Chapter 39 In 2010, the maternal mortality rate of the United States was 21 deaths per 100,000 live births, compared with 12 per 100,000 in 1990. This represents a total of about 840 women who died from maternal causes in 2010.1 Some of this increase observed over the past decades may have been caused by changes in the coding and classification of maternal deaths. An alarming trend in maternal mortality in the United States is race-specific pregnancy-related mortality rates. Analysis of rates has demonstrated that compared with white women, African American women have 2.7 times the risk of dying as a result of childbirth and that the maternal mortality rate among Hispanic women was about 0.8% less than that of white women.2 At the global level, the World Health Organization (WHO), The United Nations Children’s Fund, the United Nations Population Fund and the World Bank assessed trends in maternal mortality rate from the period 1990-2010 and revealed that the number of women dying of pregnancy- and childbirth-related complications has almost halved in 20 years. The WHO’s fifth Millennium Development Goal (MDG) aims to improve maternal health with a target of reducing maternal death by 75% in the period 1990-2015. The resulting findings were that the percentage reductions for 10 countries have already achieved the MDG by 2010, 11 countries are “on track” of achieving this goal, 50 countries are “making progress,” 14 countries have made “insufficient progress,” and 11 countries are characterized as having made “no progress” and are likely to miss the MDG target unless accelerated interventions are put in place.1 Factors that influence embryonic and fetal development may be intrinsic or extrinsic in nature. Intrinsic factors such as chromosomal abnormalities and congenital anomalies account for 25% of all birth defects. Extrinsic factors, also known as teratogens, account for those remaining.3 A teratogen is any chemical, substance, or exposure that may cause any form of birth defects in a developing fetus. This would include medicines, radiation, and agents of infection. It is important to remember that the effects of teratogens depend on maternal and fetal genotypes, the stage of development when exposure occurs, and the dose and duration of the exposure of the agent.3 Exposure to ionizing radiation is usually not a concern until more than a cumulative 100 to 200 milligrays (mGy; 10 to 20 rads) have been exceeded, but some experts recommend caution in the first 25 weeks because of fetal organogenesis and central nervous system development.4–5 At doses less than 0.05 gray (Gy), no evidence of an increased risk of fetal anomalies, intellectual disability, growth restriction, or pregnancy loss from ionizing radiation is present. A small increased risk of childhood cancer, 1 in 2000 versus the 1 in 3000 background rate, may exist.4 Medication use in critically ill obstetric patients requires analysis of the risk–benefit ratio. It is important to consider the influence that drug exposure could have on the developing fetus, but often, the benefit may outweigh the potential fetal risk when all factors are considered.6 Also, fetal and newborn outcomes may be dependent on timely maternal stabilization on potential harmful medication. Critical ill obstetric patients may be reluctant to take medication during pregnancy because of the possibility or perceived possibility of the adverse effects on the fetus. The U.S. Food and Drug Administration (FDA) placed medications into risk categories regarding use during pregnancy along with potential effects on the growing fetus. Box 39-1 describes the FDA labeling with regard to a drug’s risk to a fetus.6–7 (As of February 2011, the FDA proposed major revisions to prescription drug labeling to more completely inform the use of medicines during pregnancy and breastfeeding. They are presently in the writing and clearance process.8) During the 1970s, group B Streptococcus (GBS) was recognized as the leading infectious cause of neonatal sepsis and mortality and remains so today.9 The prevalence of neonatal infection is approximately 0.4 per 1000 live births, and approximately 1300 cases of neonatal streptococcal septicemia occur each year in the United States.10 Mortality is higher among preterm infants, with case fatality rates of approximately 20% and as high as 30% among those less than 33 weeks’ gestation, compared with 2% to 3% among full-term infants.11 Maternal colonization may be intermittent, transient, or chronic, and it is likely that nearly every woman is colonized by GBS at some time. Most women are asymptomatic, but in symptomatic women, GBS is responsible for considerable maternal morbidity from infections such as pyelonephritis, chorioamnionitis, postpartum endometrioses, sepsis, wound infections and, in rare instances, meninigitis.9 In the