26 Pregnancy and Postpartum Considerations
After reading this chapter, you should be able to:
• identify the core physiological adaptations of pregnancy pertinent to critical care nursing
• describe the antenatal assessment that would be required when caring for a woman 28 weeks pregnant in ICU
• describe the priorities of management for a postpartum woman admitted to ICU with preeclampsia
• outline the main causes of obstetric haemorrhage
• outline the standard postnatal care required by a woman in ICU, for the 48 hours following birth
• consider the resources and equipment available in your workplace that are specifically required for the care of pregnant and postpartum women
Introduction
The admission of a pregnant or postpartum woman to ICU often extends ICU staff outside of their comfort zone. Pregnant and postpartum women undergo substantial physiological adaptations. Nursing staff also need to consider the fetus and be aware of, and manage, obstetric conditions. This chapter provides an overview of the epidemiology of critical illness in pregnancy, describes the physiological adaptations of pregnancy and the puerperium, outlines some key medical conditions and their interaction with pregnancy and describes the major obstetric conditions that are associated with critical illness. Additionally, we include guidance on specific practices relating to the care of pregnant and postpartum women in ICU, for example assessment of fetal wellbeing and establishment of lactation. Further details on these topics can be found in textbooks that specifically deal with critical care obstetrics.1,2 Research into critical care obstetrics is limited and at times the evidence being drawn on is dated, but still considered to be valid.
Epidemiology of Critical Illness In Pregnancy
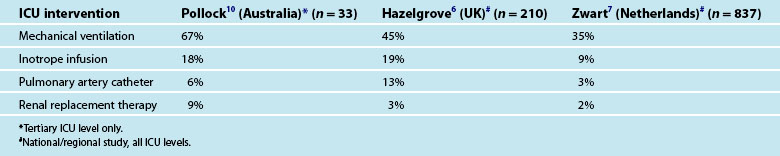
Most women experience a healthy, normal pregnancy and the development of critical illness associated with pregnancy is usually sudden and unexpected. Approximately 1 in 370 births result in a maternal ICU admission, making up about 1% of the ICU population; more than three-quarters of admissions occur following the birth of the baby.3,4 Admission of a pregnant woman to ICU is infrequent and more likely to be related to a non-obstetric diagnosis such as pneumonia or a motor vehicle crash. Conversely, in postpartum women, a condition directly associated with pregnancy is more likely, usually preeclampsia or obstetric haemorrhage.3 However, pregnant and postpartum women may be admitted to ICU with any diagnosis, which may or may not be associated with pregnancy.
Pregnant and postpartum admissions to ICU are usually short with most lengths of stay less than 24 hours. There is a vast variation in the threshold for admission to ICU with one European study of severe maternal morbidity reporting ICU admission proportions of between 0 and 50% across different regions.5 Additionally there are many women who, when admitted to ICU, do not receive any notable specific ICU intervention (Table 26.1) and the need for ICU admission for these women has been questioned.6 In general, about a third of women who experience severe maternal morbidity are admitted to ICU.7 It is feasible that admission to ICU is preventable by upskilling midwifery services6 and by early identification of severe illness resulting in prompt and appropriate treatment.6,8,9 There has been limited study of the long term outcomes for pregnant and postpartum women admitted to ICU in relation to their ongoing health and wellbeing, partner relationship and infant bonding. In developed countries like Australia, the mortality of pregnant and postpartum women admitted to ICU is relatively low at around 3% compared to the 15% mortality observed in the regular ICU population.3
Adapted Physiology of Pregnancy
Conception results in extensive physiological adaptations across most body systems (Table 26.2). The physiological adaptations most relevant to critical care nursing include cardiovascular, respiratory, renal, gastrointestinal and coagulation and the role of the placenta as the maternal–fetal interface. The uterus and breasts obviously undergo major change in pregnancy and any basic midwifery or obstetric textbook, such as Myles’ Textbook for Midwives or Midwifery: preparation for practice will describe these in detail.11,12 The physiological adaptations described in this chapter refer to a singleton pregnancy only, as women with a multiple pregnancy (i.e. twins) may undergo further changes.13 The physiological changes described refer to a non-labouring pregnant woman. Labour induces further changes to physiology, such as increased cardiac output.14
TABLE 26.2 Key physiological changes in pregnancy
| Parameter | Change during pregnancy |
|---|---|
| Cardiovascular system: | |
| Heart rate | ↑ 10–15 beats/min |
| Blood pressure | |
| Systolic | ↓ 5–9 mmHg |
| Diastolic | ↓ 6–17 mmHg |
| Cardiac output | ↑ 30–50% |
| Systemic vascular resistance | ↓ up to 35% |
| Central arterial and venous pressures | Unchanged |
| Blood and associated components: | |
| Blood volume | ↑ 40–50% |
| Plasma volume | ↑ 40–50% |
| Red blood cells | ↑ 20–40% |
| White blood cells | ↑ 100–300% |
| Platelets | Unchanged |
| Fibrinogen | ↑ 100% |
| Serum albumin level | ↓ 10–15% |
| Respiratory system | |
| Respiratory rate | Unchanged |
| Tidal volume | ↑ 25–40% |
| Minute volume | ↑ 40–50% |
| Oxygen consumption | ↑ 15–20% |
| Arterial blood gas analysis values | |
| PaO2 | 80–110 mmHg |
| PaCO2 | 28–32 mmHg |
| pH | 7.40–7.45 |
| HCO3− | 18–21 |
| SaO2 | ≥95% |
| Vital capacity | Unchanged |
| Functional reserve capacity | ↓ 17–20% |
| Airway compliance and resistance | Unchanged |
| Renal system | |
| Glomerular filtration rate | ↑ 40–50% |
| Serum urea and creatinine | ↓ Unknown |
| Urine output | <300mg/day |
| Proteinuria |
Cardiovascular System
Anatomical Changes
The heart undergoes anatomical change during pregnancy including left ventricular hypertrophy and the cross-sectional areas of the aortic, pulmonary and mitral valves increase by 12–14%. ECG changes include non-specific ST segment changes, the development of a Q wave in Lead III and a left-axis deviation pattern.15 These are evident by the end of the first trimester and remain throughout the pregnancy.16 As with the interpretation of any ECG, consider other information like the patient presentation (signs and symptoms) and blood test results to form a complete assessment of the woman’s condition.
