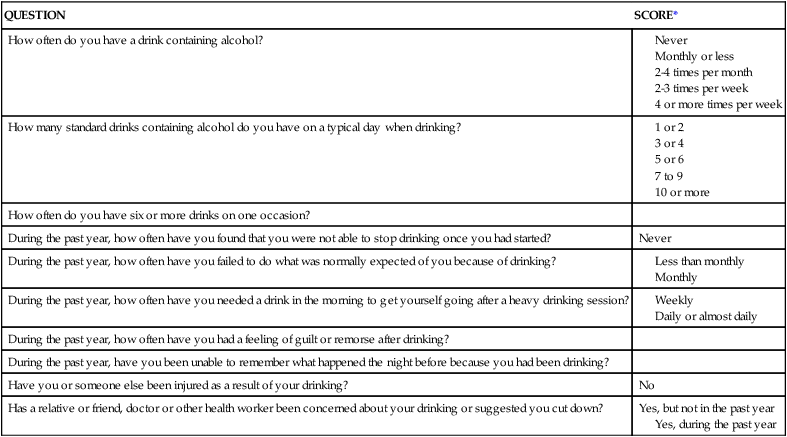
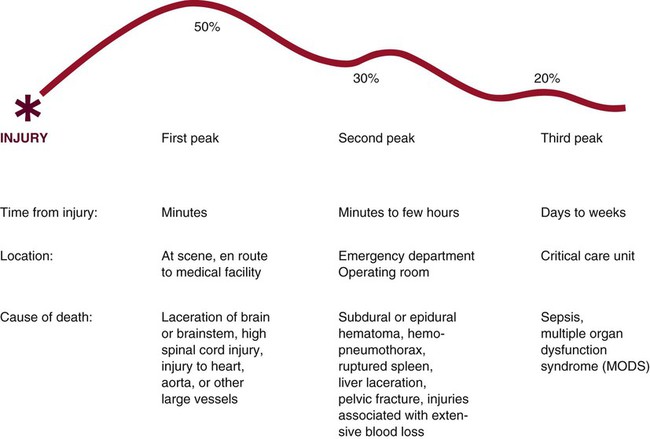
Intimate partner violence (IPV) constitutes a major public health issue in the United States. IPV is the leading cause of injury to women, in the United States it is estimated that 4.8 million women and 2.9 million men every year are raped or physically assaulted each year.1 Up to one third of injuries from IPV result in medical attention being sought and 21% of those who present with acute injuries require emergency surgery.2 The Joint Commission has a standard of practice that all patients must be screened for IPV as part of the health history.3 Studies have shown that screening for IPV in healthcare settings is effective in identifying women who are victims and that patients are not offended when asked about current or past IPV.4 Key interventions for the victim of IPV are listed in Box 34-1. In 2010, 10,228 people were killed in alcohol-impaired driving crashes, which account for 31% of the total motor vehicle traffic fatalities in the United States.5 Each year, alcohol-related crashes in the United States cost about $51 billion.6 Alcohol screening and intervention have been recommended as routine components of trauma care.7 The Alcohol Use Disorders Identification Test (AUDIT) (Table 34-1) is a screening questionnaire that can be used because it can be used to identify frequency of alcoholic drinking and problem drinking.8 TABLE 34-1 AUDIT ALCOHOL SCREENING QUESTIONNAIRE AUDIT, Alcohol Use Disorders Identification Test. *Scores for each question range from 0 to 4, with the first response for each question (never) scoring 0, the second (less than monthly) scoring 1, the third (monthly) scoring 2, the fourth (weekly) scoring 3, and the fifth response (daily or almost daily) scoring 4. For the last two questions, which only have three responses, the scoring is 0, 2, and 4. A score of 8 or more is associated with harmful or hazardous drinking, and a score of 13 or more by women or 15 or more by men is likely to indicate alcohol dependence. Penetrating injuries occur with stabbings, firearms, or impalement—injuries that penetrate the skin and result in damage to internal structures. Damage is created along the path of penetration. Penetrating injuries can be misleading inasmuch as the condition of the outside of the wound does not determine the extent of internal injury. Bullets can create internal cavities 5 to 30 times larger than the diameter of the bullet.9 Statistics demonstrate that deaths as a result of trauma occur in a trimodal distribution (Fig. 34-1).9 The first peak includes victims who die before medical attention can be provided. The second peak occurs within a few hours after injury. This peak commonly is referred to as the golden hour for those critically injured. The golden hour is a 60-minute time frame that incorporates activation of the EMS system, stabilization in the prehospital setting, transportation to a medical facility, rapid resuscitation on arrival in the emergency department, and provision of definitive care. For the critically injured patient, the primary goal is to minimize the time from injury to definitive care. The third death peak occurs days to weeks after injury as a result of complications, including infection or multiple organ dysfunction syndrome (MODS). It is a nursing challenge to influence the quality of care the trauma patient receives in an attempt to “beat” the trimodal distribution of trauma deaths. The goal of prehospital care is immediate stabilization and transportation. This is achieved through airway maintenance, control of external bleeding and shock, immobilization of the patient, and immediate transport (ground or air) to the closest appropriate medical facility.9 Prehospital personnel should communicate information needed for triage at the hospital. Advanced planning for the injured patient is essential. The American College of Surgeons developed guidelines (advanced trauma life support [ATLS]) for rapid assessment, resuscitation, and definitive care for trauma patients in the emergency department.9 These guidelines delineate a systematic approach to care of the trauma patient: rapid primary survey, resuscitation of vital functions, more detailed secondary survey, and initiation of definitive care. This process constitutes the ABCDEs of trauma care and assists in identifying injuries. On arrival of the trauma patient in the emergency department, the primary survey is initiated. During this assessment, life-threatening injuries are discovered and treated. The five steps in the trauma primary survey are performed