods. To minimize bleeding from noncompressible sites, avoid internal jugular and subclavian venous punctures. If an arterial puncture is necessary, use an upper extremity vessel that is accessible to manual compression, apply pressure for at least 30 minutes, and monitor the puncture site closely. ■ Fatal cases of hemorrhage associated with traumatic intubation in patients receiving alteplase have been reported. ■ Aspirin and heparin have been administered concomitantly with and following infusions of alteplase in the management of AMI and PE. However, the administration of heparin and aspirin concomitantly with and following infusions of alteplase for the treatment of acute ischemic stroke during the first 24 hours after symptom onset has not been investigated. Because heparin, aspirin, or alteplase may cause bleeding complications, carefully monitor for bleeding. Hemorrhage can occur 1 or more days after administration of alteplase while patients are still receiving anticoagulant therapy. ■ The risk of bleeding with alteplase therapy for all systemic indications is increased and should be weighed against the anticipated benefit in any of the following conditions: recent major surgery or procedure (e.g., CABG, obstetric delivery, organ biopsy, previous puncture of noncompressible vessels), recent intracranial hemorrhage, recent GI or GU bleeding, cerebrovascular disease, hypertension (systolic above 175 mm Hg or diastolic above 110 mm Hg), recent trauma, high likelihood of left heart thrombus (e.g., mitral stenosis with atrial fibrillation), acute pericarditis, subacute bacterial endocarditis, hemostatic defects (including those secondary to severe hepatic or renal disease), significant liver dysfunction, pregnancy, hemorrhagic ophthalmic conditions (e.g., diabetic hemorrhagic retinopathy), septic thrombophlebitis or occluded AV cannula at seriously infected site, patients taking anticoagulants, advanced age, or any situation in which bleeding might be hazardous or difficult to manage because of location. ■ Cholesterol embolism has been reported rarely; see Side Effects. ■ Orolingual angioedema has been reported in patients with acute MI and acute ischemic stroke. Onset may occur during or up to 2 hours after infusion. May be associated with concomitant use of angiotensin-converting enzyme inhibitors. Most resolved with prompt treatment; see Drug/Lab Interactions and Antidote.
Myocardial infarction:
Reperfusion arrhythmias can occur (e.g., sinus bradycardia, accelerated idioventricular rhythm, PVCs, ventricular tachycardia); have antiarrhythmic medications available.
Acute ischemic stroke:
Because of the higher risk of intracranial hemorrhage in patients treated for acute ischemic stroke, treatment facility must be able to provide evaluation and management of intracranial hemorrhage. ■ Risk of intracranial hemorrhage may be increased in patients with severe neurologic deficit at presentation. ■ Treatment may begin before coagulation study results are known in patients without recent use of oral anticoagulants or heparin. Discontinue infusion if pretreatment INR is greater than 1.7 or if an elevated aPTT is identified. ■ Use caution in patients with blood glucose values less than 50 mg/dL or greater than 400 mg/dL; risk of misdiagnosis of acute ischemic stroke is increased. ■ Safety and efficacy in patients with minor neurologic deficit or rapidly improving symptoms before treatment is begun have not been studied; use is not recommended.
Pulmonary emboli:
Treatment with alteplase does not constitute adequate treatment of underlying deep venous thrombosis. ■ Risk of re-embolization due to lysis of underlying DVT should be considered.
Cathflo activase:
Consider causes of catheter dysfunction other than thrombus formation (e.g., catheter malposition, mechanical failure, constriction by a suture, lipid deposits or drug precipitates within the catheter lumen). ■ Do not apply vigorous suction during attempts to determine catheter occlusion; may risk damage to the vascular wall or collapse of soft-walled catheters. Avoid excessive pressure during instillation of Cathflo Activase into the catheter; may cause rupture of the catheter or expulsion of the clot into the circulation. ■ Use caution with patients who have active internal bleeding, thrombocytopenia, or other hemostatic defects (including those secondary to hepatic or renal disease). ■ Use caution in patients who have conditions for which bleeding constitutes a significant hazard, who would be difficult to manage because of location of the bleeding, who are at high risk for embolic complications (e.g., venous thrombosis in the region of the catheter), or who have had any of the following within 48 hours: surgery, obstetric delivery, percutaneous biopsy of viscera or deep tissues, or puncture of noncompressible vessels. ■ Use caution in the presence of known or suspected infection in the catheter; may release a localized infection into the systemic circulation. ■ See Monitor. ■ Safety and effectiveness of doses greater than 2 mg have not been established.
Monitor:
All systemic indications:
Establish a separate IV line for alteplase. ■ Obtain appropriate laboratory studies (e.g., PT, TT, PTT, INR, aPTT, CBC, fibrinogen levels, platelets). ■ Diagnosis-specific baseline studies (e.g., ECG, troponin in myocardial infarction, noncontrast CT brain scan, neurologic assessment in acute ischemic stroke, and lung scan or pulmonary angiography in pulmonary embolism) are indicated. ■ Monitor the patient carefully and frequently for pain and signs of bleeding; observe catheter sites frequently and apply pressure dressings to any recently invaded site; watch for hematuria, hematemesis, bloody stool, petechiae, hematoma, flank pain, or muscle weakness; perform neuro checks. Continue until normal clotting function returns. ■ Monitor BP and maintain within appropriate limits with antihypertensives or vasopressors as indicated. ■ Monitor for signs of orolingual angioedema during and for several hours after infusion. ■ Watch for extravasation; may cause ecchymosis and/or inflammation. Restart IV at another site. Moist compresses may be helpful. ■ See Precautions and Drug/Lab Interactions.
Myocardial infarction:
Monitor ECG.
Acute ischemic stroke:
Before the initiation of therapy, determine actual time of onset of stroke. ■ During and following alteplase administration, frequently monitor and control BP. The 2013 AHA/ASA guidelines for early management of acute ischemic stroke recommend monitoring BP and performing neurologic assessments every 15 minutes for 2 hours, every 30 minutes for 6 hours, then every 1 hour for 18 hours. ■ Hemorrhage in the brain occurs frequently during treatment with alteplase; monitor carefully. Acute neurologic deterioration, new headache, acute hypertension, or nausea and vomiting may indicate the occurrence of intracranial hemorrhage. If intracranial hemorrhage is suspected, discontinue alteplase and obtain a CT scan.
