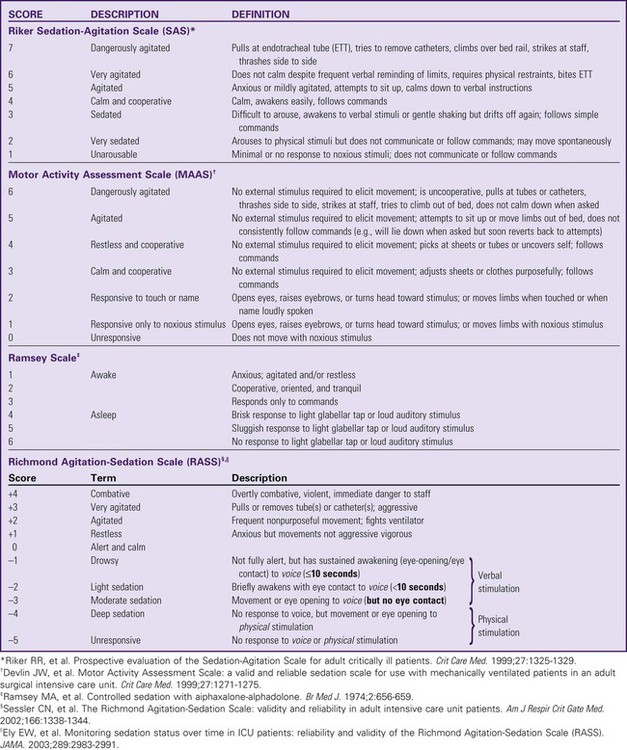
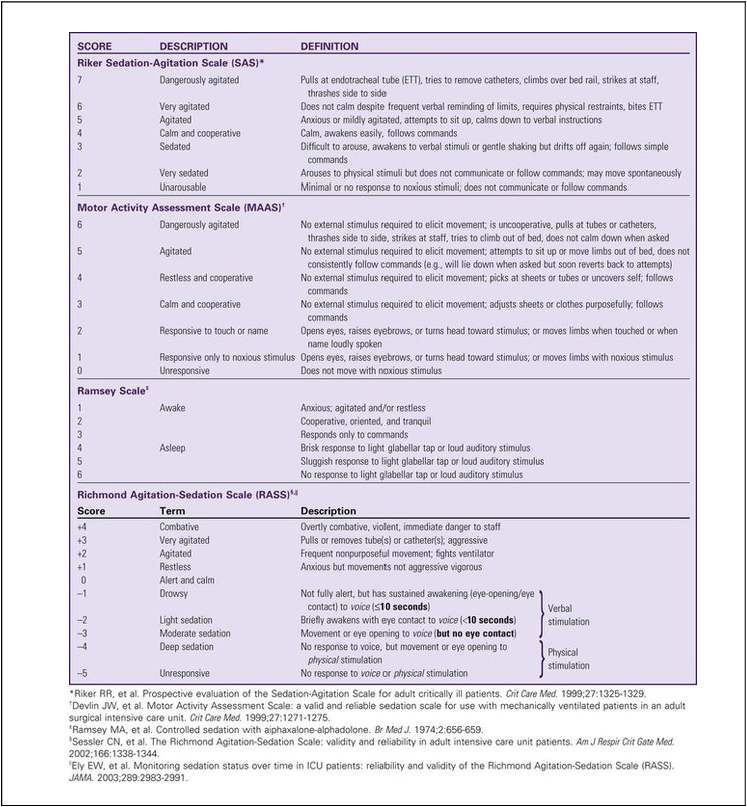
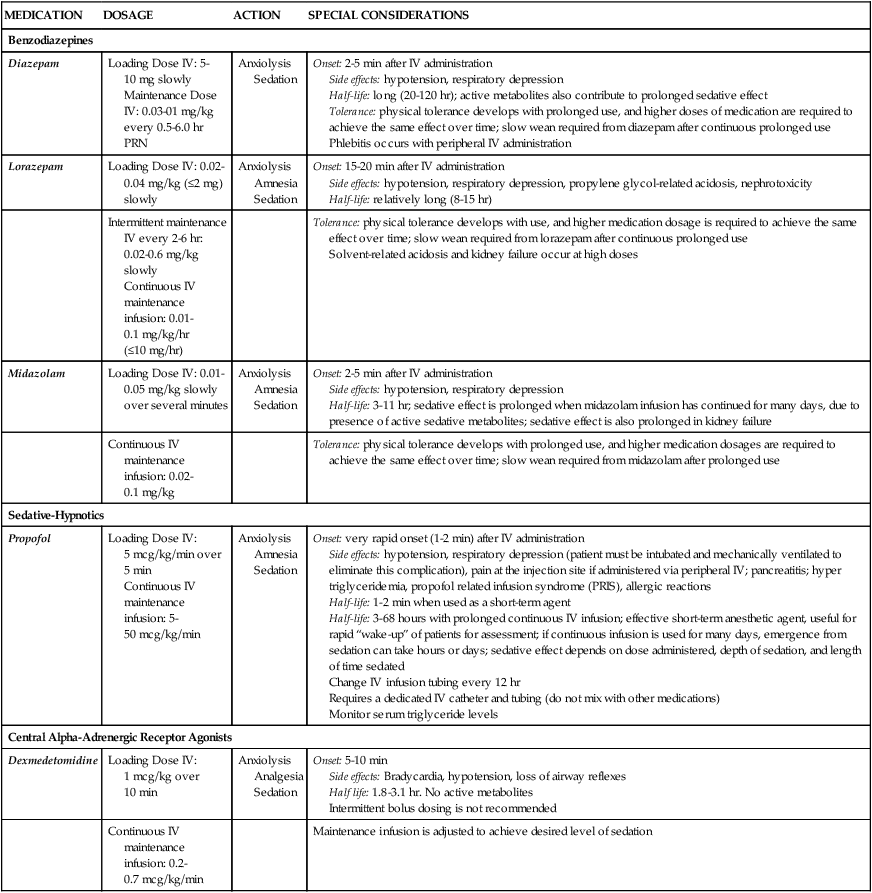
One of the challenges facing clinicians is how to provide a therapeutic healing environment for patients in the alarm-filled, emergency-focused critical care unit. Many critical care patients demonstrate agitation and discomfort caused by painful procedures, invasive tubes, sleep deprivation, fear, anxiety, and physiologic stress. Clinical practice guidelines were developed by the Society of Critical Care Medicine (SCCM) to increase awareness of these issues in the critically ill. The initial guidelines were released in 20021 and substantially revised in 2013.2 The use of scoring systems to assess and record levels of sedation and agitation is now strongly recommended.1,2 Four frequently used scales are the Ramsay Scale,3 the Riker Sedation-Agitation Scale (SAS),4 the Motor Activity Assessment Scale (MAAS),5 and the Richmond Agitation-Sedation Scale (RASS)6,7 (Table 10-1). The two scales that are recommended for assessment of agitation and sedation in the adult critically ill patient are the SAS and the RASS.2 Because individuals do not metabolize sedative medications at the same rate, the use of a standardized scale will ensure that continuous infusions of sedatives such as propofol or dexmedetomidine are titrated to a specific goal. Collaboratively, the critical care team must determine which level of sedation is most appropriate for each individual patient. The first step in assessing the agitated patient is to rule out any sensations of pain.1,2 Clinical assessment is more challenging when the patient is obtunded or has an artificial airway in place. If the patient can communicate, the verbal pain scale of 0 to 10 is very useful. If the patient is intubated and cannot vocalize, pain assessment becomes considerably more complex. The Behavioral Pain Scale (BPS) and the Critical-Care Pain Observation Scale (CPOT) are the most reliable behavioral pain scales for monitoring of pain in critically ill adults.2 These pain scales are shown in Table 9-1 (BPS) and Table 9-2 (CPOT) in Chapter 9. Pre-emptive analgesia should be provided before painful procedures.2 The next step is to determine the minimum level of sedation required1,2 (Box 10-1). The SCCM guidelines recommend that all critically ill, intubated, mechanically ventilated patients have stated goals for analgesia and sedation.2 Assessment, treatment and prevention strategies for the management of pain are listed in Figure 9-6 in Chapter 9. Several categories of sedatives are available. If the patient is experiencing pain, analgesia must be administered in addition to any sedative agents. Sedative agents include the benzodiazepines, sedative-hypnotic agents such as propofol, and the central alpha agonist dexmedetomidine (Table 10-2).1 Currently dexmedetomidine-based and propofol-based sedative regimens are recommended for routine sedation of mechanically ventilated adult patients.2 TABLE 10-2 