infant, GBS may result in unexpected intrapartum stillbirth.12 Early-onset neonatal infection results almost exclusively from vertical transmission from a colonized mother to her infant and is often characterized by signs of serious illness, including respiratory distress, apnea, and shock. Late-onset disease occurs 1 week or more after birth. These infants often develop meningitis. Long-term neurologic complications are common in survivors of both types of GBS.9 At the present time, the standard for diagnosis of GBS infection is bacteriologic culture using the Todd-Hewitt broth or selective blood agar. The specimen for culture should be obtained from the lower vagina, perineum, and anus using a simple cotton swab.13 Prevention of early onset neonatal GBS infection is based on the guidelines from the Centers for Disease Control and Prevention (CDC), which were published in 1996 and updated in 2010.14 Cytomegalovirus (CMV) is a deoxyribonucleic acid (DNA) virus belonging to the herpes simplex virus (HSV) group. It causes both congenital and acquired disorders. The significance of this virus in pregnancy is related to its ability to be transmitted by asymptomatic women across the placenta to the fetus or by the cervical route during birth.15 Although the virus is usually innocuous in adults and children, it may be fatal to the fetus. The virus is passed between humans by close contact such as during kissing, breastfeeding, and sexual intercourse.15 The diagnosis of CMV infection can be confirmed by isolation of the virus in tissue culture, with the highest concentrations of virus typically in the urine, seminal fluid, saliva, and breast milk. About 50% to 80% of adult women in the United States have serologic evidence of past CMV infection. The overall risk of congenital infection is greatest when maternal infection occurs in the third trimester, but the probability of severe fetal injury is highest when maternal infection develops in the first trimester.16 Although 85% to 90% of infected fetuses will be asymptomatic at birth, the remaining 10% to 15% will have abnormalities of varying severity.16 Mortality rate among the symptomatic infants is 20% to 30%, and 90% of the these survivors have significant neurologic complications. The most common severe neonatal infections are hepatosplenomegaly, intracranial calcification, growth restriction, microcephaly, chorioretinitis, hearing loss, thrombocytopenia, hyperbilirubinemia, and intellectual disability. The virus may be identified in amniotic fluid by culture or polymerase chain reaction (PCR); however, mere identification of the virus does not necessarily delineate the severity of the fetal injury.17 Ultrasonographic findings may include fetal hydrops, growth restriction, hydramnios, cardiomegaly, and fetal ascites. Currently, no effective therapies are available to manage this infection. A recent placebo-controlled, randomized, double-blind trail evaluated a CMV vaccine and showed that it has the potential to decrease cases of maternal and congenital CMV infection.18 Ideally, preventive measures should be employed to ensure that women do not contract CMV infection during pregnancy. One simple measure is encouraging women to use careful hand-washing techniques. Toxoplasmosis is caused by the protozoan Toxoplasma gondii (T. gondii), with both farm animals, especially cattle, pigs, and sheep, and domestic cats playing an important role in the life cycle of the Toxoplasma organism. Cats are the usual host for the protozoan, and the infective oocytes are passed in feces and subsequently ingested by grazing farm animals. The organism disseminates throughout the animal’s body, ultimately forming cysts in brain tissue and muscle. Cats acquire the organism through ingestion of undercooked or uncooked meat, possibly infected rodents. Human infection occurs when infected, undercooked meat is ingested or when food has been contaminated by cat feces.19 In the United States, congenital toxoplasmosis is found to occur in 0.8 per 10,000 live births annually, and in Europe, it is estimated to be 10 cases per 10,000 live births.12 It is innocuous in most adults, resembling a minor viral illness, but it affects the fetus profoundly, creating long-term sequelae if the mother contracted the disease shortly before or during pregnancy.19 Maternal infection occurring in the first trimester typically results in more severe fetal damage and often ends in spontaneous abortion. The fetal infection occurring during the last month of pregnancy results in infants being born without any clinical signs of infections, although 50% will become symptomatic if left untreated.12,16 Severe neonatal disorders associated with congenital infection include convulsions, coma, microcephaly, and hydrocephalus, causing many infant to die soon after birth. Survivors often have