in ABCDE sequence (Table 34-2): TABLE 34-2 PRIMARY SURVEY OF THE TRAUMA PATIENT ACLS, Advanced cardiac life support; ECG, electrocardiogram; IV, intravenous line. The patient’s airway is assessed for ineffective airway clearance and airway obstruction. The trauma patient is at risk for ineffective airway clearance, especially in the presence of altered consciousness, drugs and alcohol, and maxillofacial or thoracic injuries. Airway obstruction can be caused by foreign bodies, blood clots, or broken teeth. Airway patency should be assessed by inspecting the oropharynx for foreign body obstruction, listening for air movement at the nose and mouth, and auscultation of lung fields. Airway assessment must incorporate cervical spine immobilization. The patient’s head should not be rotated, hyperflexed, or hyperextended to establish and maintain an airway. The cervical spine must be immobilized in all trauma patients until a cervical spinal cord injury has been definitively ruled out. If the patient can verbally communicate, it is likely that the airway is patent. Patients who display nonpurposeful motor movements or who have a Glasgow Coma Scale (GCS) score of 8 or less usually require the placement of a definitive airway.9 The patient is assessed for ineffective breathing patterns and impaired gas exchange; an open, clear airway does not ensure adequate ventilation and gas exchange. Assessment includes chest wall integrity and respiratory rate, depth, and symmetry. Auscultation is performed to assess gas flow in the lungs. Air or blood in the chest may be identified by percussion. Decreased breath sounds or alteration in chest wall integrity necessitate chest tube placement. Endotracheal intubation may be required for patients who have compromised airways caused by mechanical factors, who are unconscious, or who have ventilatory problems.9 Supplemental oxygen is administered to all injured patients.9 The next step is to assess for decreased cardiac output, impaired tissue perfusion, and deficient fluid volume. External exsanguination is identified and controlled by direct manual pressure on the wound. Rapid assessment of the circulatory status includes assessment of level of consciousness, skin color, and pulse.9 Level of consciousness provides data on cerebral perfusion. Ashen, gray facial skin color or white, pale extremities may be ominous signs of hypovolemia.9 Central pulses (femoral or carotid artery) are assessed bilaterally for rate, regularity, and quality. If a pulse is not present, advanced cardiac life support (ACLS) protocols are instituted. ECG monitoring is initiated to assess for rhythm disturbances. Life-threatening dysrhythmias are treated according to ACLS protocols. After the primary survey the resuscitation phase begins. Hypovolemic shock is the most common type of shock that occurs in trauma patients.9 Hemorrhage must be identified and treated rapidly. Two large-bore (14- to 16-gauge) peripheral intravenous catheters, intraosseous catheter, or central venous catheter is inserted. During the initiation of intravenous lines, blood samples are drawn (Box 34-2). Resuscitation is aimed at ensuring adequate perfusion of tissues with oxygen and nutrients to support cellular function. Resuscitation end points (variables or parameters) must be viewed across the continuum of resuscitation from shock. During resuscitation from traumatic hemorrhagic shock, normalization of standard clinical parameters such as blood pressure, heart rate, and urine output are not adequate. The optimal resuscitation end point is a major focus of research in trauma care. During resuscitation, attempts should be made to improve cellular oxygenation. Base deficit and lactate and other assessment parameters have been well-studied to determine adequacy of cellular oxygenation during trauma resuscitation.10 Resuscitation practices have been a focus for research in trauma care. Damage control resuscitation (DCR) is an emerging concept in trauma care. DCR is a strategy to provide only interventions to control hemorrhage and contamination. The strategy involves permissive hypotension, the use of blood products over isotonic fluid for volume replacement, and the rapid and early correction of coagulopathy with component therapy and begins in the field and continues through the emergency departments, operating rooms, and critical care units.11 Given the emphasis on use of blood products over crystalloids and correction of traumatic coagulopathy, many trauma centers have developed massive transfusion protocols for the 1% to 3% of all trauma patients who require DCR.12 