PHARMACOLOGIC MANAGEMENT Barr J, et al. Clinical Practice Guidelines for the Management of Pain, Agitation, and Delirium in Adult Patients in the Intensive Care Unit. Crit Care Med. 2013;41(1):278. All sedatives are to be administered to a specific target level identified by a RASS or SAS level appropriate to the patient’s clinical condition (Figure 10-1).2 A target level is specified every day and reevaluated whenever there is a change in sedation dosage or patient condition. One likely sedation goal is that the patient is awake and calm, specified as a target sedation level of RASS = 0, or SAS = 4. This describes a patient who follows simple verbal commands without agitation and who can sustain eye contact for at least 10 seconds, as described in Table 10-1. When the intent is to provide deep sedation, the target sedation level is specified as RASS = -3 to -5 or SAS = 1 to 2.2 This describes a mechanically-ventilated patient who is not responsive to voice, but who may respond to physical stimulation (see Table 10-1). Pharmacologic neuromuscular blockade (NMB) induces a state of physical paralysis. NMB is administered to achieve ventilator synchrony and improve oxygenation when this cannot be achieved by sedation and analgesia alone. Table 21-10 in Chapter 21 lists NMB medications, dosages, and management. Brain function monitoring is recommended to assess level of consciousness following NMB administration (see Figure 10-1).2 In the operating room, brain function or activity is routinely monitored during anesthesia to ensure the patient does not have awareness during surgery. Similar monitoring technologies are now recommended for critically ill patients receiving NMB in addition to standard sedation assessments with RASS or SAS.2 Various technologies are available that monitor the electrical activity of the brain frontal lobes and include electromyelogram (EMG) sensors to record forehead muscle movements. Benzodiazepines have powerful amnesic properties that inhibit reception of new sensory information.1,7 Benzodiazepines do not confer analgesia. The most frequently used benzodiazepines are diazepam (Valium), midazolam (Versed), and lorazepam (Ativan). Once a mainstay of sedation, benzodiazepines are no longer recommended for sedation of the mechanically ventilated critically ill adult.2 This is because benzodiazepine-based sedative regimens are associated with worse patient outcomes, including longer duration of mechanical ventilation and delirium.8–10 Short-acting benzodiazepines (midazolam) are still helpful for the treatment of acute short-term agitation,2 because the intravenous (IV) onset of action is less than 3 minutes. However, when midazolam is administered for longer than 24 hours as a continuous infusion, the sedative effect is prolonged by its active metabolites.1 Longer acting benzodiazepines (lorazepam and diazepam) also have a role in the management of delirium tremens (described later in the chapter) and seizures,2 but these are different