Blood Volume
Very early in the pregnancy there is generalised vasodilatation resulting in sodium and water retention. The causes of the vasodilatation are likely to include hormonal factors (e.g. progesterone), peripheral vasodilators like nitric oxide, and potentially, an as-yet unidentified pregnancy-specific vasodilatory substance.17 The end result is a 40–50% increase in blood volume as well as reduced normal serum sodium level, from 140 to 136 mmol/L and a reduced plasma osmolality from 290 to 280 mosmol/kg. These changes persist throughout pregnancy and the osmoreceptor system resets to accept these values as normal.18
The red cell mass increases 20–40% whilst the plasma volume increases 40–50%. The resultant physiological haemodilution produces a relative anaemia which is thought to be beneficial for utero-placental perfusion. Venous haematocrit typically falls from a non-pregnant value of 40% to 34% near term.19 The increase in blood volume is evident from seven weeks’ gestation and peaks at around 30–32 weeks’ gestation, normally remaining at a stable level until delivery.17,20 Women who do not experience this normal increase in blood volume are more prone to adverse outcomes such as preeclampsia or small- for-gestational-age infant.21 The additional blood volume is also thought to accommodate the normal blood loss associated with birth (<500 mL). Pregnant women are renowned for being able to maintain stable vital signs, with blood losses as much as 1500 mL, before acutely deteriorating.
Blood Pressure
Blood pressure reduces in pregnancy, with the lowest normal blood pressure recorded during the second trimester (16–28 weeks), and returns to pre-pregnancy levels near term (see Table 26.2). Blood pressure begins dropping as early as 8 weeks’ gestation, in association with the generalised vasodilatation occurring at this time. If a woman does not experience the characteristic lowering of blood pressure, particularly during the second trimester, it is viewed with suspicion and as a potentially abnormal sign.
Heart Rate, Stroke Volume and Cardiac Output
Maternal heart rate increases by 10–15 beats per minute during pregnancy with an increase noted as early as 5 weeks’ gestation.16,22 The increase in heart rate may be a compensatory response related to the generalised vasodilatation, although a hormone-related effect cannot be ruled out.23 Tachycardia (>100 beats/min) is an abnormal sign and warrants further investigation.24 The stroke volume is noted to increase between 18 and 32%, beginning as early as 8 weeks’ gestation.25,26 An increase in cardiac output is detectable from 5 weeks gestation and continues to be 30–50% higher by 32 weeks gestation.17,26 Hence, a normal cardiac output in pregnancy may be as high as 8 L/min. The increased cardiac output is achieved by a combination of the increases in heart rate and stroke volume.
Systemic Vascular Resistance
The generalised vasodilatation observed in early pregnancy reduces systemic vascular resistance by up to 35%, with some reduction already detectable by 8 weeks’ gestation.27 The development of the low-resistance utero-placental junction was thought to act as an arteriovenous shunt and contribute to the lowered SVR seen in pregnancy. However, the very-early-observed decrease in SVR argues against this theory and perhaps circulating substances that exert a vasodilatory effect on the vasculature is a more likely proposition.
Effect of Posture on Maternal Haemodynamics
It is evident that from as early as 5–8 weeks’ gestation, pregnancy is characterised by general vasodilatation, increased blood volume, increased cardiac output and is generally a hyperdynamic state. As the pregnancy advances, the bulk of the uterus begins to have an impact on maternal haemodynamics. After 20 weeks’ gestation, a woman lying flat on her back may experience supine hypotension, secondary to compression of the inferior vena cava and aorta with subsequent reduction in venous return, cardiac output and placental flow. A reduction in placental flow may occur even without a recorded drop in blood pressure. Consequently, it is inadvisable to nurse a pregnant woman more than 20 weeks’ gestation, flat on her back. A left lateral lying position results in the best cardiac output, although manually displacing the uterus to the left whilst the woman remains supine is also effective in relieving the aorto-caval compression.28 Otherwise, the use of a wedge or pillows to maintain a left lateral tilt of at least 15 degrees is recommended to minimise aorto-caval compression.29
Postpartum Cardiovascular Changes
Heart rate returns to pre-pregnancy levels by 10 days postpartum; blood pressure has normally returned to pre-pregnancy levels by term and does not change during the puerperium.23,27 The first few days of the puerperium are associated with a diuresis which reduces the circulating volume and results in haemoconcentration of blood. Consequently a postpartum haemoglobin level will increase over the first few days and the risk of thromboembolism is higher during the postpartum period than during pregnancy. Due care should be paid to postpartum women in ICU to prevent deep vein thrombosis, particularly as many of these women are in ICU with complications of preeclampsia or severe obstetric haemorrhage, both of which further increase the likelihood of thromboembolism.30
Cardiac output increases briefly in the immediate postpartum period to compensate for blood losses and tends to increase by 50% of the pre-delivery value, at this point in the post partum phase stroke volume is increased while the maternal heart rate is often slowed.23 For most women, the immediate postpartum elevation in cardiac output only lasts for an hour or so. By 2 weeks postpartum, many haemodynamic parameters have returned to pre-pregnancy levels for the majority of women, although some have been recorded as remaining above pre-pregnancy levels at 12 months postpartum, including cardiac output.14,27 There is increasing acknowledgement that for many women following childbirth, there is a permanent modification to the cardiovascular system, although whether this persists into the menopausal era is not known and whether it impacts on cardiovascular disease risk is also unknown.27