Having a defined protocol serves as a system-based strategy to facilitate early, timely release of blood products in what can be an often-chaotic situation. The massive transfusion protocol outlines the ratio of packed red blood cells, fresh frozen plasma, platelets, and cryoprecipitate to be administered.12 The optimal ratio for these blood components has not yet been determined and is a focus for research. Additional interventions during the resuscitation phase involve placement of urinary and gastric catheters. A gastric tube is inserted to reduce gastric distention and lower the risk of aspiration.9 The secondary survey begins when the primary survey is completed, resuscitation is well established, and the patient is demonstrating normalization of vital signs. During the secondary survey, a head-to-toe approach is used to thoroughly examine each body region. The history is one of the most important aspects of the secondary survey. Often, head injury, shock, or the use of drugs or alcohol may preclude a good history, so the history must be pieced together from other sources. The prehospital providers (paramedics, emergency medical technicians) usually can provide most of the vital information pertaining to the unintentional injury. Specific information that must be elicited pertaining to the mechanism of injury is summarized in Box 34-3. This information can help predict internal injuries and facilitate rapid intervention. The patient’s pertinent past history can be assessed by use of the mnemonic AMPLE: Critically ill trauma patients are admitted into the critical care unit as direct transfers from the emergency department or operating room. Information the nurse must obtain from the emergency department or operating room nurse, or both, is summarized using the SBAR method: Situation, Background, Assessment, and Recommendations (Box 34-4). This information must be obtained before the patient’s admission to the critical care unit to ensure availability of needed personnel, equipment, and supplies. This information also helps the nurse to assess the impact of trauma resuscitation on the patient’s presentation and course. Table 34-3 summarizes the prehospital, emergency department, and operating room resuscitative measures that can affect the trauma patient’s care. TABLE 34-3 EFFECTS OF TRAUMA RESUSCITATION ARDS, Acute respiratory distress syndrome; MODS, multiple organ dysfunction syndrome. One of the most important nursing roles is assessment of the balance between oxygen delivery and oxygen demand. Oxygen delivery must be optimized to prevent further system damage. The trauma patient is at high risk for impaired oxygenation as a result of a variety of factors (Table 34-4). Risk factors must be promptly identified and treated to prevent life-threatening sequelae. Prevention and treatment of hypoxemia depend on accurate assessment of the adequacy of pulmonary gas exchange, oxygen delivery, and oxygen consumption. TABLE 34-4 FACTORS PREDISPOSING THE TRAUMA PATIENT TO IMPAIRED OXYGENATION More than 1.7 million traumatic brain injuries (TBIs) occur annually; approximately 52,000 Americans die each year of TBI with 275,000 hospitalized as a result of their injury.13 Children aged 0 to 4 years, older adolescents aged 15 to 19 years, and adults aged 65 years and older are most likely to sustain a TBI.13 TBIs occur when mechanical forces are transmitted to brain tissue. Mechanisms of injury include penetrating or blunt trauma to the head. The leading causes of TBI include falls (35%), MVCs (17%), struck by or against objects (17%), and assaults (10%).14 Penetrating trauma can result from the penetration of a foreign object such as a bullet, which causes direct damage to cerebral tissue. Blunt trauma can be the result of deceleration, acceleration, or rotational forces. Deceleration injury causes the brain to crash against the skull after it has hit a hard surface such as the dashboard of a car. Acceleration injury occurs when the brain has been forcefully hit, such as with a baseball bat. In many instances, TBIs can be caused by acceleration and deceleration. Acceleration injuries occur when the skull is hit by a force that causes the brain to move forward to the point of impact, and then as the brain reverses direction and hits the other side of the skull, deceleration injuries occur. Secondary injury is the biochemical and cellular response to the initial trauma that can exacerbate the primary injury and cause loss of brain tissue not originally damaged. Secondary injury can be caused by ischemia, hypercapnia, hypotension, cerebral edema, sustained