Cathflo activase:
Aseptic technique imperative. ■ Avoid force while attempting to clear catheters; may rupture catheter or dislodge clot into the circulation. ■ To prevent air from entering the open catheter and the circulatory system, instruct the patient to exhale and hold his/her breath any time the catheter is not connected to the IV tubing or a syringe. ■ See Precautions.
Patient education:
Compliance with all measures to minimize bleeding (e.g., strict bed rest) is very important. ■ Avoid use of razors, toothbrushes, and other sharp items. Use caution while moving to avoid excessive bumping. ■ Report all episodes of bleeding and apply local pressure if indicated. Expect oozing from IV sites.
Maternal/child:
Systemic alteplase:
Category C: safety for use in pregnancy and breast-feeding not established. ■ Safety and effectiveness for use in pediatric patients not established.
Cathflo activase:
Category C: use during pregnancy only if benefits justify potential risk to the fetus. ■ Use caution during breast-feeding. ■ Has been used in patients 2 weeks to 17 years of age. Rates of serious adverse events as well as restoration of catheter function similar to adults.
Elderly:
Systemic alteplase:
Possible increased risk of bleeding with advanced age (e.g., age greater than 77 years). In acute ischemic stroke, efficacy results suggest a reduced but still favorable clinical outcome for elderly patients treated with alteplase.
Cathflo activase:
No incidents of intracranial hemorrhage, embolic events, or major bleeding events were observed during studies. ■ Use caution in the elderly with conditions known to increase the risk of bleeding; see Precautions.
Drug/lab interactions
Risk of bleeding may be increased by any medicine that affects blood clotting, including anticoagulants (e.g., heparin, warfarin [Coumadin]); any medication that may cause hypoprothrombinemia, thrombocytopenia, or GI ulceration or bleeding (e.g., selected antibiotics [e.g., cefotetan], aspirin, NSAIDs [e.g., ibuprofen (Advil, Motrin), naproxen (Aleve, Naprosyn)]); and/or any other medication that inhibits platelet aggregation (e.g., clopidogrel [Plavix], dipyridamole [Persantine], glycoprotein GPIIb/IIIa receptor antagonists [e.g., abciximab (ReoPro), eptifibatide (Integrilin), tirofiban (Aggrastat)]). If concurrent or subsequent use is indicated, monitor patient closely. ■ Orolingual angioedema has been reported. Many patients, including patients with acute ischemic stroke, were receiving angiotensin-converting enzyme inhibitors (e.g., enalapril [Vasotec], enalaprilat [Vasotec IV], lisinopril [Zestril]). ■ Coagulation tests and measures of fibrinolytic activity may be unreliable; specific procedures can be used; notify the lab of alteplase use.
Side effects
Systemic alteplase:
Bleeding is most common: internal (GI tract, GU tract, retroperitoneal, or intracranial sites) and superficial or external bleeding (ecchymosis, epistaxis, gingival bleeding, venous cutdowns, arterial punctures, sites of recent surgical intervention). Mild to serious hypersensitivity reactions (e.g., anaphylactoid reaction, laryngeal edema, orolingual angioedema, rash, urticaria) have occurred. Cholesterol embolization can occur with thrombolytics but has been reported rarely. It may present with acute renal failure, bowel infarction, cerebral infarction, gangrenous digits, hypertension, livedo reticularis, myocardial infarction, pancreatitis, purple toe syndrome, retinal artery occlusion, rhabdomyolysis, or spinal cord infarction. Fatalities have been reported.
Post-marketing:
The following events may be life threatening:
Acute ischemic stroke (AIS):
Cerebral edema, cerebral herniation, new ischemic stroke, seizure.
Acute myocardial infarction (AMI):
Arrhythmias, AV block, cardiac arrest, cardiac tamponade, cardiogenic shock, electromechanical dissociation, fever, heart failure, hypotension, mitral regurgitation, myocardial reinfarction, myocardial rupture, nausea and/or vomiting, pericardial effusion, pericarditis, pulmonary edema, recurrent ischemia, thromboembolism.
Pulmonary embolism (PE):
Hypotension, pleural effusion, pulmonary edema, pulmonary re-embolization, thromboembolism.
Cathflo activase:
Gastrointestinal bleeding, sepsis, and venous thrombosis have occurred. There were no reports of intracranial hemorrhage or pulmonary emboli during clinical trials.
Antidote
Systemic alteplase:
Notify physician of all side effects. Note even the most minute bleeding tendency. Oozing at IV sites is expected. Control minor bleeding by local pressure. For severe bleeding in a critical location or suspected intracranial bleeding, discontinue alteplase and any heparin therapy immediately. Obtain PT, aPTT, platelet count, and fibrinogen. Draw blood for type and cross-match. Transfuse as indicated. Consider protamine if heparin has been used. Treat reperfusion arrhythmias with appropriate antiarrhythmic; VT or VF may require cardioversion. If anaphylaxis occurs, discontinue infusion immediately and initiate appropriate treatment. Treat minor hypersensitivity reactions symptomatically. If angioedema occurs, treat promptly with antihistamines (e.g., diphenhydramine [Benadryl]), IV corticosteroids, or epinephrine and consider discontinuing the alteplase infusion; resuscitate as necessary.
Cathflo activase:
Discontinue Cathflo Activase and withdraw it from the catheter if serious bleeding in a critical location (e.g., intracranial, gastrointestinal, retroperitoneal, pericardial) occurs. Discontinue drug and treat anaphylaxis as indicated; resuscitate as necessary. In the event of accidental administration of a 2-mg dose directly into the systemic circulation, the concentration of circulating levels of alteplase would be expected to return to exogenous levels of 5 to 10 ng/mL within 30 minutes.
Amifostine
(am-ih-FOS-teen)
Ethyol
Antidote
Antineoplastic adjunct
Cytoprotective agent
Usual dose
Prophylactic reduction of cisplatin-induced nephrotoxicity, prophylactic reduction of cisplatin-induced neurotoxicity (unlabeled), and prophylactic reduction of antineoplastic agent bone marrow toxicity (unlabeled)
Must be administered in conjunction with cisplatin. Adequate hydration and premedication required before administration; see Premedication below, Monitor, and cisplatin monograph.
Premedication:
Premedication to prevent severe nausea and vomiting is recommended before each dose. Usual regimen includes dexamethasone 20 mg IV and a serotonin 5HT3 receptor antagonist (e.g., ondansetron [Zofran], granisetron [Kytril]) given before and in conjunction with amifostine infusion. Additional antiemetics (e.g., aprepitant [EMEND]) may be required based on the chemotherapy drugs administered.
Amifostine:
910 mg/M2. Cisplatin dose must be given within 30 minutes of starting the amifostine infusion, but only after the full dose of amifostine is administered.
Reduction of moderate to severe xerostomia from radiation of the head and neck
Premedication:
Premedication to prevent nausea and vomiting is recommended before each dose. Oral 5HT3 receptor antagonists alone or in combination with other antiemetics are recommended. Adequate hydration required; see Monitor.
Amifostine:
200 mg/M2 once daily as a 3-minute infusion, starting 15 to 30 minutes before standard fraction radiation therapy.
Dose adjustments
Dosing should be cautious in the elderly. Consider potential for decreased organ function and concomitant disease or drug therapy.
Reduction of cumulative renal toxicity with chemotherapy:
Temporarily discontinue the infusion if the systolic BP decreases significantly from the baseline value. See the following chart.
| Guideline for Interrupting Amifostine Infusion Due to Decrease in Systolic Blood Pressure | |||||
| Baseline Systolic Blood Pressure (mm Hg) | |||||
| <100 | 100-119 | 120-139 | 140-179 | ≥180 | |
| Decrease in systolic blood pressure during infusion of amifostine (mm Hg) | 20 | 25 | 30 | 40 | 50 |

Infusion may be restarted to deliver the full dose if the BP returns to normal within 5 minutes and the patient is asymptomatic. If the BP does not return to baseline within 5 minutes and/or the patient is symptomatic (e.g., bradycardia, fainting, unconscious), the full dose cannot be delivered and subsequent doses should be reduced to 740 mg/M2.
Dilution
Each 500-mg vial should be reconstituted with 9.7 mL of NS (50 mg/mL). May be further diluted with NS to concentrations from 5 to 40 mg/mL. An additional 2.5 mL of NS will yield 40 mg/mL; 90 mL NS will yield 5 mg/mL.
Filters:
Not required by manufacturer. Additional data not available.
Storage:
Store at CRT before reconstitution. Reconstituted or diluted solution prepared in PVC infusion bags is stable for 5 hours at room temperature or 24 hours if refrigerated.
Compatibility
Consider any drug NOT listed as compatible to be INCOMPATIBLE until consulting a pharmacist; specific conditions may apply.
Use of any additive, diluent, or solution other than NS is not recommended by manufacturer.
One source suggests the following compatibilities:
Y-site:
Amikacin, aminophylline, ampicillin, ampicillin/sulbactam (Unasyn), aztreonam (Azactam), bleomycin (Blenoxane), bumetanide, buprenorphine (Buprenex), butorphanol (Stadol), calcium gluconate, carboplatin (Paraplatin), carmustine (BiCNU), cefazolin (Ancef), cefotaxime (Claforan), cefotetan, cefoxitin (Mefoxin), ceftazidime (Fortaz), ceftriaxone (Rocephin), cefuroxime (Zinacef), ciprofloxacin (Cipro IV), clindamycin (Cleocin), cyclophosphamide (Cytoxan), cytarabine (ARA-C), dacarbazine (DTIC), dactinomycin (Cosmegen), daunorubicin (Cerubidine), dexamethasone (Decadron), diphenhydramine (Benadryl), dobutamine, docetaxel (Taxotere), dopamine, doxorubicin (Adriamycin), doxycycline, droperidol (Inapsine), enalaprilat (Vasotec IV), etoposide (VePesid), famotidine (Pepcid IV), fluconazole (Diflucan), fludarabine (Fludara), fluorouracil (5-FU), furosemide (Lasix), gallium nitrate (Ganite), gemcitabine (Gemzar), gentamicin, granisetron (Kytril), heparin, hydrocortisone sodium succinate (Solu-Cortef), hydromorphone (Dilaudid), idarubicin (Idamycin), ifosfamide (Ifex), imipenem-cilastatin (Primaxin), leucovorin calcium, lorazepam (Ativan), magnesium sulfate, mannitol (Osmitrol), mechlorethamine (nitrogen mustard), meperidine (Demerol), mesna (Mesnex), methotrexate, methylprednisolone (Solu-Medrol), metoclopramide (Reglan), metronidazole (Flagyl IV), mitomycin (Mutamycin), mitoxantrone (Novantrone), morphine, nalbuphine, ondansetron (Zofran), pemetrexed (Alimta), potassium chloride (KCl), promethazine (Phenergan), ranitidine (Zantac), sodium bicarbonate, streptozocin (Zanosar), sulfamethoxazole/trimethoprim, teniposide (Vumon), thiotepa, ticarcillin/clavulanate (Timentin), tobramycin, vancomycin, vinblastine, vincristine, zidovudine (AZT, Retrovir).
Rate of administration
Reduction of cumulative renal toxicity with chemotherapy:
A single dose evenly distributed over 15 minutes. Complete amifostine dose but begin cisplatin within 30 minutes after beginning amifostine. Amifostine must be given over 15 minutes; longer infusion times increase the risk of side effects, especially hypotension. Shorter infusion times have not been studied.
Reduction of moderate to severe xerostomia from radiation of the head and neck:
A single dose evenly distributed over 3 minutes. Begin infusion 15 to 30 minutes before standard fraction radiation therapy.
Actions
A cytoprotective agent. Rapidly metabolized by alkaline phosphatase in tissues to an active metabolite that can reduce the nephrotoxic effects of cisplatin and the toxic effects of radiation on normal oral tissues. This protective metabolite is generated in greater amounts in normal tissues versus tumor tissues and is available to bind to and detoxify reactive metabolites of cisplatin and/or radiation. It reduces the incidence of cisplatin toxicity including nephrotoxicity but does not cause other toxic reactions. Its protective metabolite can also scavenge reactive oxygen species generated by exposure to either cisplatin or radiation. May adversely affect antitumor effects of cisplatin. Rapidly cleared from plasma with an elimination half-life of 8 minutes. Pretreatment with antiemetics does not alter its actions. Measurable levels of the metabolite have been found in bone marrow; minimal excretion in urine.
Indications and uses
Reduce the cumulative renal toxicity associated with repeated administration of cisplatin in patients with advanced ovarian cancer. May allow higher cumulative doses of cisplatin and cyclophosphamide. ■ Reduce the incidence of moderate to severe xerostomia (dryness of the mouth from salivary gland dysfunction) in patients undergoing postoperative radiation treatment for head and neck cancer, where the “radiation port” includes a substantial portion of the parotid glands.