Respiratory System
Changes to the Upper Airways and Thorax
Changes also occur to the chest wall with relaxation of ligaments resulting in an outwards flaring of the lower ribs and a 50% increase in the subcostal angle.31 Both the diameter and the circumference of the thorax increase by 2 cm and 5–7 cm respectively.31,32 These physical changes are thought to cause the diaphragm to rise by 5 cm, with this occurring early in pregnancy and well before there is any pressure from the advancing uterus.32 Respiratory muscle function does not change significantly during pregnancy and rib cage compliance is unaltered.31 The functional reserve capacity (the amount of air left in the lungs after expiration) is reduced 17–20% making the pregnant woman more vulnerable to hypoxaemia during any apnoeic period. Chest X-ray interpretation is unchanged during pregnancy, despite the variety of changes to cardiovascular and respiratory flows.23
Changes to the Physiology of Breathing
From as early as 5 weeks’ gestation, multiple factors result in an increased respiratory drive. The increase in progesterone levels is thought to lower the PaCO2 threshold in the respiratory centre to stimulate respiration resulting in hyperventilation.15 Other related factors include an oestrogen-mediated progesterone response, lower serum osmolality, strong ion difference and increased level of wakefulness that are also present in pregnancy.33–35 Increased minute ventilation begins soon after conception and peaks at 40–50% at term.15 The increase in minute ventilation is achieved by a 30–50% increase in tidal volume (e.g. an increase of 200 ± 50 mL at term), with no increase in respiratory rate.15
Due to the altered respiratory function, normal arterial blood gas values are different in pregnancy compared to the non-pregnant values (see Table 26.2). The reduced PaCO2 level creates the necessary gradient for the fetal CO2 to passively cross the placenta for maternal excretion. PaO2 normally increases by 10 mmHg, although the PaO2 level is affected by posture, particularly as the pregnancy progresses.36 In advanced pregnancy, the supine position is associated with a reduction in PaO2 of up to 10 mmHg when compared with the same woman in the sitting position.37 The kidneys compensate for the lowered PaCO2 by increasing bicarbonate excretion, which serves to maintain a normal pH.36,38,39 Normal oxygen saturation in pregnancy has not been well investigated, however, it is likely to be 97–100% at sea level, with a healthy pregnant woman’s saturation not dropping below 95% during moderate exercise.40,41
The notable hyperventilation of pregnancy is associated with a feeling of breathlessness in up to 75% of healthy pregnant women when attending to activities of daily living.33 Distinguishing what is considered ‘physiological dyspnoea’ from pathological dyspnoea, for example developing cardiomyopathy, can present a challenge in pregnancy. Dyspnoea at rest is usually an abnormal sign in pregnancy.42
Postpartum Respiratory Changes
There is complete resolution of the spirometry and arterial blood gas changes by 5 weeks postpartum.36 Unfortunately there has been no study reporting the daily transition of these parameters over the first week postpartum – the timing when a postpartum woman is likely to be in ICU. One very old study reported that CO2 levels took between two and five days to return to normal non-pregnant values postpartum.43 Regardless, with the fetus delivered, it is probable that no harm will be done to a woman by the titration of her ventilation requirements according to non-pregnant conventions and arterial blood gas values.
Renal System
All smooth muscle dilates in early pregnancy, most likely in response to progesterone. This includes the renal tract, involving the renal pelvis, calyces, ureters and urethra. The placental hormone, relaxin, has also been shown to have an effect on renal tract dilatation.44 Each kidney lengthens by about 1 cm, which is explained by the dilatation and associated mild hydronephrosis and increased vascularity of the kidneys, with no hypertrophy of renal tissue.17 Another effect of widespread dilatation is urinary stasis and an increased likelihood of urinary tract infection. Acute pyelonephritis is one of the most common renal complications of pregnancy and is associated with the onset of preterm labour.45
The kidneys receive a proportion of the additional cardiac output resulting in a 30% increase in renal blood flow. The glomerular filtration rate (GFR) increases 40–50% during the first trimester and then reduces slightly towards the end of the third trimester.18 The increase in GFR may result in the tubule active transport systems for both glucose and proteins to be exhausted, with both glycosuria and proteinuria common in pregnancy. Glycosuria is not related to blood sugar levels and is unhelpful in monitoring diabetes. Proteinuria, up to 300 mg per 24 hours, is considered normal in pregnancy. Conversely, the high GFR results in lowered serum levels of both urea and creatinine. A plasma urea level exceeding 4.5 mmol/L and plasma creatinine level higher than 75 µmol/L, should be viewed as abnormal and indicative of potential renal impairment.18,46 There is conflicting information regarding normal urine output during pregnancy, with some studies suggesting no difference to that during non-pregnancy and others reporting an increase in 24-hour urine volume after 12 weeks’ gestation.45,47
Postpartum Renal Changes
The most significant renal change is the diuresis that occurs in the 1–3 days postpartum. This diuresis serves to offload the additional blood volume that the woman has had circulating for the duration of the pregnancy. There has been little examination of ‘normal urine output’ with the standard 0.5 mL/kg/hr reported as a minimum acceptable level, however a true ‘normal’ level is likely to be closer to 0.8 mL/kg/hr.48 Creatinine levels are within the normal non-pregnancy range within 24 hours postpartum, whilst the lower urea levels remain for at least 48 hours.46 The bladder returns to the pelvis in the early postpartum period as the uterus and other organs resume their pre-pregnancy position.