visual, hearing, and intellectual impairment.20 The goal is to identify the woman at risk and to treat the disease promptly if diagnosed. Diagnosis is made by serologic testing, including the immunoglobulin M (IgM) and IgG fluorescent antibody test.20 PCR for T. gondii DNA in amniotic fluid is the best way to diagnose fetal infection, with ultrasonography revealing findings such as ascites, ventriculomegaly, microcephaly, and growth restriction.20 If fetal infection is suspected, pyrimethamine, sulfadiazine, or folinic acid should be given to the mother after the 18th week of pregnancy.12 Rubella is one of the most teratogenic of all viruses. Although it presents as a mild illness in most children and adults, rubella infection in the fetus can have overwhelming consequences. Estimates suggest that in the United States up to 10% of women are susceptible to rubella.12 The period of greatest risk for the teratogenic effects of rubella on the fetus is during the first trimester, when maternal infection results in up to 80% of maternal–fetal transmission.12 Defects are rare when infection develops after 20 weeks’ gestation.21 Infants born with congenital rubella syndrome are infectious and should be isolated at birth. Prognosis of these infants is poor, with 10% to 20% of the affected infants dying during the first year of life. Rubella syndrome manifests in the newborn most commonly as congenital cataract, sensorineural deafness, and congenial heart defects. Mental retardation and cerebral palsy may become evident in infancy. Diagnosis is made when these conditions and an elevated rubella IgM antibody titer are present at birth.21 The best therapy for rubella is prevention. Vaccination with live, attenuated vaccines should be given to all women of childbearing age who are susceptible and prior to pregnancy. Although no fetal infection has resulted from immunization of a pregnant woman, pregnancy should be avoided for 1 month after immunization. Testing for immunity involves the serology test of hemagglutination inhibition (HAI); the presence of a 1 : 18 titer or greater is evidence of immunity, and less than 1 : 8 indicates susceptibility to rubella. If pregnant when diagnosed as being “nonimmune,” the immunization should be administered in the postpartum period.22 HSV is a DNA virus with two principle strains: 1) HSV-I and 2) HSV-II. HSV is estimated to infect 1 in 6 people between the ages of 14 and 49 years (16.2%) in the United States.23 The incidence of neonatal herpes infection is 1 per 3500 live births.22 A primary herpes simplex infection may increase the risk of spontaneous abortion when infection occurs in the first trimester, whereas preterm labor, intrauterine growth restriction (IUGR), and neonatal infection are greater risks if the infection occurs late in the second trimester or early in the third trimester. If a primary lesion develops close to the time of labor, the risk of transmission is 30% to 60% for a vaginal birth.21 Exposure of the newborn to a recurrent lesion drops the risk of transmission to between 2% and 5%.24 The most likely mechanism of infection is exposure of the neonate to the viruses in the lower genital tract during the process of vaginal delivery; therefore, in a woman with either a primary or secondary outbreak of genital herpes during labor, the preferred method of delivery is by cesarean section. An estimated number of 1500 to 2000 newborns contact herpes each year, with 85% resulting from viral transmission near the time of birth from asymptomatic women.21 Neonatal HSV infection may take the form of disseminated mucocutaneous eruption, central nervous system (CNS) infection, or disseminated visceral infection. Approximately 30% of infants with disseminated disease die despite antiviral therapy, and 40% of the survivors have severe neurologic damage. Many times, the infected infant is asymptomatic at birth, with symptoms occurring any time after birth and up to 4 weeks of age.16 These symptoms include jaundice, fever, seizures, vesicular skin lesion, and poor feeding. CNS symptoms generally occur during the second or third week. All infants who have neonatal herpes should be evaluated and treated with acyclovir.25 Any woman who is planning a pregnancy and who might have been exposed to the herpesvirus should have type specific serology testing to determine her risk of acquiring HSV. If she has HSV and has experienced recurrent outbreaks during pregnancy, antiviral therapy (with acyclovir, famciclovir, and valacyclovir) is recommended after 36 weeks’ gestation to reduce the need for a cesarean section.24 Currently, no evidence of any adverse fetal effects exists in relation to exposure to these antiviral drugs used in HSV treatment during any trimester.26 Parvovirus is caused by the DNA organism, the B19 parvovirus. It causes erythema infectiosum, or “fifth