hypertension, calcium toxicity, or metabolic derangements. Hypoxia or hypotension, the best known culprits for secondary injury, typically are the result of extracranial trauma.15 A self-perpetuating cycle develops that may cause the expansion of a relatively focal primary injury as a result of uncontrolled refractory secondary injury.9,15 Initial hypertension in the patient with severe TBI is common. As a result of the loss of autoregulation, increased blood pressure results in increased intracranial blood volume and elevates ICP. Every effort must be made to control hypertension to prevent the secondary injury caused by increased ICP (see “Intracranial Pressure Monitoring” in Chapter 24 and “Intracranial Hypertension” in Chapter 25). The effects of increased ICP are varied. As pressure increases inside the closed skull vault, cerebral perfusion decreases, which further compromises the brain. The combined effects of increasing pressure and decreasing perfusion precipitate a downward spiral of events. A concussion is a brain injury accompanied by a brief loss of neurologic function, especially loss of consciousness.14 When loss of consciousness occurs, it may last for seconds to an hour. The neurologic dysfunctions include confusion, disorientation, and sometimes a period of antegrade or retrograde amnesia. Other clinical manifestations that occur after concussion are headache, dizziness, nausea, irritability, inability to concentrate, impaired memory, and fatigue. The diagnosis of concussion is based on the loss of consciousness inasmuch as the brain remains structurally intact despite functional impairment. Contusion, or “bruising” of the brain, usually is related to acceleration–deceleration injuries, which result in hemorrhage into the superficial parenchyma. Frontal or temporal lobe contusions are most common and can be seen in a coup–contrecoup mechanism of injury (Fig. 34-2). Coup injury affects the cerebral tissue directly under the point of impact. Contrecoup injury occurs in a line directly opposite the point of impact. The clinical manifestations of contusion are related to the location of the contusion, the degree of contusion, and the presence of associated lesions. Contusions can be small, in which localized areas of dysfunction result in a focal neurologic deficit. Larger contusions can evolve over 2 to 3 days after injury as a result of edema and further hemorrhaging. A large contusion can produce a mass effect that can cause a significant increase in ICP. Contusions are almost always associated with subdural hematoma (SDH).9 Extravasation of blood creates a space-occupying lesion within the cranial vault that can lead to increased ICP. Three types of hematomas are discussed here (Fig. 34-3). The first two, epidural and SDH, are extraparenchymal (outside of brain tissue) and produce injury by pressure effect and displacement of intracranial contents. The third type, intracerebral hematoma, directly damages neural tissue and can produce further injury as a result of pressure and displacement of intracranial contents. Epidural hematoma (EDH) is a collection of blood between the inner skull and the outermost layer of the dura (Fig. 34-3A). EDHs are most often associated with patients with skull fractures and middle meningeal artery lacerations (two thirds of patients) or skull fractures with venous bleeding.9 A blow to the head that causes a linear skull fracture on the lateral surface of the head may tear the middle meningeal artery. As the artery bleeds, it pulls the dura away from the skull, creating a pouch that expands into the intracranial space. The classic clinical manifestations of EDH include brief loss of consciousness followed by a period of lucidity. Rapid deterioration in the level of consciousness should be anticipated because arterial bleeding into the epidural space can occur quickly. A dilated and fixed pupil on the same side as the impact area is a hallmark of EDH.9 The patient may complain of a severe, localized headache and may be sleepy. Diagnosis of EDH is based on clinical symptoms and evidence of a collection of epidural blood identified on the CT scan. Treatment of EDH requires surgical intervention to remove the blood and to cauterize the bleeding vessels. Subdural hematoma (SDH), which is the accumulation of blood between the dura and underlying arachnoid membrane, most often is related to a rupture in the bridging veins between the cerebral cortex and the dura (Fig. 34-3B). Acceleration–deceleration and rotational forces are the major causes of SDH, which often is associated with cerebral contusions and