Gastrointestinal System And Liver
The uterus pushes abdominal organs aside as it advances making assessment and diagnosis of an acute abdomen difficult. For example, the appendix is progressively displaced upwards and laterally from McBurney’s point at the third month, reaching the level of the iliac crest by late pregnancy.49 The bowel and other organs are generally displaced by the enlarging uterus; women with prior abdominal surgery and adhesions are predisposed to intestinal obstruction as a result.50 Additionally, there is an increase in intraabdominal pressure which may contribute to another common pregnancy symptom, heartburn.
Hepatobiliary Changes in Pregnancy
There is no significant increase in hepatic arterial blood flow during pregnancy, despite the 40–50% increased cardiac output.51 There is, however, a doubling of bloodflow to the liver supplied by the portal vein,51 which may have an impact on oral medication metabolism in the liver. There are also changes in other hepatic enzymes responsible for drug metabolism, resulting in a change in pharmacokinetics of some medications, e.g. higher plasma levels of midazolam. Serum albumin levels reduce to 30–40 g/L for the majority of pregnancy, with levels as low as 25 g/L normal during the second postpartum week.46 This low albumin level reduces colloid osmotic pressure that contributes to the dependent oedema, for example swollen ankles, that is common in pregnancy.
Haemostasis System
During pregnancy, the woman’s body prepares for the separation of the placenta, a time of potential large blood loss. The blood flow to the placental bed at term is in the range of 600–800 mL/min. Both elements of the haemostasis system are activated during pregnancy (coagulation and fibrinolysis), with pregnancy and particularly the postpartum period associated with an increased risk of thrombus formation. Thromboembolic events remain a leading cause of maternal death in developed countries.24,52 A number of changes to the haemostatic system occur during pregnancy (Table 26.3).
TABLE 26.3 Haemostatic changes during pregnancy56–58
| Haemostatic component | Changes during pregnancy |
|---|---|
| Platelets: | |
| Count Function and lifespan | UnchangedUnchanged |
| Clotting factors: | |
| Factors VII, VIII & IX Fibrinogen Other clotting factors | IncreasedDoubles by termMainly unchanged |
| Fibrinolysis: | |
| D-Dimer level | Progressively increases throughout pregnancy By term, level >0.5 mg/L common |
Of note, gestational thrombocytopenia – a platelet level between 80–150 × 109/L – occurs in 6–8 % of women.53,54 It generally has no negative impact on the woman or fetus at these levels, as there is no pathology associated with the low platelet count.55
Changes In White Blood Cells And The Immune System
There is continued debate on whether the pregnant state increases vulnerability to infection, secondary to some protective mechanism that prevents the woman’s body from reacting to the fetus as a foreign body.17 Pregnant women have increased innate immune system activity (non-specific response) and a lowered adaptive immune system (specific antibody response), with pregnant women more vulnerable to some infections like malaria and varicella.17,59,60 Pregnant women are often in contact with small children and potentially have an increased exposure to various infections. The white blood cell number increases throughout pregnancy, peaking around delivery when a normal level may be as high as 25 × 109/L.46
The Maternal–Fetal Interface
Placenta
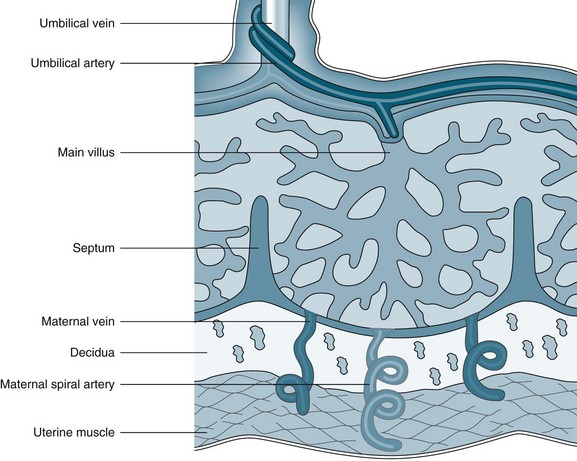
The placenta develops from the trophoblastic layer of the fertilised ovum and is completely formed and functioning ten weeks following fertilisation.61 The chorionic villi constitute the undersurface of the placenta and attach to the uterine wall via the decidua. The end result is an interface whereby maternal blood fills a space in which the nutritive villi float and are bathed in the maternal blood (Figure 26.1). A few villi are more deeply anchored in the decidua and these are referred to as anchoring villi.61 The blood drains back into the maternal circulation via maternal sinuses and the endometrial veins. Approximately 150 mL of maternal blood, replenished three to four times per minute, bathes the villi in the intervillous space.61 The chorionic villi maximise the available surface area to optimise the exchange of products across the maternal–placental interface. By term, this surface area is said to be as large as 13 m2.62 Initially, four layers of cells separate the maternal blood from the fetal blood, reducing to three after 20 weeks’ gestation; these cell layers are collectively referred to as the ‘placental membrane’ or ‘placental barrier’.63 Damage to villi, such as a threatened abortion or blunt trauma, may result in mixing of the blood circulations.