disease,” in children and a mild disease in adults that produces a characteristic “slapped cheek” rash but a potential extremely serious fetal outcome. Although the risk of fetal morbidity is low, fetal infection is associated with spontaneous abortion, fetal hydrops, and stillbirth. Severe effects occur most frequently with maternal infection before 20 weeks’ gestation.27 The major concern for the fetus is nonimmune hydrops and fetal anemia, which, if left untreated, may result in death. If hydrops and fetal anemia are diagnosed, intrauterine fetal transfusion may reduce the mortality from about 50% to 18%.27 Fetal death may occur at 4 to 12 weeks after infection; therefore, fetal surveillance should be maintained from 8 to 12 weeks. In fetuses who survive the infection, long-term development appears to be normal. Nonimmune women with school-age children are more likely to acquire parvovirus, and serologic evaluation should be performed if the pregnant woman has been exposed to a child diagnosed with fifth disease.28 The condition or illness in a critically ill mother may justify an early termination of pregnancy to prevent serious complications in or death of the patient. Depending on the duration of gestation, a severely premature infant may result. Technologic advances, improvements in maternal–fetal diagnostics, and aggressive neonatal interventions have improved the survival of extremely low-birth-weight infants. Research has placed minimal viability parameters between 23 and 24 weeks’ gestation and fetal weight between 500 and 1000 g (0.5 and 1 kg).22 Critical care clinicians may encounter situations in which extrauterine viability, fetal outcomes, and maternal stability are uncertain. Clinical decisions must be made in light of the maternal–fetal risk–benefit ratio. Personal, cultural, spiritual, and social beliefs regarding viability may affect the clinical decision-making process. Parental and family beliefs and desires may conflict with those of the health care team. When confronting the dilemma of viability, the parameters of gestational age, fetal weight, parental desires, and maternal–fetal mortality must be considered. During pregnancy, the woman’s body undergoes profound physiologic changes. These changes are necessary to maintain the pregnancy and to allow for fetal growth and development. The changes are so dramatic that they would probably be considered pathologic in the nonpregnant woman. Adaptations occur in nearly every organ system, beginning during the first week of gestation and continuing until up to 6 weeks after delivery. The only system in which no documented characteristic changes occur is the nervous system. Boxes 39-2 and 39-3 and Tables 39-1 to 39-4 summarize the various systems and alterations during pregnancy.12,22,23,29 Understanding the physiologic adaptations is important to the management of the critically ill pregnant woman. More detailed information can be found in textbooks dedicated to obstetric issues. TABLE 39-1 POSITIONAL CARDIAC OUTPUT CHANGES IN PREGNANCY TABLE 39-2 HEMODYNAMIC CHANGES ASSOCIATED WITH TERM PREGNANCY TABLE 39-3 PHYSIOLOGIC ADAPTATION OF THE GASTROINTESTINAL SYSTEM DURING PREGNANCY TABLE 39-4 RENAL PHYSIOLOGIC CHANGES IN PREGNANCY Labor and delivery bring additional stresses to the maternal system, especially as a result of the pain and anxiety associated with labor. The most dramatic requirements are for the cardiopulmonary system. During labor, uterine contractions produce cyclic auto-transfusions of approximately 300 to 500 milliliters (mL). Immediately after birth, cardiac output peaks, with an 80% increase over prelabor values.12 This occurs because of the contracted uterus shunting its blood, sudden removal of fetal supply demands, and resolution of vena cava compression. Table 39-5 summarizes the cardiac output changes in labor and delivery.12,23,29 TABLE 39-5 CARDIAC OUTPUT CHANGES IN LABOR AND DELIVERY Normal blood loss from a vaginal birth is typically 500 mL; blood loss from cesarean deliveries usually is 1000 mL. Clinical estimates tend to underestimate actual blood loss by up to 50%.30 The cardiopulmonary changes occurring during labor and delivery are of significant concern because they occur over a short period and because maternal decompensation may occur. Cardiac disease ranks fourth after pregnancy-induced hypertension, hemorrhage, and infection as a cause of maternal mortality.31 Several factors have to be taken into consideration in the care of the pregnant woman with cardiac disease. Cardiac disease during pregnancy may be a result of pre-existing conditions such as congenital diseases, or it may be a result of primary cardiac disease arising prior to or during pregnancy. The woman with heart disease has decreased cardiac reserve, making it more difficulty for her heart to accommodate the higher