intracerebral hemorrhage. SDH is common, representing about 30% of severe head injuries. The three types of SDH—acute, subacute, and chronic—are based on the timeframe from injury to clinical symptoms. Intracerebral hematoma (ICH) results when bleeding occurs within cerebral tissue. Traumatic causes of ICH include depressed skull fractures, penetrating injuries (bullet, knife), or sudden acceleration–deceleration motion. The ICH can act as a rapidly expanding lesion; however, late ICH into the necrotic center of a contused area also is possible (Fig. 34-3C). Sudden clinical deterioration of a patient 6 to 10 days after trauma may be the result of ICH. Missile injuries are caused by objects that penetrate the skull to produce a significant focal damage but little acceleration–deceleration or rotational injury. The injury may be depressed, penetrating, or perforating (Fig. 34-4). Depressed injuries are caused by fractures of the skull, with penetration of bone into cerebral tissue. Penetrating injury is caused by a missile that enters the cranial cavity but does not exit. A low-velocity penetrating injury (knife) may involve only focal damage and no loss of consciousness. A high-velocity missile (bullet) can produce shock waves that are transmitted throughout the brain in addition to the injury caused by the bullet. Perforating injuries are missile injuries that enter and then exit the brain. Perforating injuries have much less ricochet effect but are still responsible for significant injury. The neurologic assessment is the most important tool for evaluating the patient with a severe TBI, because it can indicate the severity of injury, provide prognostic information, and dictate the speed with which further evaluation and treatment must proceed.16 The cornerstone of the neurologic assessment is the GCS, although it is not a complete neurologic examination. Pupils and motor strength assessment must be incorporated into the early and ongoing assessments. After injuries are specifically identified, a more thorough, focused neurologic assessment, such as examination of the cranial nerves, is warranted. To assist with the initial assessment, TBIs are divided into three descriptive categories—mild, moderate, or severe—on the basis of the patient’s GCS score and duration of the unconscious state. Level of consciousness, motor movements, pupillary response, respiratory function, and vital signs are all part of a complete neurologic assessment of the patient with a TBI. Level of consciousness can be elicited to assess wakefulness. Consciousness is assessed by obtaining the patient’s response to verbal and painful stimuli. Determination of orientation to person, place, and time assesses mental alertness. Pupils are assessed for size, shape, equality, and reactivity. Asymmetry must be reported immediately. Pupils are also assessed for constriction to a light source (parasympathetic innervation) or dilation (sympathetic innervation). Because parasympathetic fibers are present in the brainstem, pupils that are slow to react to light may indicate a brainstem injury. A “blown” pupil can be caused by compression of the third cranial nerve or transtentorial herniation. Bilateral fixed pupils can indicate midbrain involvement (see “Pupillary Function” in Chapter 23).
Trauma
QUESTION
SCORE*
How often do you have a drink containing alcohol?
How many standard drinks containing alcohol do you have on a typical day when drinking?
How often do you have six or more drinks on one occasion?
During the past year, how often have you found that you were not able to stop drinking once you had started?
Never
During the past year, how often have you failed to do what was normally expected of you because of drinking?
During the past year, how often have you needed a drink in the morning to get yourself going after a heavy drinking session?
During the past year, how often have you had a feeling of guilt or remorse after drinking?
During the past year, have you been unable to remember what happened the night before because you had been drinking?
Have you or someone else been injured as a result of your drinking?
No
Has a relative or friend, doctor or other health worker been concerned about your drinking or suggested you cut down?
Yes, but not in the past year
Yes, during the past year
Mechanisms of Injury
Penetrating Trauma
Phases of Trauma Care
Prehospital Resuscitation
Emergency Department Resuscitation
Primary Survey
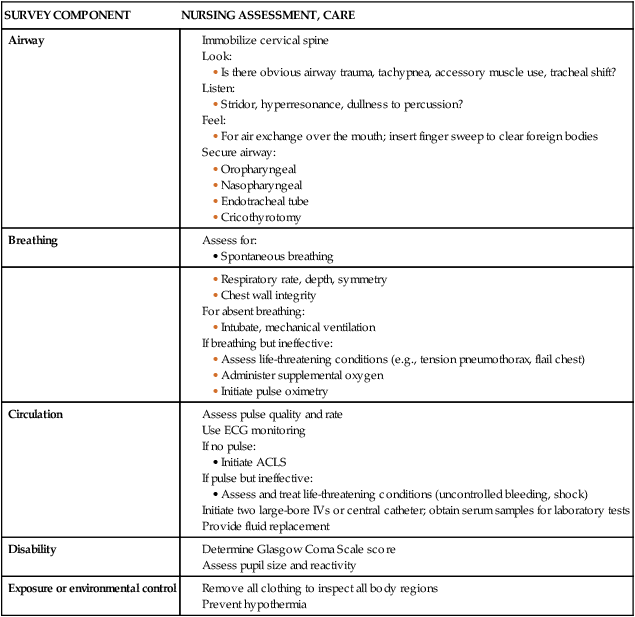
SURVEY COMPONENT
NURSING ASSESSMENT, CARE
Airway
Breathing
Circulation
Disability
Exposure or environmental control
Airway.