Role of the Placenta
The placenta provides six major functions to sustain the pregnancy and fetus: respiration, nutrition, storage, excretion, protection and endocrine.61 Fetal lungs are filled with fluid and all oxygenation and removal of carbon dioxide must be provided via the placenta. Fetal haemoglobin has a slightly different structure to adult haemoglobin and has a higher affininity for oxygen. Both oxygen and carbon dioxide cross the placental membrane by simple diffusion. Nutrients are actively transported across the placental membrane, with the placenta able to select the substances needed by the fetus, even at the expense of the mother if necessary.61 The placenta is able to store glucose by converting it to glycogen and reconverting it to glucose as required and is also able to store iron and some fat-soluble vitamins.
Impact of Impaired Utero–placental Gas Exchange
Effective gas exchange across the placental membrane depends on sufficient maternal blood pressure and adequate O2 and CO2 gradients for passive diffusion to occur. In response to hypoxaemia, a fetal brain-sparing mechanism goes into effect that increases fetal arterial pressure and redirects blood delivery to the main organs, namely the brain, heart and adrenal glands.64 This centralisation of fetal blood flow is more apparent in response to maternal hypoxaemia than to reduced utero–placental blood flow. It appears that a less mature fetus (i.e. earlier gestation) may be less susceptible to asphyxia than a fetus at term.64
Clinical Implications Of The Physiological Adaptations Of Pregnancy
The beginning point of any nursing practice is an understanding of normal anatomy and physiology. The normal physiological adaptations of pregnancy can be used to explain the so-called ‘minor discomforts’ of pregnancy, including constipation, varicose veins, indigestion, breathlessness and fatigue. For a critically ill pregnant woman being nursed in ICU, these normal physiological changes are also highly relevant for her care. ICU nurses need to accommodate for, and take into account, the likely impact of the normal physiology of pregnancy on common ICU monitoring, interventions and care (Table 26.4).
TABLE 26.4 Clinical relevance of physiological adaptations in pregnancy
Diseases and Conditions Unique to Pregnancy
Preeclampsia
The umbrella term ‘hypertension in pregnancy’ is used to describe a myriad of conditions in pregnancy where hypertension is a major feature. These include gestational hypertension, pre-existing essential hypertension and preeclampsia which incorporates eclampsia and Haemolysis Elevated Liver enzymes and Low Platelets (HELLP) syndrome (Table 26.5). Comprehensive descriptions of these conditions and their management have been published by the Australian and New Zealand College of Obstetricians and Gynaecologists (RANZCOG) and the Society of Obstetric Medicine Australia and New Zealand (SOMANZ).65,66
TABLE 26.5 Definitions of conditions characterised by hypertension in pregnancy
| Term | Definition |
|---|---|
| Hypertension in pregnancy | |
| Essential hypertension | |
| Gestational hypertension | |
| Preeclampsia (Also referred to as pregnancy induced hypertension (PIH), toxaemia) | |
| Eclampsia | |
| HELLP syndrome |
DIC – disseminated intravascular coagulopathy; HELLP – haemolysis, elevated liver enzymes and low platelets.
Preeclampsia is a condition unique to human pregnancy in that, whilst characterised by hypertension and proteinuria, it is a multisystem disorder consisting of variable clinical features caused by widespread vasospasm. The basis for preeclampsia remains unknown. The indication for ICU admission is usually related to organ failure, caused by the widespread vasospasm and reduced organ perfusion that characterises the disease.67 Preeclampsia can be a very serious condition and remains a leading cause of maternal death in both developed and developing countries.68
Aetiology
The placenta is strongly implicated in the cause of preeclampsia; its removal is the only definitive treatment for the condition. However, the exact mechanisms of the aetiology of the disease remain elusive and are likely to be complex and multifactorial. Theories explaining the pathophysiology of preeclampsia include immune maladaptation, abnormal trophoblast embedding, endothelial activation and excessive inflammatory response, and a genetic susceptibility (Box 26.1).71 The contribution of each component and whether all components are relevant in all cases of preeclampsia is not known. It is feasible that there are differing types of pathophysiology for mild preeclampsia that occurs at term, compared with severe preeclampsia that often occurs prior to 34 weeks’ gestation.
Box 26.1
Theories on the pathophysiology of preeclampsia71
Placentation and the immune theory of preeclampsia:
Placental debris hypothesis: syncytiotrophoblast shedding
• increased syncytiotrophoblast shedding
• placental ischaemia and reperfusion with subsequent oxidative stress
• increased circulating levels of inflammatory cytokines, corticotropin-releasing hormone, free-radical species and activin A
Endothelial activation and inflammation:
• enhanced vascular sensitivity to angiotensin II and noradrenaline with subsequent vasoconstriction and hypertension
• a fall in production and activity of vasodilator prostaglandins, especially prostacyclin and nitric oxide
Genes, the genetic-conflict hypothesis, and genetic imprinting:
Preeclampsia is associated with impaired remodelling of the uterine spiral arteries and abnormal placental implantation. It is thought that maternal–fetal immune maladaptation could be the main cause for this superficial placentation.71 Placental flow defects are detected as early as 12 weeks in some women who go on to develop preeclampsia.72 Placental ischaemia and reperfusion with subsequent oxidative stress have been regarded as major pathogenetic drivers. It is likely that there is an excessive or atypical maternal immune response to trophoblasts and the disease represents a failed interaction between the mother’s and fetus’ genetic make-up.68 The excessive systemic inflammatory response and associated endothelial dysfunction and enhanced vascular reactivity, results in widespread vasospasm which precedes the onset of clinical signs, such as hypertension.68 Other common clinical manifestations in preeclampsia include enhanced endothelial-cell permeability and platelet aggregation, explaining the increased likelihood for oedema and thrombosis.71
In summary, preeclampsia presents post 20 weeks’ gestation, but the foundation for the disease relates to abnormal placentation early in the first trimester. Whilst a number of ‘biomarkers’ attempting to predict the onset of preeclampsia have been identified, there is no reliable predictive test in clinical use.68
Risk Factors
A number of maternal characteristics are associated with an increased likelihood for the development of preeclampsia; these include:71,73
• preexisting medical conditions including diabetes, chronic hypertension, chronic renal disease, antiphospholipid antibodies
• preeclampsia in a prior pregnancy, particularly if the previous preeclampsia presented prior to 34 weeks
• family history of preeclampsia, particularly on the maternal side of the family
• multiple pregnancy e.g. twins
• body mass index >25 prior to pregnancy
• a new fathering partner for the index pregnancy
• achieving conception using assisted techniques, such as in vitro fertilisation.