workload of pregnancy. Thus, prepregnancy counseling is highly recommended for women with known cardiac disease. Counseling would include determining the New York Heart Association (NYHA) functional class, which is especially useful for pregnant women with structural heart disease, and determining the maternal and fetal risks associated with the pregnancy (Box 39-4).29,32 Major fetal risks include fetal development of congenital heart disease, prematurity, IUGR, and intrauterine fetal demise (IUFD). The method and timing of delivery are decided primarily by obstetric considerations, taking into account the woman’s ability to tolerate the labor process and associated physiologic changes. Selection of anesthesia techniques involves weighing the risks and benefits of the procedures. As a general rule, most patients tolerate epidural anesthesia more favorably than they tolerate general anesthesia (see Chapter 42). Congenital heart defects have become a more common finding in pregnant women, as improved surgical techniques have enabled females born with heart defects to live to childbearing age. When surgical repair can be accomplished with no remaining evidence of organic heart disease, pregnancy may be undertaken with confidence. When congenital heart disease is associated with cyanosis, whether the defect was originally uncorrected or the correction failed to relieve the cyanosis, the woman should be counseled about risks to both herself and to her fetus. Of equal concern in pregnant women with congenital heart disease is the risk of fetal congenital cardiac anomalies, which is approximately 5%. During the antepartum period, serial ultrasonography should be performed to access the fetus for appropriate interval growth.33 Atrial septal defect (ASD) is the most common congenital anomaly seen during pregnancy, and most women with ASD tolerate pregnancy, labor, and delivery without complications. The decrease in systemic vascular resistance (SVR) lessens the degree of left-to-right shunt, whereas the hypervolemic state may slightly worsen the shunt and increase right ventricular workload. The most common complications seen with ASD are dysrhythmias, heart failure, and thromboembolism.34 The outcome for the pregnant woman with ventricular septal defect (VSD) and resultant left-to-right shunt depends on the size of the defect, with larger defects producing a less favorable prognosis. The majority of VSDs are diagnosed and repaired before the women reaches childbearing age. In the absence of significant symptoms and pulmonary hypertension, pregnancy is typically well tolerated. Therapy is aimed at early recognition and treatments of signs of heart failure.35 Common complications include tachycardia, heart failure, and pulmonary hypertension. During pregnancy patent ductus arteriosus (PDA) is an unusual finding, as it is generally detected and closed during the newborn period. Patients who present with a PDA during pregnancy, usually tolerate the hemodynamic stress of labor and delivery without difficulty. Precautions against the risks of infective endocarditis and thromboembolism may be taken. Severe PDA may produce large left-to-right shunts, causing acute heart failure or pulmonary hypertension that is associated with significant maternal mortality.30 Tetralogy of Fallot (ToF) is the most common cyanotic heart defect in individuals who survive to adulthood.36 The four primary lesions associated with ToF include: 1) VSD, 2) overriding aorta, 3) right ventricular hypertrophy, and 4) pulmonary stenosis. Women with corrected ToF generally can tolerate pregnancy well. Although rare, if the congenital anomalies are not corrected, the maternal mortality rate and fetal complications increase significantly.36 Cardiopulmonary function must be maximized by measures that include treatment of dysrhythmias and use of prophylaxis for endocarditis. Considerations during labor and delivery include maintenance of adequate preload and blood pressure. Coarctation of the aorta may occur in isolation or, most often, in combination with valvular or septal anomalies. Patients with uncomplicated coarctation of the aorta who are relatively asymptomatic (NYHA class I or II) have demonstrated good prognosis and minimal risk of complications or death.12 Assessment of the aortic gradient may also be useful in predicting pregnancy outcome in patients with coarctation of the aorta. In general, aortic gradients across the site of coarctation that are less than 20 mm Hg are associated with good maternal and fetal outcomes.34 Intrapartum management focuses on the prevention of hypertension to avoid aortic wall stress. Careful management of fluid balance and left ventricular function must occur to prevent CHF and to promote adequate perfusion. Eisenmenger syndrome is not a single congenital defect but a complication that may