Unlabeled uses:
Reduce acute and cumulative hematologic toxicity associated with various chemotherapy regimens (e.g., cisplatin, cyclophosphamide, carboplatin; see literature). ■ Decrease frequency or severity of cisplatin-induced neurotoxicity. ■ Prevention of radiation proctitis in rectal cancer.
Contraindications
Known sensitivity to aminothiol compounds or mannitol. ■ Not recommended for patients receiving chemotherapy for malignancies if chemotherapy can produce significant survival benefit or cure; may interfere with effectiveness of chemotherapy regimen and reduce incidence of cure. ■ Not recommended for patients who are hypotensive, dehydrated, or for those receiving antihypertensive therapy that cannot be stopped for 24 hours before amifostine is administered. ■ Not recommended for patients receiving definitive radiotherapy. Tumor-protective effect in this setting has not been ruled out.
Precautions
Use caution in the elderly and in patients with pre-existing cardiovascular or cerebrovascular conditions (e.g., arrhythmias, congestive heart failure, history of stroke, history of ischemic heart disease). ■ Hypotension and nausea and vomiting can be severe; use caution in any situation in which these side effects may have serious consequences. ■ Discontinue antihypertensive therapy 24 hours before administering amifostine. ■ Hypersensitivity reactions, including anaphylaxis and severe cutaneous reactions, have been reported; see Monitor and Side Effects. Deaths have occurred. ■ Facilities for monitoring the patient and responding to any medical emergency must be available.
Monitor:
All indications:
Adequate hydration required before administration of amifostine. ■ Monitor fluid balance carefully, especially in conjunction with highly emetogenic chemotherapy (e.g., cisplatin). ■ See Usual Dose for premedication requirements. Additional antiemetics may be required to offset nausea and vomiting of chemotherapy drugs. ■ Keep patient in supine position during and immediately after the infusion. ■ Hypersensitivity and/or severe cutaneous reactions may occur during or after infusion; monitor closely before, during, and after administration. Serious cutaneous reactions may develop weeks after initiation of therapy. ■ Monitor serum calcium. Risk of hypocalcemia increased in some patients (e.g., nephrotic syndrome or patients receiving multiple doses). Calcium supplements may be required. ■ See Dose Adjustments, Rate of Administration, and Antidote.
Reduction of cumulative renal toxicity with chemotherapy:
Discontinue antihypertensive therapy if indicated; see Contraindications, Precautions, Drug/Lab Interactions. ■ Obtain baseline BP and monitor at least every 5 minutes during and immediately after the infusion. Continue monitoring BP as indicated. ■ Hypertension may be exacerbated by several causes (e.g., interruption of antihypertensive therapy, IV hydration). Monitor BP closely. ■ Hypotension can occur at any time but is more frequent toward the end of the infusion, and recovery usually begins within 5 to 6 minutes after infusion is discontinued.
Reduction of xerostomia from radiation:
Monitor BP before and immediately after the infusion and as indicated by results.
Patient education:
Void before administration. ■ May produce significant hypotension. Effects may be additive with medications currently being taken. Review all medications (prescription and nonprescription) with nurse and/or physician. ■ Must remain in supine position until BP is stabilized, then request assistance for ambulation. ■ Report feelings of faintness or nausea promptly. ■ Promptly report development of any rash or skin condition.
Maternal/child:
Category C: use only if potential benefits justify risk to fetus; embryotoxic in rabbits at doses lower than required for humans. ■ Discontinue breast-feeding. ■ Safety for use in pediatric patients not established; experience is limited.
Elderly:
Response similar to younger adults; however, dosing should be cautious; see Dose Adjustments. ■ Monitor fluid balance closely; avoid dehydration. ■ Hypotension may be sudden and severe; monitor closely. ■ See Contraindications and Precautions.
Drug/lab interactions
Antihypertensive therapy (e.g., ACE inhibitors [e.g., enalapril], calcium channel blocking agents [e.g., nicardipine, verapamil], diuretics [e.g., furosemide, torsemide], nitroglycerin, nitroprusside sodium) should be discontinued 24 hours before amifostine administration; see Contraindications. ■ Use extreme caution in any patient receiving medications with hypotensive effects (antidepressants, benzodiazepines, beta-adrenergic blocking agents [e.g., atenolol, esmolol, metoprolol, propranolol], lidocaine, magnesium, narcotics, nitrates, paclitaxel, procainamide); will cause additive hypotension.
Side effects
Hypotension and severe nausea and vomiting occur frequently. Hypotension may be associated with apnea, arrhythmias (e.g., atrial fibrillation/flutter, bradycardia, extrasystoles, tachycardia), chest pain, dyspnea, hypoxia, myocardial ischemia, and rarely renal failure, respiratory and cardiac arrest, seizures, and unconsciousness. Dehydration, dizziness, feelings of cold or warmth, flushing, hiccups, hypocalcemia, loss of consciousness, somnolence, and/or transient hypertension or exacerbation of pre-existing hypertension may occur. Hypersensitivity reactions (e.g., anaphylaxis [rare], arrhythmias, chest tightness, chills, cutaneous eruptions, dyspnea, fever, hypoxia, laryngeal edema, pruritus, sneezing, urticaria) have occurred. Most cutaneous eruptions, pruritus, and urticaria were mild; however, serious skin reactions such as erythema multiforme, exfoliative dermatitis, rash, Stevens-Johnson syndrome (rare), toxic epidermal necrolysis, and urticaria have been reported. Serious skin reactions have been reported more frequently when amifostine is used as a radioprotectant.
Overdose:
Hypotension is the most likely symptom. Anxiety and reversible urinary retention have occurred at higher doses. Up to 3 doses have been given within 24 hours without unexpected side effects.
Antidote
Keep physician informed of side effects. Treatment of nausea and vomiting is imperative to encourage patients to continue treatment with full doses of chemotherapeutic agents. Hypotension may be dose-limiting. If the systolic BP decreases significantly (see Dose Adjustments chart), temporarily discontinue the amifostine still infusing, place the patient in Trendelenburg position, and administer an infusion of NS at a separate site. Vasopressors (e.g., dopamine, norepinephrine [Levophed]) may be required. If indicated, restart infusion if BP returns to baseline within 5 minutes and patient is asymptomatic. Discontinue amifostine immediately and permanently if an acute hypersensitivity or cutaneous reaction occurs. Dermatologic consult may be required. Treat anaphylaxis and resuscitate as necessary.
Amikacin sulfate 
(am-ih-KAY-sin SUL-fayt)
Antibacterial (aminoglycoside)
pH 3.5 to 5.5
Usual dose
Up to 15 mg/kg of body weight/24 hr equally divided into 2 or 3 doses at equally divided intervals (5 mg/kg every 8 hours or 7.5 mg/kg every 12 hours). Dosage based on ideal weight of lean body mass. Do not exceed a total adult dose of 15 mg/kg/24 hr in an average weight patient or 1.5 Gm in heavier patients by all routes in 24 hours.
Studies suggest that in certain populations a single daily dose of 15 to 20 mg/kg (instead of divided into 2 or 3 doses) may provide higher peak levels and enhance drug effectiveness while actually reducing or having no adverse effects on risk of toxicity. Various procedures for monitoring blood levels are in use. Some health facilities are monitoring with trough levels; others may draw levels at predetermined times and plot the concentration on nomograms. Depending on the protocol in place, doses or intervals may be adjusted. See Dose Adjustments and Precautions.
Pediatric dose
15 to 22.5 mg/kg/24 hr equally divided into 2 or 3 doses and given every 8 to 12 hours (5 to 7.5 mg/kg every 8 hours or 7.5 to 11.25 mg/kg every 12 hours). Do not exceed 1.5 Gm/24 hr. A single daily dose is also being used in pediatric patients. See comments under Usual Dose.
Newborn dose
See Maternal/Child.
10 mg/kg of body weight as a loading dose, then 7.5 mg/kg/dose. Intervals of 7.5 mg/kg dose adjusted based on age as follows:
Under 28 weeks’ gestation and less than 7 days of age:
Give every 24 hours.
Under 28 weeks’ gestation and over 7 days or 28 to 34 weeks’ gestation and under 7 days of age:
Give every 18 hours.
28 to 34 weeks’ gestation and over 7 days of age or over 34 weeks’ gestation and under 7 days of age:
Give every 12 hours.
Over 34 weeks’ gestation and over 7 days of age:
Give every 8 hours.
Dose adjustments
Reduce daily dose commensurate with amount of renal impairment and/or increase intervals between injections. ■ Once-daily dosing is not usually used in patients with ascites, burns covering more than 20% of the total body surface area, CrCl less than 40 mL/min (including patients requiring dialysis), CrCl greater than 120 mL/min, cystic fibrosis, endocarditis, mycobacterium infections, or in infants or pregnancy. ■ Reduced dose or extended intervals may be required in the elderly. ■ See Drug/Lab Interactions.
Dilution
Each 500 mg or fraction thereof is diluted with 100 to 200 mL D5W, D5NS, or NS. Amount of diluent may be decreased proportionately with dosage for infants and other pediatric patients. Available for pediatric injection as 50 mg/mL.
Storage:
Stable for 24 hours at RT at concentrations of 0.25 and 5 mg/mL when diluted in specific solutions; see prescribing information.
Compatibility (underline indicates conflicting compatibility information)
Consider any drug NOT listed as compatible to be INCOMPATIBLE until consulting a pharmacist; specific conditions may apply.
Do not physically premix with other drugs; administer separately as recommended by manufacturer. Inactivated in solution with beta-lactam antibiotics (e.g., cephalosporins, penicillins) and vancomycin. Do not mix in the same solution. Appropriate spacing required because of physical incompatibilities. See Drug/Lab Interactions.
One source suggests the following compatibilities:
Solution:
Manufacturer lists D5W, D5/1/4NS, D5/1/2NS, NS, LR, D5 in Normosol M (Plasma-Lyte 56 in D5W), D5 in Normosol R (Plasma-Lyte 148 in D5W). Other sources list additional solutions.
Y-site:
Acyclovir (Zovirax), amifostine (Ethyol), amiodarone (Nexterone), anidulafungin (Eraxis), aztreonam (Azactam), bivalirudin (Angiomax), caspofungin (Cancidas), cefepime (Maxipime), ceftaroline (Teflaro), ceftazidime (Fortaz), cisatracurium (Nimbex), cyclophosphamide (Cytoxan), dexamethasone (Decadron), dexmedetomidine (Precedex), diltiazem (Cardizem), docetaxel (Taxotere), doripenem (Doribax), enalaprilat (Vasotec IV), esmolol (Brevibloc), etoposide phosphate (Etopophos), fenoldopam (Corlopam), filgrastim (Neupogen), fluconazole (Diflucan), fludarabine (Fludara), foscarnet (Foscavir), furosemide (Lasix), gemcitabine (Gemzar), granisetron (Kytril), hetastarch in electrolytes (Hextend), hydromorphone (Dilaudid), idarubicin (Idamycin), labetalol, levofloxacin (Levaquin), linezolid (Zyvox), lorazepam (Ativan), magnesium sulfate, melphalan (Alkeran), meperidine (Demerol), midazolam (Versed), milrinone (Primacor), morphine, nicardipine (Cardene IV), ondansetron (Zofran), paclitaxel (Taxol), pemetrexed (Alimta), remifentanil (Ultiva), sargramostim (Leukine), teniposide (Vumon), thiotepa, tigecycline (Tygacil), vinorelbine (Navelbine), warfarin (Coumadin), zidovudine (AZT, Retrovir).
Rate of administration
A single dose over at least 30 to 60 minutes. Infants should receive a 1- to 2-hour infusion.
Actions
An aminoglycoside antibiotic with neuromuscular blocking action. Bactericidal against many gram-negative organisms resistant to other antibiotics including other aminoglycosides such as gentamicin, kanamycin, and tobramycin. Well distributed through all body fluids. Usual half-life is 2 to 3 hours. Crosses the placental barrier. Excreted in the kidneys. Cross-allergenicity does occur between aminoglycosides.
Indications and uses
Short-term treatment of serious infections caused by susceptible organisms (e.g., gram-negative bacteria) generally resistant to alternate drugs that have less potential toxicity. ■ Effective in infections of the respiratory and urinary tracts, CNS (including meningitis), skin and soft tissue, intra-abdominal (including peritonitis), bacterial septicemia (including neonatal sepsis), burns, and postoperative infections. ■ Considered initial therapy in suspected gram-negative infections after culture and sensitivity is drawn. ■ In certain severe infections (e.g., neonatal sepsis), empiric concomitant treatment with an antibiotic effective against gram-positive organisms (e.g., penicillin) may be required until the results of C/S are obtained.