Incidence
The incidence of preeclampsia is reported between 2–8%, with variations based on severity of the disease.73 The incidence of eclampsia in developed countries has reduced since the routine use of magnesium sulphate has been adopted; in the UK, the rate is about 3 cases of eclampsia for every 10,000 births.74 A prospective binational study on the incidence of eclampsia in Australia and New Zealand is underway by the Australasian Maternity Outcomes Surveillance System (AMOSS), and intends to document Australian and New Zealand population-based incidences for the first time.75 The incidence of HELLP syndrome is reported to be between 0.11% and 0.67% of all pregnancies.76,77 Preeclampsia is one of the most common indications for ICU admission at approximately one ICU admission for every 1000 deliveries.3
Clinical Presentation and Diagnosis
The clinical presentation of preeclampsia is often subtle, resulting in delayed diagnosis and treatment. Common symptoms include feeling ‘generally unwell’, headache, heartburn, nausea and vomiting, and oedema; all non-specific symptoms experienced by many pregnant women who do not have preeclampsia. Severe preeclampsia is associated with severe headache, hypereflexia, vision disturbances, severe epigastric pain, right upper quadrant pain and even blindness. There is also evidence of impaired systolic and diastolic myocardial function. Diagnosis is made when the woman has hypertension (BP ≥140/90), in association with evidence of multisystem involvement (Box 26.2). Severe preeclampsia is diagnosed when the BP is ≥160/110, in association with multisystem involvement. Additionally, eclampsia and HELLP syndrome are considered severe variants of preeclampsia even if the woman is normotensive.
Box 26.2
Diagnostic features of preeclampsia
Hypertension ≥140/90 accompanied by one or more of the following:
This clinical diagnosis has replaced the traditional triad of signs of hypertension, proteinuria and oedema, in accordance with the increased understanding of the multisystem nature of the disease. Raised blood pressure is commonly, but not always, the first sign of the condition. Although proteinuria is the most commonly recognised additional feature after hypertension, it is not mandatory to make a clinical diagnosis. Oedema is no longer a specific sign of preeclampsia, though women who develop non-dependent oedema, such as facial oedema, should be investigated for evidence of preeclampsia.66 Common investigations include urea, creatinine and electrolytes, full blood examination, liver function tests, serum uric acid, spot urine protein/creatinine ratio and 24 hour urine collection. Additional tests, such as coagulation studies, may be required as indicated by the clinical condition. Intra-uterine fetal growth restriction is a sign of placental involvement (i.e. impairment) and investigation into fetal wellbeing, including an ultrasound for fetal growth estimation and amniotic fluid volume, and umbilical artery Doppler flow patterns should be done routinely following a diagnosis of severe preeclampsia.
The presentation of preeclampsia is usually restricted to women ≥20 weeks’ gestation unless they have a co-existing condition that is known to be associated with the <20 weeks presentation of preeclampsia including hydatidiform mole, multiple pregnancy, fetal triploidy, severe maternal renal disease or antiphospholipid antibody syndrome.66
The old adage is that approximately one-third of eclampsia occurs during pregnancy, one-third during labour and one-third postpartum; the UKOSS study found 45% of first eclamptic fits were during pregnancy, 19% during labour and 36% postpartum.74 The majority of postpartum eclampsia occurs in the first 48 hours, although late-onset eclampsia may occur at two to three weeks postpartum. Despite the nomenclature, eclampsia can occur without any preceding signs and symptoms of preeclampsia. In the UKOSS eclampsia study, only 38% of women had established hypertension and proteinuria in the week preceding the eclamptic fit and 21% of women had no sign or symptom prior to the first eclamptic fit.74 HELLP syndrome commonly presents during pregnancy with about 30% postpartum.78
Management Priorities
Women with mild preeclampsia at term may be managed with induction of labour and delivery and experience few complications. The management of women with severe preeclampsia is focused on stablising the woman’s condition, optimal timing of delivery of the baby (and placenta) and preventing complications of the condition. Women with eclampsia and HELLP syndrome require the same treatments as other women with severe preeclampsia, even though they may or may not have the same degree of hypertension.69,79
Prevention of eclampsia
Magnesium sulphate has received the most attention as an anticonvulsant in preeclampsia, with its mechanism of action thought to be connected to the release of prostacyclin from the endothelium, reversing the vasoconstriction that is the basis of the disease.80,81 Magnesium is the anticonvulsant of choice to reduce the incidence of eclampsia.82,83 A common magnesium regimen is:68,82
• 4g IV loading dose given over 15–20 minutes
• an ongoing infusion of 1 g/hr
• an additional 2–4 g IV loading dose should be administered over 10 minutes to treat a recurrent eclamptic seizure
• continue infusion until 24 hours following delivery or 24 hours following the last eclamptic fit; whichever occurs the later.