be the result of other cardiac lesions that cause left-to-right shunting. This syndrome is more likely to occur with VDS or ASD because of the high pressure and high flow associated with these defects. This shunting may result in progressive pulmonary hypertension, leading to shunt reversal or bidirectional shunting.37 Regardless of the cause, the risk of sudden death because of pulmonary hypertension in pregnancy is 40%, which has remained unchanged for the past 50 years. Avoidance or termination of pregnancy is commonly recommended. If pregnancy is continued, therapeutic management is directed at avoidance of pulmonary vasoconstrictors, thromboembolism, and hypotension; maintenance of adequate preload and oxygenation; fetal surveillance; and reduction of stress at the time of delivery.37 The presence of a stenotic mitral valve is the most common rheumatic valve disease of pregnancy. The primary concern with mitral stenosis during pregnancy is the impedance to ventricular filling, which produces a relatively fixed cardiac output. Additional risks include thromboembolism, heart failure, and arrhythmias, especially atrial fibrillation. Cardiac output in the face of mitral stenosis is determined by two primary factors: 1) length of diastolic filling and 2) left ventricular preload. The length of diastolic filling may be negatively affected because of the increased pulse rate during a normal pregnancy. Discomfort or anxiety associated with labor may produce a tachycardic state, which may drastically impede ventricular filling, producing an even lower cardiac output, with resultant heart failure and pulmonary edema. As pregnancy is a hypercoagulable state, thrombi may rapidly form, and fibrillation may dislodge the thrombi and cause arterial embolism. Thus, prophylactic anticoagulation should be considered in this subset of women.38,39 Maintenance and management of left ventricular preload is the second important consideration in mitral stenosis. Patients may require high-normal or slightly elevated left ventricular filling pressures to maintain adequate flow across the stenotic mitral valve. It is especially important to assess the patient’s fluid status. Caution must be exercised when employing therapies that decrease preload, for example, diuresis or epidural anesthesia.38 Invasive hemodynamic monitoring may be indicated to carefully tailor therapy. In the immediate postpartum period, careful monitoring is essential because of the massive fluid shifts and large increases in cardiac output. Authorities recommend that optimal predelivery pulmonary artery occlusion pressures be maintained at 14 mm Hg or less to accommodate the increase in occlusion pressure of up to 16 mm Hg that may be associated in the immediate postpartum period.38 Aortic stenosis often is accompanied by other valvular disease, especially disease affecting the mitral valve. The hallmark of aortic stenosis is decreased left ventricular ejection.12 Mild aortic stenosis is usually well tolerated during pregnancy because of the natural hypervolemic state. Significant aortic stenosis can produce left ventricular hypertrophy and dilation. Thromboembolic prophylaxis is recommended. Critical to successful management is maintenance of cardiac output through prevention of hypovolemia, especially at the time of delivery. The maintenance of adequate cardiac output and oxygen transport is vital in the clinical management of pregnant women with aortic stenosis. Any factor that diminishes venous return or produces hypotension worsens the effects of aortic stenosis and significantly reduces cardiac output. Heart failure occurs in less than 10% of patients with severe aortic stenosis and arrhythmias in 3% to 25%.40 Mortality is now rare if careful management is provided. Marfan syndrome is an autosomal dominant disorder of connective tissue, in which serious cardiovascular involvement, usually dissection or rupture of the aorta, may occur. Prognosis is based on aortic root and wall involvement, with most authorities citing 40 millimeters (mm) as maximal root diameter, after which significant increases in mortality occur.39,41 Prevention of tachydysrhythmias and hypertension is recommended, along with endocarditis prophylaxis. Beta-blockade therapy may be initiated for cardiac rate control and to decrease pressure on the weakened aortic wall. Goals of management include maintenance of cardiac output to meet physiologic needs without producing undue stress on the aortic wall, use of regional anesthesia, and avoidance of Valsalva maneuver by shortening the second stage of labor. Careful blood pressure maintenance is essential. Differential diagnosis of chest and back pain is essential, along with recognition of other signs of aortic dissection. Because of its inheritance pattern there is a 50% risk that the disease will be transmitted to