indications from routine sedation in critical illness. The major unwanted side effects associated with benzodiazepines are dose-related respiratory depression and hypotension. If needed, flumazenil (Romazicon) is the antidote used to reverse benzodiazepine overdose in symptomatic patients. Flumazenil should be avoided in patients with benzodiazepine dependence, because rapid withdrawal can induce seizures.11 Propofol is a powerful sedative and respiratory depressant used for sedation in mechanically ventilated patients in critical care. It is immediately identifiable by its white milky appearance, always in a glass container. At high doses (>100 to 200 mcg/kg/min), propofol is intended to produce a state of general anesthesia in the operating room.12 In the critical care unit, propofol is prescribed as a continuous infusion at lower doses (5 to 50 mcg/kg/min) to induce a state of deep sedation.12 Because propofol is lipid soluble it quickly crosses cell membranes, including the cells that comprise the blood-brain barrier. This allows rapid onset of sedation (about 30 seconds), with immediate loss of consciousness. In addition to a rapid onset of action propofol has very short half-life with initial use (2 to 4 minutes), is rapidly eliminated from the body (30 to 60 minutes), and does not have metabolites.12,13 This makes it an ideal sedative for use when a patient needs to be quickly awakened for a spontaneous awakening trial (SAT) and spontaneous breathing trial (SBT), or to assess neurologic status. This feature makes propofol an ideal medication to manage rapid ventilator weaning postsurgery. Propofol is both clinically effective and cost effective because it shortens time to extubation. It is important to add an opiate to ensure adequate pain control and amnesia. Propofol is not a reliable amnesic, and patients sedated with only propofol can have vivid recollections of their experiences. Risks of unwanted complications increase when propofol is administered long term at high doses (>4.00 mg/kg/hr for longer than 48 hours). Propofol is delivered in a fat-based emulsion, and side effects may be related to disruption of fatty acid metabolism, muscle injury, and release of toxic intracellular contents. Complications have been collectively grouped under the term Propofol Related Infusion Syndrome (PRIS) including: metabolic acidosis, muscular weakness, rhabdomyolysis, myoglobinuria, acute kidney injury, and cardiovascular dysrhythmias. PRIS affects about 1% of all patients who receive propofol; almost one third of those affected will not survive.8,13,14 Other secondary side effects related to the fat-emulsion carrier include hyperlipidemia, hypertriglyceridemia, and acute pancreatitis. Nursing vigilance is required to monitor sedation levels, and to be alert for the rare but significant risk of propofol-related complications. Serum triglycerides should be measured on all patients who receive propofol for longer than 48 hours. For additional information see Table 10-2.