The optimal therapeutic level of magnesium required to reduce the risk of fitting is not well understood and many advocate against the need to monitor serum magnesium levels on the basis that clinical assessment of deep tendon reflexes, urine output and respiratory rate is adequate to identify potentially toxic magnesium levels,68,82 although evidence is inconsistent. Other opinions suggests a therapeutic serum magnesium level of 2 mmol/L but there is no rationale provided for this level.84
Control hypertension
Obtaining control of high blood pressure remains a priority not only to improve organ perfusion but to minimise the risk of cerebral haemorrhage, a well-demonstrated hazard of hypertension in preeclampsia.24 Both systolic and diastolic pressures are important and care should be taken to ensure a controlled lowering of blood pressure, as a rapid drop can compromise fetal wellbeing. There is no evidence for the superiority of any specific antihypertensive, although there is some evidence that diazoxide may result in a potentially-harmful rapid drop in the woman’s blood pressure, and that ketanserin may not be as effective as hydralazine.83 Intravenous hydralazine is the most common drug used to treat very high blood pressure with IV labetalol increasingly being used. Severe hypertension may be treated with IV GTN or nitroprusside. The target blood pressure is not well described, other than to avoid precipitous drops in BP and to maintain adequate placental perfusion. Research has used a target diastolic BP of 85–95 mmHg.85
Optimal fluid management
Despite being hypertensive, preeclamptic women are usually plasma-volume depleted.86 In the past, intravenous fluid was administered in an attempt to restore the deficit, with no advantage noted between colloids and crystalloids. More recently, there has been a move towards more conservative plasma volume expansion due to the risk of pulmonary oedema. In reviews of maternal deaths associated with preeclampsia, it was noticed that some women were dying from complications of fluid overload. Careful titration of intravenous fluid is required with the use of pulmonary artery catheters advocated by some to guide the administration of fluid in women with severe preeclampsia, to optimise plasma volume and organ perfusion without the development of pulmonary oedema.87 Central venous pressure is universally accepted as unhelpful to guide fluid management in preeclampsia. See also Box 26.3.
Box 26.3
Management of women with HELLP syndrome using steroids
The use of steroids has been evaluated in the management of HELLP syndrome in the belief that steroids may mitigate the severity of the disease. However, a Cochrane Review concluded that there was insufficient evidence to determine whether steroid use as a treatment for HELLP syndrome had a favourable outcome for mothers and babies, although steroids may be beneficial if an increase in platelet count was imperative.88
Thrombophylaxis
Preeclampsia is an independent risk factor for thromboembolic disease and when combined with prolonged bed rest, as may occur with caesarean section, ICU admission, obesity and age ≥35 years, due consideration must be made on the need for thrombophylaxis (in the absence of any contraindications). Thus women with severe preeclampsia admitted to ICU may meet the requirements for treatment with compression stockings and low molecular weight heparin for a minimum of 7 days.30
Betamethasone
Women in late pregnancy with severe preeclampsia diagnosed prior to 34 weeks’ gestation are normally prescribed a single dose of betamethasone (11.4 mg IM), to promote fetal lung maturity and surfactant production. A Cochrane Review has shown that treatment with antenatal corticosteroids reduces the risk of neonatal death, respiratory distress syndrome, cerebroventricular haemorrhage, necrotising enterocolitis, infectious morbidity, need for respiratory support and neonatal intensive care unit admission, with no adverse effects on the mother.89
Optimal timing of delivery
Women with severe preeclampsia can only be definitively cured by delivery, no matter what the gestation. A number of studies have trialled ‘temporising treatments’, aimed at prolonging the pregnancy especially when a woman develops early onset severe preeclampsia (<34 weeks’ gestation). Whilst some have found that treatment with vasodilators and fluid administration prolongs pregnancy with no adverse effect, the general belief is that prolonging the pregnancy is associated with an increased chance of the maternal complications of preeclampsia, such as eclampsia, pulmonary oedema and cerebral haemorrhage.68,90 Consequently, a woman with severe preeclampsia is usually stabilised (magnesium sulphate commenced and hypertension controlled) and arrangements for delivery are made. Ideally, women <34 weeks’ gestation should be transferred to a tertiary obstetric centre prior to delivery.
Obstetric Haemorrhage
Obstetric haemorrhage is a leading cause of maternal mortality across the world and directly accounts for an estimated 127,000 deaths each year. Postpartum haemorrhage (PPH) is responsible for the majority of these maternal deaths. The past decade has seen an increase in both the incidence and severity of obstetric haemorrhage, with more women requiring a blood transfusion for postpartum haemorrhage than in the past.91 Severe bleeding in childbirth is estimated to occur once in every 200–250 births, although incidence is highly dependent on how ‘severe bleeding’ is defined.24 Major obstetric haemorrhage is often sudden and unexpected, and is frequently associated with an acute coagulopathy. Early recognition and treatment of major obstetric haemorrhage is vital to ensure the best outcome for mother and fetus. A repeated finding in maternal death reviews is a delay by obstetric providers in recognising the severity of haemorrhage and a consequent deterioration in maternal condition.24
Obstetric haemorrhage may occur after the 20th week gestation up to the birth (antepartum haemorrhage) and after the birth of the baby (postpartum haemorrhage). Severe obstetric haemorrhage is a common reason for postpartum women to be admitted to ICU at 0.7/1000 deliveries, with many women experiencing haemorrhage before and after the birth of the baby.3 Although not classified technically as an obstetric haemorrhage, ruptured ectopic pregnancy can also result in life-threatening haemorrhage and result in ICU admission. The common causes of antepartum and postpartum haemorrhage are described below with common management strategies presented at the end of the section. See also Box 26.4.