the infant.41 Peripartum cardiomyopathy (PPCM) is a type left ventricle dysfunction of unknown origin that occurs in the last month of pregnancy and up to 5 months after delivery in women with no previous history of heart disease. Thus, PPCM is a diagnosis of exclusion. Occurring in 1 in 3000 to 4000 live births, it is a relatively rare but serious condition. Early report suggested a mortality rate of nearly 50%, but more recent studies indicate a 0% to 5% rate in the United States.42,43 Controversy regarding exact causes of peripartum cardiomyopathy continues, as symptoms that are often attributable to viral and immune sources, chronic hypertension, mitral stenosis, obesity, and myocarditis have all been proposed.43 The definitive diagnosis of PPCM depends on the echocardiographic identification of new-onset heart failure during a limited period toward the end of pregnancy or in the months following delivery.40,43,44 Symptoms are identical to those of classic heart failure, but treatment depends on the pregnancy status of the patient. Women who present with PPCM during pregnancy require joint cardiac and obstetric care, but as soon as the baby is born and the patient is hemodynamically stable, standard therapy for heart failure may be applied.40 This would include treatment of diuretics, digoxin, beta-blockade, and afterload reduction. Angiotensin-converting enzyme (ACE) inhibitors, angiotensin receptor blockers (ARBs), and renin inhibitors are contraindicated during pregnancy because of fetotoxicity but may be used once the infant is born.40,44 Once the bleeding has been stopped after delivery, anticoagulation is commonly employed to prevent thromboembolism and the formation of left ventricular thrombus, which is associated with a worse prognosis.44 The clinical course of peripartum cardiomyopathy is quite variable, but 50% to 60% of patients show clinical recovery within the first 6 months after delivery.45 The prognosis of PPCM is positively related to the recovery of ventricular function. Medical therapy as outlined in the American College of Cardiology Foundation and American Heart Association (ACCF/AHA) guidelines should be continued when a woman does not recover function.44 Subsequent pregnancy carries a recurrence risk for PPCM of 30% to 50%.40 Although acute myocardial infarction (AMI) during pregnancy is rare, pregnancy has been shown to increase the risk of AMI three- to fourfold.46 With the continuing trend of childbearing at older ages and advances in reproductive technology enabling many older women to conceive, it may be expected that the occurrence of AMI will increase. Mortality rates from an AMI during pregnancy range from 37% to 50%, depending on the timing of the myocardial event.47 However, the majority of AMI in pregnancy is not related to atherosclerosis; about 50% of ischemic heart disease in pregnancy is linked to idiopathic dissections of the coronary artieries.40 Other reported conditions that contribute to AMI risk are eclampsia, thrombophilia, postpartum infections, and severe postpartum hemorrhage.40 Increased mortality is associated with many factors, including occurrence of the event during the third trimester, multigravidas older than 33 years, cesarean section, and delivery occurring within 2 weeks of infarction.47 Clinical diagnostics are similar to those for standard AMI detection, although diagnosis must be made with consideration of the normal physiologic cardiovascular changes. Coronary angiography with the possibility of coronary intervention is preferred to thrombolysis, as it will also diagnose coronary artery dissection.47 Treatment of AMI during pregnancy is focused on restoring myocardial blood flow and balancing myocardial oxygen supply and demand.47 Management may include percutaneous coronary intervention; nitrate therapy, beta-blockade therapy, or both; cardiac monitoring; oxygen therapy; management of pain and anxiety; and afterload reduction. Again ACE inhibitors, ARBs, and renin inhibitors are not indicated during pregnancy.40 Coronary angiograms, angioplasty, bypass surgery, and intra-aortic balloon pumps all have been successfully used during pregnancy. Special consideration is given to the maternal physiologic demands required during the labor and delivery processes. Operative delivery interventions, such as forceps or cesarean section, may be necessary (see “Myocardial Infarction” in Chapter 15). The outcome in the pregnant woman with prior myocardial damage depends on many factors. The length of time between the myocardial event and delivery is especially important.47 Increased myocardial oxygen demands during pregnancy must be considered, and therapy is usually supportive in nature. Careful attention to preload is essential to prevent burdening the heart and producing congestive failure (see “Heart Failure” in Chapter 15).