Breathing.
Circulation.
Resuscitation Phase
Damage Control Resuscitation.
Massive Transfusion Protocols.
Secondary Survey
Critical Care Phase
ASPECT OF INJURY OR RESUSCITATION
IMPACT ON CRITICAL CARE MANAGEMENT
Prolonged extrication time
Gives an indication of length of time patient may have been hypotensive and/or hypothermic before medical care
Period of respiratory or cardiac arrest
Effects of loss of perfusion to brain (anoxic injury), kidneys, and other vital organs
Time on backboard
Potentiates risk of sacral or occipital breakdown
Number of units of blood; whether any were not fully cross-matched; packed cells versus whole blood used
Potentiates risk of ARDS, MODS
FACTOR
IMPAIRMENT
Impaired ventilation
Injury to airway structures, loss of central nervous system regulation of breathing, impaired level of consciousness
Impaired pulmonary gas diffusion
Pneumothorax, hemothorax, aspiration of gastric contents
Shifts to the left of the oxyhemoglobin dissociation curve (can result from infusion of large volumes of banked blood, hypocarbia or alkalosis, or hypothermia)
Decreased oxygen supply
Reduced hemoglobin (from hemorrhage)
Reduced cardiac output (cardiovascular injury, decreased preload)
Increased oxygen supply
Increased metabolic demands (associated with the stress response to injury)
Specific Trauma Injuries
Traumatic Brain Injuries
Mechanism of Injury
Pathophysiology
Secondary Injury.
Brain Edema.
Classification of Brain Injuries
Concussion.
Contusion.
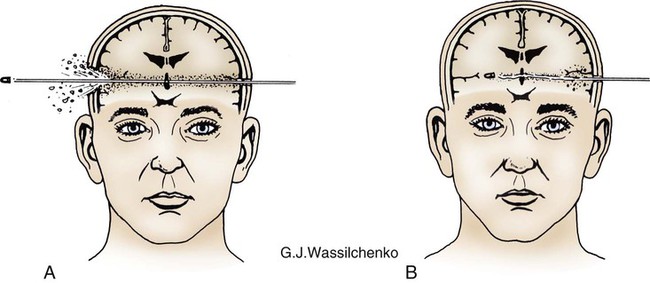
Cerebral Hematomas.
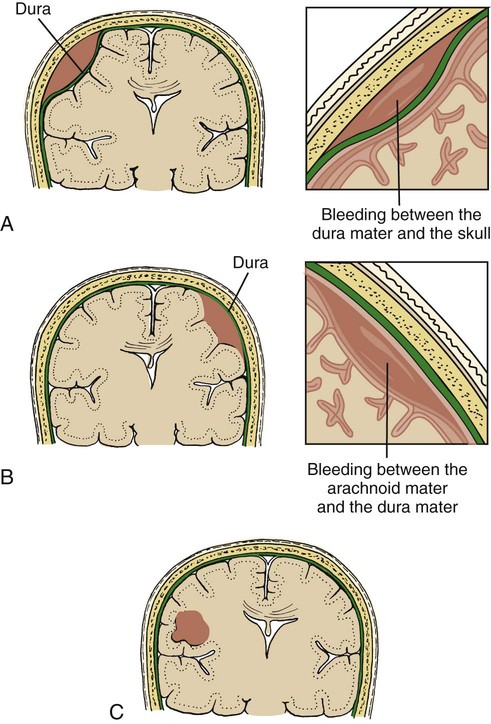
Epidural Hematoma.
Subdural Hematoma.
Intracerebral Hematoma.
Missile Injuries.
Neurologic Assessment of Traumatic Brain Injury
Nursing Assessment of the Patient with Traumatic Brain Injury.
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Trauma
Get Clinical Tree app for offline access