Sedation, Agitation, Delirium
Assessment and Management
Sedation
Sedation and Agitation Assessment Scales
Pain Assessment Scales
Pharmacologic Management of Sedation
Sedation
MEDICATION
DOSAGE
ACTION
SPECIAL CONSIDERATIONS
Benzodiazepines
Diazepam
Loading Dose IV: 5-10 mg slowly
Maintenance Dose IV: 0.03-01 mg/kg every 0.5-6.0 hr PRN
Anxiolysis
Sedation
Onset: 2-5 min after IV administration
Side effects: hypotension, respiratory depression
Half-life: long (20-120 hr); active metabolites also contribute to prolonged sedative effect
Tolerance: physical tolerance develops with prolonged use, and higher doses of medication are required to achieve the same effect over time; slow wean required from diazepam after continuous prolonged use
Phlebitis occurs with peripheral IV administration
Lorazepam
Loading Dose IV: 0.02-0.04 mg/kg (≤2 mg) slowly
Anxiolysis
Amnesia
Sedation
Onset: 15-20 min after IV administration
Side effects: hypotension, respiratory depression, propylene glycol-related acidosis, nephrotoxicity
Half-life: relatively long (8-15 hr)
Intermittent maintenance IV every 2-6 hr: 0.02-0.6 mg/kg slowly
Continuous IV maintenance infusion: 0.01-0.1 mg/kg/hr (≤10 mg/hr)
Tolerance: physical tolerance develops with use, and higher medication dosage is required to achieve the same effect over time; slow wean required from lorazepam after continuous prolonged use
Solvent-related acidosis and kidney failure occur at high doses
Midazolam
Loading Dose IV: 0.01-0.05 mg/kg slowly over several minutes
Anxiolysis
Amnesia
Sedation
Onset: 2-5 min after IV administration
Side effects: hypotension, respiratory depression
Half-life: 3-11 hr; sedative effect is prolonged when midazolam infusion has continued for many days, due to presence of active sedative metabolites; sedative effect is also prolonged in kidney failure
Continuous IV maintenance infusion: 0.02-0.1 mg/kg
Tolerance: physical tolerance develops with prolonged use, and higher medication dosages are required to achieve the same effect over time; slow wean required from midazolam after prolonged use
Sedative-Hypnotics
Propofol
Loading Dose IV: 5 mcg/kg/min over 5 min
Continuous IV maintenance infusion: 5-50 mcg/kg/min
Anxiolysis
Amnesia
Sedation
Onset: very rapid onset (1-2 min) after IV administration
Side effects: hypotension, respiratory depression (patient must be intubated and mechanically ventilated to eliminate this complication), pain at the injection site if administered via peripheral IV; pancreatitis; hyper triglyceridemia, propofol related infusion syndrome (PRIS), allergic reactions
Half-life: 1-2 min when used as a short-term agent
Half-life: 3-68 hours with prolonged continuous IV infusion; effective short-term anesthetic agent, useful for rapid “wake-up” of patients for assessment; if continuous infusion is used for many days, emergence from sedation can take hours or days; sedative effect depends on dose administered, depth of sedation, and length of time sedated
Change IV infusion tubing every 12 hr
Requires a dedicated IV catheter and tubing (do not mix with other medications)
Monitor serum triglyceride levels
Central Alpha-Adrenergic Receptor Agonists
Dexmedetomidine
Loading Dose IV: 1 mcg/kg over 10 min
Anxiolysis
Analgesia
Sedation
Onset: 5-10 min
Side effects: Bradycardia, hypotension, loss of airway reflexes
Half life: 1.8-3.1 hr. No active metabolites
Intermittent bolus dosing is not recommended
Continuous IV maintenance infusion: 0.2-0.7 mcg/kg/min
Maintenance infusion is adjusted to achieve desired level of sedation
Targeted Sedation

CAM-ICU, Confusion Assessment Method for the Intensive Care Unit; HTN, hypertension; ICDSC, Intensive Care Delirium Screening Checklist; NMB, neuromuscular blockade; RASS, Richmond Agitation-Sedation Scale; SAS, Riker Sedation-Agitation Scale; SBT, spontaneous breathing trial. (From Barr J, Fraser GL, Puntillo KA, Ely WE, Gelinas C, Dasta JF, et al. Clinical practice guidelines for the management of pain, agitation, and delirium in adult patients in the intensive care unit. Critical Care Medicine. 2013, in press.)
Neuromuscular Blockade
Brain Function Monitoring.
Benzodiazepines
Sedative-Hypnotic Agents
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Sedation, Agitation, Delirium: Assessment and Management
Get Clinical Tree app for offline access