The Obstetric Patient
Risks to Fetal Development
Perinatal Infection
Group B Streptococcal Infection
Cytomegalovirus Infection
Toxoplasmosis
Rubella
Herpes Simplex Virus
Prematurity
Physiologic Alterations in Pregnancy
MATERNAL POSITION
CARDIAC OUTPUT (L/min)
Knee-chest
6.9 (± 2.1)
Right lateral
6.8 (± 1.3)
Left lateral
6.6 (± 1.4)
Sitting
6.2 (± 0.0)
Supine
6.0 (± 0.4)
Standing
5.4 (± 2.0)
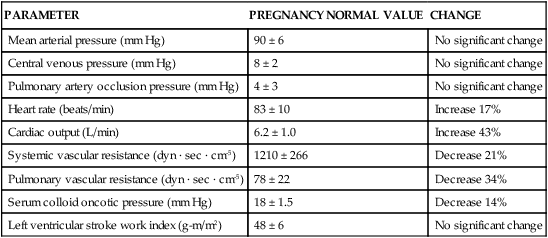
PARAMETER
PREGNANCY NORMAL VALUE
CHANGE
Mean arterial pressure (mm Hg)
90 ± 6
No significant change
Central venous pressure (mm Hg)
8 ± 2
No significant change
Pulmonary artery occlusion pressure (mm Hg)
4 ± 3
No significant change
Heart rate (beats/min)
83 ± 10
Increase 17%
Cardiac output (L/min)
6.2 ± 1.0
Increase 43%
Systemic vascular resistance (dyn · sec · cm-5)
1210 ± 266
Decrease 21%
Pulmonary vascular resistance (dyn · sec · cm-5)
78 ± 22
Decrease 34%
Serum colloid oncotic pressure (mm Hg)
18 ± 1.5
Decrease 14%
Left ventricular stroke work index (g-m/m2)
48 ± 6
No significant change
GASTROINTESTINAL FUNCTION CHANGE
PRESUMED CAUSE
Heartburn
Progesterone and estrogen; size of gravid uterus impeding gastroesophageal junction
Bleeding gums
Hyperemia
Constipation
Progesterone, causing decreased motility and intestinal secretion, enhanced water absorption
Hemorrhoids
Hyperemia, pelvic congestion, obstruction of venous return
“Morning sickness” or nausea
Increased levels of estrogen and human chorionic gonadotropin (hCG)
Risk for aspiration
Displacement of lower esophageal sphincter and reduced gastric motility
Gallstones
Decreased gallbladder activity, impaired emptying
PARAMETER
PERCENT CHANGE
NORMAL LEVELS IN PREGNANCY
Renal blood flow
Increase 25%-50%
1250-1500 mL/min
Glomerular filtration rate
Increase 50%
140-170 mL/min
Renal plasma flow
Increase 35%
700-900 mL/min
Physiologic Changes during Labor and Delivery
STAGE OF LABOR OR DELIVERY
CHANGE IN CARDIAC OUTPUT
Early first stage of labor
↑ 15% plus additional 15% with each contraction
Last first state of labor
↑ 30% plus additional 15% with each contraction
Second stage of labor
↑ 45% plus additional 15% with each contraction
First 5 minutes postpartum
↑ 80% secondary to auto-transfusion
First hour postpartum
↑ 25%
Cardiac Disorders in Pregnancy
Congenital Cardiac Disorders
Atrial Septal Defect
Ventricular Septal Defect
Patent Ductus Arteriosus
Tetralogy of Fallot
Coarctation of the Aorta
Eisenmenger Syndrome
Acquired Cardiac Disorders
Mitral Stenosis
Aortic Stenosis
Marfan Syndrome
Peripartum Cardiomyopathy
Ischemic Cardiac Disease
Acute Myocardial Infarction
Prior Myocardial Infarction
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


The Obstetric Patient
Get Clinical Tree app for offline access