Unlabeled uses:
Treatment of Mycobacterium avium complex, a common infection in AIDS (part of a multiple [3 to 5] drug regimen), tuberculosis.
Contraindications
Known amikacin or aminoglycoside sensitivity. Sulfite sensitivity may be a contraindication.
Precautions
Sensitivity studies indicated to determine susceptibility of causative organism to amikacin. ■ Response should occur in 24 to 48 hours. Safety for use longer than 14 days not established. ■ Superinfection may occur from overgrowth of nonsusceptible organisms. ■ May contain sulfites; use caution in patients with asthma. ■ Single daily dosing has been used effectively in abdominal, pelvic inflammatory, and GU infections in patients with normal renal function. Not recommended in bacteremia caused by Pseudomonas aeruginosa, endocarditis, meningitis, during pregnancy or in patients less than 6 weeks postpartum. Limited data available for use in all other situations (e.g., burns, cystic fibrosis, elderly, pediatrics, renal impairment). ■ Potentially nephrotoxic, ototoxic, and neurotoxic. Risk of nephrotoxicity and neurotoxicity (e.g., auditory and vestibular ototoxicity) increased in patients with pre-existing renal damage or in normal renal function with prolonged use. Partial or total irreversible deafness may continue to develop after amikacin is discontinued. ■ Use with caution in patients with muscular disorders (e.g., myasthenia gravis, parkinsonism) because these drugs may aggravate muscle weakness. ■ Clostridium difficile–associated diarrhea (CDAD) has been reported. May range from mild diarrhea to fatal colitis. Consider in patients who present with diarrhea during or after treatment with amikacin. ■ See Monitor and Drug/Lab Interactions.
Monitor:
Maintain good hydration. ■ Narrow range between toxic and therapeutic levels. Periodically monitor peak and trough concentrations. Manufacturer recommends avoiding peak serum concentrations greater than 35 mcg/mL and trough serum concentrations above 10 mcg/mL. ■ Monitor urine protein, presence of cells and casts, and decreased specific gravity. Watch for decreased urine output, rising BUN and SCr, and declining CrCl levels. Dose adjustment may be necessary. ■ Closely monitor patients with impaired renal function for nephrotoxicity and neurotoxicity (e.g., auditory and vestibular ototoxicity); nephrotoxicity may be reversible. ■ In extended treatment, monitor serum levels, electrolytes, and renal, auditory, and vestibular functions frequently. ■ See Drug/Lab Interactions.
Patient education:
Report promptly any changes in balance, hearing loss, weakness, or dizziness. ■ Consider birth control options. ■ Promptly report diarrhea or bloody stools that occur during treatment or up to several months after an antibiotic has been discontinued; may indicate CDAD and require treatment.
Maternal/child:
Category D: avoid pregnancy. Potential hazard to fetus. ■ Safety for use during breast-feeding not established; use extreme caution. ■ Peak concentrations are generally lower in infants and young children. ■ Use extreme caution in premature infants and neonates; immature kidney function will result in prolonged half-life.
Elderly:
Consider less toxic alternatives. ■ Longer intervals between doses may be more important than smaller doses. ■ Monitor renal function and drug levels carefully. Measurement of CrCl more useful than BUN or SCr to assess renal function. ■ Half-life prolonged.
Drug/lab interactions
Synergistic when used in combination with beta-lactam antibiotics (e.g., cephalosporins, penicillins) and vancomycin. Synergism may be inconsistent; see Compatibility. ■ Concurrent use topically or systemically with any other ototoxic or nephrotoxic agents should be avoided. May have dangerous additive effects with anesthetics (e.g., enflurane), other neuromuscular blocking antibiotics (e.g., kanamycin), diuretics (e.g., furosemide [Lasix]), beta-lactam antibiotics (e.g., cephalosporins), vancomycin, and many others. ■ Neuromuscular blocking muscle relaxants (e.g., atracurium [Tracrium], succinylcholine) are potentiated by aminoglycosides. Apnea can occur. ■ Aminoglycosides are potentiated by anticholinesterases (e.g., edrophonium), antineoplastics (e.g., nitrogen mustard, cisplatin).
Side effects
Occur more frequently with impaired renal function, higher doses, prolonged administration, in dehydrated or elderly patients, and in patients receiving other ototoxic or nephrotoxic drugs. Fever, headache, hypotension, nausea, paresthesias, seizures, skin rash, tremor, vomiting.
Major:
Albuminuria, anemia, arthralgia, azotemia, CDAD, eosinophilia, loss of balance, neuromuscular blockade, oliguria, ototoxicity, RBCs and WBCs or casts in urine, respiratory depression or arrest, rising SCr.
Antidote
Notify physician of all side effects. If minor side effects persist or any major symptom appears, discontinue drug and notify physician. Treatment is symptomatic, or a reduction in dose may be required. In overdose hemodialysis may be indicated. Monitor fluid balance, CrCl, and plasma levels carefully. Complexation with ticarcillin may be as effective as hemodialysis. Consider exchange transfusion in the newborn. Calcium salts or neostigmine may reverse neuromuscular blockade. Treat CDAD with fluids, electrolytes, protein supplements, and oral vancomycin (Vancocin) or metronidazole (Flagyl) as indicated. In severe cases, surgical evaluation may be indicated. Resuscitate as necessary.
Aminocaproic acid
(a-mee-noh-ka-PROH-ick AS-id)
Amicar
Antifibrinolytic
Antihemorrhagic
pH 6 to 7.6
Usual dose
4 to 5 Gm initially over 1 hour. Follow with 1 Gm/hr for 8 hours or until bleeding is controlled. In acute bleeding syndromes, the 4- to 5-Gm dose may be given as a continuous infusion over the first hour, followed by a continuous infusion of 1 Gm/hr for 8 hours or until bleeding is controlled. Maximum dose is 30 Gm/24 hr.
Prevent recurrence of subarachnoid hemorrhage (unlabeled):
36 Gm/24 hr. One source suggests administering 18 Gm in 400 mL D5W every 12 hours for 10 days. Follow with oral therapy.
Prevention of perioperative bleeding during cardiac surgery (unlabeled):
10 Gm as an infusion over 20 to 30 minutes before skin incision. Follow with 1 to 2.5 Gm/hr (usually 2 Gm/hr) until the end of the operation. Infusion may be continued for 4 hours after protamine reversal of heparin. In addition, 10 Gm may be added to the cardiopulmonary bypass circuit priming solution. An alternate regimen is 10 Gm over 20 to 30 minutes before skin incision, followed by 10 Gm after heparin administration, then 10 Gm when cardiopulmonary bypass is discontinued and before protamine reversal of heparin. Another source suggests a loading dose of 80 mg/kg over 20 minutes followed by 30 mg/kg/hr, or a loading dose of 60 mg/kg over 20 minutes followed by 30 mg/kg/hr plus a 10-mg/kg dose in the priming solution of the cardiopulmonary bypass pump.
Pediatric dose
Acute bleeding syndromes (unlabeled):
See Maternal/Child. Loading dose: 100 to 200 mg/kg. Follow with a maintenance dose of 100 mg/kg/dose every 4 to 6 hours. Maximum dose is 30 Gm/24 hr.
Dose adjustments
May accumulate in patients with renal impairment, and reduced doses are suggested; however, specific recommendations are not available from the manufacturer. Another source suggests decreasing dose to 15% to 25% of the normal dose in patients with renal impairment.
Dilution
1 Gm equals 4 mL of prepared solution. Further dilute with compatible infusion solutions (NS, D5W, SWFI, or Ringer’s solution). Up to 50 mL of diluent may be used for each 1 Gm.
Storage:
Before use store at CRT. Do not freeze.
Compatibility
Compatible in D5W, Ringer’s solution, and NS. One source suggests compatibility at the Y-site with fenoldopam (Corlopam).
Rate of administration
5 Gm or fraction thereof over first hour in 250 mL of solution; then administer each succeeding 1 Gm over 1 hour in 50 to 100 mL of solution. Use of an infusion pump for accurate dose recommended. Rapid administration or insufficient dilution may cause hypotension, bradycardia, and/or arrhythmia.
Actions
A 6-aminohexanoic acid that acts as an inhibitor of fibrinolysis. Inhibits plasminogen activator substances; to a lesser degree inhibits plasmin activity. Increases fibrinogen activity in clot formation by inhibiting the enzyme required for destruction of formed fibrin. Onset of action is prompt, but will last less than 3 hours. Partially metabolized. Half-life is 2 hours. Excreted in urine. Easily penetrates RBCs and tissue cells after prolonged administration.
Indications and uses
Useful in enhancing hemostasis when fibrinolysis contributes to bleeding. ■ Treatment of fibrinolytic bleeding, which may be associated with surgical complications following heart surgery (with or without cardiac bypass procedures) and portacaval shunt, hematologic disorders such as aplastic anemia, acute and life-threatening abruptio placentae, hepatic cirrhosis, and neoplastic disease such as carcinoma of the prostate, lung, stomach, and cervix. ■ Urinary fibrinolysis (normal physiologic phenomenon), which may result from severe trauma, anoxia, shock, surgical hematuria complications following prostatectomy and nephrectomy, or nonsurgical hematuria resulting from polycystic or neoplastic disease of the GU system.
Unlabeled uses:
Prevent recurrence of subarachnoid hemorrhage. ■ Control of bleeding in thrombocytopenia. ■ Prevention of perioperative bleeding during cardiac surgery. ■ Prophylaxis and treatment during dental surgical procedures (hemophilia and/or hemorrhage).
Contraindications
Evidence of an active intravascular clotting process. ■ Uncertainty as to whether the cause of bleeding is primary fibrinolysis (PF) or disseminated intravascular coagulation (DIC). This distinction must be made before administration; see Precautions. ■ Do not use aminocaproic acid in the presence of DIC without concomitant heparin.
Precautions
Should not be administered without a definite diagnosis and/or lab findings indicative of hyperfibrinolysis. ■ The following tests are used to differentiate primary fibrinolysis (PF) from disseminated intravascular coagulation (DIC). Platelet count should be normal in PF but is usually decreased in DIC; protamine paracoagulation test is negative in PF and positive in DIC (a precipitate forms when protamine sulfate is dropped into citrated plasma); euglobulin clot lysis test is abnormal in PF but normal in DIC. ■ In life-threatening situations, transfusion and other appropriate emergency measures may be required. ■ Avoid use in patients with hematuria of upper urinary tract origin. Has caused glomerular capillary thrombosis in the renal pelvis and ureters, leading to intrarenal obstruction. ■ Use with caution in patients with cardiac disease. May cause hypotension and bradycardia. Endocardial hemorrhage and fatty degeneration of the myocardium have been reported in animals. ■ Use with caution in patients with hepatic disease. Etiology of bleeding may be multifactorial and difficult to diagnose. ■ Use with caution in patients with renal impairment; see Dose Adjustments. Kidney stones have been reported in animal studies. ■ Skeletal muscle weakness with necrosis of muscle fibers has been reported after prolonged use; see Monitor. ■ An increased incidence of certain neurologic deficits (e.g., cerebral ischemia, cerebral vasospasm, hydrocephalus) associated with the use of antifibrinolytic agents in the treatment of subarachnoid hemorrhage has been reported. Relationship to drug therapy versus natural disease process or diagnostic procedures (e.g., angiography) is unclear.
Monitor:
See Precautions and Contraindications. ■ Use only in conjunction with general and specific tests to determine the amount of fibrinolysis present (e.g., fibrinogen, PT, aPTT). ■ Monitor lab evaluations as appropriate for diagnosis (e.g., platelet count, clotting factors, CPK, AST). ■ Vital signs, intake and output, any signs of bleeding, and neurologic assessment should be monitored based on patient condition. ■ Observe for thromboembolic complications (e.g., chest pain, dyspnea, edema, hemoptysis, leg pain, or positive Homans’ sign). ■ Monitor for S/S of skeletal muscle damage. May range from mild myalgias with weakness to severe proximal myopathy with rhabdomyolysis, myoglobinuria, and acute renal failure. ■ Monitor lab evaluations as appropriate for diagnosis (e.g., platelet count, clotting factors, CPK, AST).
Patient education:
Move slowly with help to avoid orthostatic hypotension.
Maternal/child:
Category C: safety for use in pregnancy and breast-feeding not established. ■ Safety for use in pediatric patients not established but is used. ■ Contains benzyl alcohol, which has been associated with “gasping syndrome” in neonates (sudden onset of gasping respirations, hypotension, bradycardia, and cardiovascular collapse).
Elderly:
Consider age-related impaired organ function; reduced dose may be indicated.
Drug/lab interactions
Potential for thrombus formation increased with concurrent use of estrogens. ■ Frequently used with clotting factor complexes (e.g., factor IX complex, anti-inhibitor coagulant complex), but risk of thrombus formation may be increased. Delay administration for 8 or more hours after clotting factor complexes. ■ Prolongation of template bleeding time has been reported during continuous infusions exceeding 24 Gm/day.
Side effects
Generally well tolerated. Abdominal pain, agranulocytosis, bradycardia, coagulation disorder, confusion, cramps, decreased vision, diarrhea, dizziness, dyspnea, edema, grand mal seizure, hallucinations, headache, hypersensitivity reactions (including anaphylaxis), hypotension, increased BUN and CPK, injection site reactions, intracranial hypertension, leukopenia, malaise, muscle weakness, myalgia, myopathy, myositis, nausea, peripheral ischemia, pruritus, pulmonary embolism, renal failure, rhabdomyolysis, rash, stuffy nose, stroke, syncope, tearing, thrombocytopenia, thrombophlebitis, thrombosis, tinnitus, vomiting.
Overdose:
Acute renal failure, convulsions, death.
Antidote
Treat side effects symptomatically. Discontinue use of drug with any suspicion of thrombophlebitis, thromboembolic complications, or if CPK is elevated (myopathy). In life-threatening situations, fresh whole blood transfusions, fibrinogen infusions, and other emergency measures may be required. May be removed by hemodialysis or peritoneal dialysis.
Aminophylline
(am-ih-NOFF-ih-lin)
(79% Theophylline), Theophylline Ethylenediamine
Bronchodilator
Respiratory stimulant
pH 8.6 to 9
Usual dose
To obtain maximum benefit with minimal risk of adverse effects, dosing must be individualized based on serum theophylline concentration and patient response. Monitor frequently to avoid toxicity. Only aminophylline premixed in solution or aminophylline containing 20 mg of theophylline for each 25 mg of aminophylline is intended for IV use (approximately 79% theophylline). All doses are based on lean body weight; theophylline does not distribute into fatty tissue. All doses listed are milligrams of aminophylline to be administered.
Bronchodilation in acute asthma or bronchospasm
With an average mean volume of distribution of 0.5 L/kg (range is 0.3 to 0.7 L/kg), each mg/kg of theophylline given over 30 minutes should result in an average 2 mcg/mL increase in serum theophylline concentration.
Adults, children, infants and neonates who have not received a theophylline preparation in the previous 24 hours:
An initial loading dose of 5 to 6 mg/kg of lean body weight (5.7 mg of aminophylline is equal to 4.6 mg/kg of theophylline) should produce a serum concentration of 10 mcg/mL (range 6 to 16 mcg/mL). Measure serum theophylline concentration in 30 minutes to determine if additional loading doses are indicated. Once a serum concentration of 10 to 15 mcg/mL is obtained with loading dose(s), it should be maintained with a continuous infusion. Rate of infusion is based on the pharmacokinetic parameters (e.g., volume of distribution, clearance, concomitant disease states) of the specific patient population and should achieve a target serum concentration of 10 mcg/mL. See Dose Adjustments and Monitor for recommendations of serum theophylline testing after an infusion is started.
Adults, children, infants and neonates who have received a theophylline preparation in the previous 24 hours: a serum theophylline concentration must be obtained before considering any loading dose.
Calculate the appropriate loading dose with the following formula.
D = (Desired C – Measured C) × (V)
D is the loading dose, C is the serum theophylline concentration, and V is the volume of distribution (0.5 L/kg). Desired serum concentration in this situation should be conservative (e.g., 10 mcg/mL) to allow for variability in the volume of distribution; carefully evaluate the patient condition and risk versus benefit. It is not recommended by the manufacturer, but another source suggests that a smaller loading dose may be considered if it is not immediately possible to obtain a theophylline serum concentration. Carefully evaluate the patient’s condition and risk versus benefit. The potential for theophylline toxicity must be ruled out. For example, if significant respiratory distress is present, a smaller loading dose of 2.5 mg/kg should increase the theophylline level by approximately 5 mcg/mL. In the absence of toxicity, this increase is unlikely to cause significant side effects and may improve the clinical picture. In all situations, measure serum theophylline concentration in 30 minutes to determine if additional loading doses are indicated. Once a serum concentration of 10 to 15 mcg/mL is obtained with or without loading dose(s), it should be maintained with a continuous infusion. Rate of infusion is based on the pharmacokinetic parameters (e.g., volume of distribution, clearance, concomitant disease states) of the specific patient population and should achieve a target serum concentration of 10 mcg/mL. See Dose Adjustments and Monitor for recommendations of serum theophylline testing after an infusion is started.
Maintenance infusion:
Desired theophylline serum concentration is 10 mcg/mL. Most maintenance doses can be reduced within the first 12 hours based on serum theophylline levels and depending on patient condition and response; see Dose Adjustments. Because of a large interpatient variability in theophylline clearance, each patient may differ from the mean value used to calculate these infusion rates. Another serum concentration is recommended one expected half-life after starting the continuous infusion; see Dose Adjustments.
| Aminophylline Infusion Rates Following an Appropriate Loading Dose | |
| Patient Population | Aminophylline Infusion Rate in mg/kg/hra (Actual theophylline administered in mg/kg/hr is in parentheses) |
| Neonates up to 24 days of age | 1.27 mg/kg every 12 hours (1 mg/kg every 12 hours)b |
| Neonates over 24 days of age | 1.9 mg/kg every 12 hours (1.5 mg/kg every 12 hours)b |
| Infants 6 to 52 weeks of age | mg/kg/hr = [(0.008 × Age in weeks) + 0.21c] ÷ 0.79 |
| Children 1 to 9 years | 1 mg/kg/hr (0.8 mg/kg/hr) |
| Children 9 to 12 years | 0.875 mg/kg/hr (0.7 mg/kg/hr) |
| Adolescent smokers 12 to 16 years | 0.875 mg/kg/hr (0.7 mg/kg/hr) |
| Adolescent nonsmokers 12 to 16 years | 0.625 mg/kg/hr (0.5 mg/kg/hr)d |
| Adults (healthy nonsmokers 16 to 60 years) | 0.5 mg/kg/hr (0.4 mg/kg/hr)d |
| Elderly over 60 years | 0.375 mg/kg/hr (0.3 mg/kg/hr)e |
| Cardiac decompensation, cor pulmonale, liver dysfunction, sepsis with multiorgan failure, or shock | 0.25 mg/kg/hr (0.2 mg/kg/hr)e |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