Antepartum Haemorrhage
Antepartum haemorrhage (APH) is defined as any bleeding from the genital tract occurring between the 20th week of gestation and the birth of the baby and occurs in 2–5% of all pregnancies.92 Bleeding from the vagina prior to 20 weeks’ gestation is referred to in terms of miscarriage (e.g. threatened) and is not classified as an APH. The two main causes of APH are placental abruption and placenta praevia.
Placental abruption (or abruptio placentae)
Placental abruption is premature separation (i.e. before the birth of the baby) of a normally-sited placenta from the uterine wall and is responsible for about 25% of APH.92 Only a portion of the placenta separates with two-thirds separation considered severe. There are two relevant matters to consider with placental abruption: how much blood the woman has lost and how much placenta remains attached and functionally able to support the fetus. If the placenta partially separates along an edge of the placenta, blood loss is usually visible via the vagina. In some cases the centre part of the placenta detaches, leaving the rim attached all the way around (like the rim of a dinner plate) and in these cases, the blood loss is usually not visible via the vagina (i.e. is concealed). However, the woman may have lost substantial blood volume and be in hypovolaemic shock. This type of placental abruption is usually accompanied by severe abdominal pain and DIC commonly develops in response to blood being forced into uterine muscle tissue; referred to as a couvelaire uterus. Once half to two-thirds of the placenta is detached, the likelihood of fetal survival is low, especially if the woman is also hypotensive. In the majority of cases, only women with severe placental abruption are admitted to ICU and usually admission occurs following an emergency caesarean section. Understanding of the aetiology of placental abruption is not complete with approximately 20% of cases unexplained. For most women, placental abruption is associated with a known related factor like preeclampsia, blunt trauma (e.g. car crash) and sudden reduction in uterine volume (e.g. after delivery of the first baby in a twin pregnancy).
Placenta praevia
Placenta praevia is when some or the entire placenta is abnormally sited in the lower segment of the uterus, often referred to as a low-lying placenta. Placenta praevia is graded into four categories of severity according to the location of the placenta in relation to the cervix (Box 26.5). A vaginal birth is not possible with Grades III and IV as the placenta blocks the passage for the baby, necessitating a caesarean section. The lower uterine segment does not fully form until 28–32 weeks’ gestation and the shearing stress as the lower uterine segment forms may precipitate detachment of the placenta from the uterine wall causing maternal bleeding. However, bleeding can occur at any time, is usually painless and may be massive. Placenta praevia is the main cause of APH accounting for 30% of cases.92 As with placental abruption, management is dictated by the size of the blood loss and maternal condition, how much functioning placenta remains and fetal wellbeing, and whether bleeding is ongoing. In severe cases, the woman is usually taken to theatre for an emergency caesarean section.
Box 26.5
Categories of severity of placenta praevia
• Type I (low-lying placenta): The placenta is located in the lower uterine segment but does not impede on the internal cervical os.
• Type II (marginal): The placenta edge is aligned with the internal cervical os.
• Type III (partial): The placenta lies over and partially covers the internal cervical os.
• Type IV (complete): The placenta is centrally located over the cervix and completely covers it.
Placenta accreta is a serious complicating condition that may occur in conjunction with placenta praevia. The attachment of the placenta to the uterine wall is abnormal and is considered morbidly adherent. There are three levels of severity, although often all three are referred to as placenta accreta (Box 26.6). Placenta accreta is strongly associated with prior caesarean section and a woman with an anterior placenta praevia and a prior caesarean section should be actively screened for placenta accreta (by ultrasound or MRI) prior to any elective caesarean section. Placental tissue can be very invasive and may infiltrate local structures like the bladder. Many women with placenta accreta undergo emergency hysterectomy at the time of caesarean section, as a means to remove the placenta and control bleeding. An alternative management is to deliver the baby by caesarean section and leave the placenta in situ.93 As long as a portion of the placenta does not detach, there will be no bleeding and in most cases, the placenta will autolyse and be re-absorbed by the woman.
Postpartum Haemorrhage
Postpartum haemorrhage (PPH), a major cause of maternal death in developed and developing countries, is defined as ≥500 mL blood loss from the genital tract after the birth of the baby. The incidence and severity of PPH is increasing, in both caesarean and vaginal births.91,94–96 PPH rates commonly sit at around 10% of all births. Severe PPH lacks an agreed definition, with published definitions ranging from ‘≥1000 mL’ to ‘estimated blood loss ≥2500 mL or transfused ≥5 units of blood or received treatment for coagulopathy during the acute event’.12,97 Consequently, the incidence of severe PPH varies depending on how it has been defined and ranges from 3.7/1000 deliveries to 4.6/1000 deliveries.5,97 Additionally, PPH is also classified according to the timing of the haemorrhage in relation to the birth. Primary PPH occurs within the first 24 hours after birth whilst secondary PPH occurs from 24 hours up to six weeks following birth. Primary PPH is often caused by uterine atony, whilst secondary PPH is more likely to be associated with retained products and associated infection.
The causes of PPH are varied and have been classified by the four ‘Ts’: tone, tissue, trauma and thrombin (Box 26.7). The cause of the PPH should be identified and targeted with specific management, in conjunction with the general principles of haemorrhage management. See also Box 26.8.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree