• Discuss the physiologic adaptations of the neonate during the transition to extrauterine life. • Describe the sequence to follow in assessing the newborn. • Recognize deviations from normal physiologic findings during the examination of the newborn. • Explain thermoregulation in the neonate and the types of heat loss. • Describe the behavioral adaptations of the newborn, including periods of reactivity and sleep-wake states. • Discuss the sensory and perceptual functioning of the neonate. Additional related content can be found on the companion website at http://evolve.elsevier.com/Lowdermilk/Maternity/ • Anatomy Review: Sutures and Fontanels • Assessment Videos: Buttocks, Cremasteric Reflex, Legs (Symmetry, Length), Male Breasts (Supine Position), Moro Reflex, Neck (Posterior), Plantar Grasp, Rooting and Sucking, Upper Extremities • Case Study: Newborn Health Problems With the cutting of the umbilical cord the infant undergoes rapid and complex physiologic changes. The most critical and immediate adjustment a newborn makes at birth is the establishment of respirations. With a vaginal birth some lung fluid is squeezed from the newborn’s trachea and lungs; in infants who are born by cesarean birth some lung fluid may be retained within the alveoli. With the first breath of air the newborn begins a sequence of cardiopulmonary changes (Table 16-1). TABLE 16-1 Characteristics of the Respiratory System of the Neonate PCO2: Partial pressure of carbon dioxide. From Blackburn, S. (2007). Maternal, fetal, & neonatal physiology: A clinical perspective (3rd ed.). St. Louis: Saunders. The cardiovascular system changes significantly after birth. The infant’s first breaths, combined with increased alveolar capillary distention, inflate the lungs and reduce pulmonary vascular resistance to the pulmonary blood flow from the pulmonary arteries. Pulmonary artery pressure drops, and pressure in the right atrium declines. Increased pulmonary blood flow to the left side of the heart increases pressure in the left atrium, which causes a functional closure of the foramen ovale. During the first few days of life, crying may reverse the flow through the foramen ovale temporarily and lead to mild cyanosis (Table 16-2). TABLE 16-2 Cardiovascular Changes at Birth Unless a specific indication exists, BP is not usually measured in the newborn on a routine basis except as a baseline. The practice of obtaining four extremity BPs in the early newborn period to detect coarctation of the aorta (COA) has been recently questioned (Razmus & Lewis, 2006). This is based on evidence that COA defects do not occur in the immediate postbirth period but more typically at approximately 12 to 14 days of age, a time when the ductus arteriosus closes (Taylor, 2005). Early or late clamping of the umbilical cord changes the circulatory dynamics of the newborn. Late clamping expands the blood volume from the so-called placental transfusion of blood to the newborn. Delayed cord clamping (≥2 minutes after birth) has been reported to result in polycythemia with subsequent clinical signs of hyperviscosity (hematocrit ≥65%, plethoric or ruddy red appearance, sluggish circulation leading to possible emboli in the microvasculature and organ damage, respiratory distress, and possibly hyperbilirubinemia as a result of red cell breakdown) (Armentrout & Huseby, 2003). However, recent data showed delayed cord clamping (no longer than 2 minutes after birth) in full-term neonates was found to be beneficial in improving hematocrit and iron status and in decreasing anemia; such benefits were observed over ages 2 to 6 months. Polycythemia occurred with delayed clamping but was not harmful (Hutton & Hassan, 2007). Heat loss in the newborn occurs by four modes: • Convection is the flow of heat from the body surface to cooler ambient air. Because of heat loss by convection the ambient temperature in the nursery is kept at approximately 24° C, and newborns in open bassinets are wrapped to protect them from the cold. • Radiation is the loss of heat from the body surface to a cooler solid surface not in direct contact but in relative proximity. To prevent this type of loss, nursery cribs and examining tables are placed away from outside windows, and care is taken to avoid direct air drafts. • Evaporation is the loss of heat that occurs when a liquid is converted to a vapor. In the newborn, heat loss by evaporation occurs as a result of vaporization of moisture from the skin. This heat loss can be intensified by failure to dry the newborn directly after birth or by drying the infant too slowly after a bath. The less mature the newborn is, the more severe the evaporative heat loss will be. Evaporative heat loss, as a component of insensible water loss, is the most significant cause of heat loss in the first few days of life. • Conduction is the loss of heat from the body surface to cooler surfaces in direct contact. When admitted to the nursery the newborn is placed in a warmed crib to minimize heat loss. The scales used for weighing the newborn should have a protective cover to minimize conductive heat losses as well. Loss of heat must be controlled to protect the infant. Control of such modes of heat loss is the basis of caregiving policies and techniques. One method for promoting maternal-newborn interaction is to place the naked healthy newborn next to the mother’s skin and cover both with a blanket. This skin-to-skin contact enhances newborn temperature control and interaction (Fig. 16-1). The basal metabolic rate increases with cold stress (Fig. 16-2). If cold stress is protracted, anaerobic glycolysis occurs, resulting in increased production of acids. Metabolic acidosis develops, and if a defect in respiratory function is present, respiratory acidosis also develops. Excessive fatty acids may displace the bilirubin from the albumin-binding sites and exacerbate hyperbilirubinemia. At birth the lower intestine is filled with meconium. Meconium is formed during fetal life from the amniotic fluid and its constituents, intestinal secretions (including bilirubin), and cells (shed from the mucosa). Meconium is greenish black and viscous and contains occult blood. The first meconium passed is usually sterile, but within hours, all meconium passed contains bacteria. The majority of healthy term infants pass meconium within the first 12 to 24 hours of life, and almost all do so by 48 hours (Blackburn, 2007). The number of stools passed varies during the first week, being most numerous between the third and sixth days. Newborns fed early pass stools sooner. Progressive changes in the stooling pattern indicate a properly functioning gastrointestinal tract (Box 16-1). At birth the newborn is cut off from its maternal glucose supply and, as a result, has an initial decrease in serum glucose levels. The newborn’s increased energy needs, decreased hepatic release of glucose from glycogen stores, increased RBC volume, and increased brain size may initially contribute to the rapid depletion of stored glycogen within the first 24 hours after birth. In most healthy term newborns, blood glucose levels stabilize at 50 to 60 mg/dl during the first several hours after birth; by the third day of life the blood glucose levels should be approximately 60 to 70 mg/dl. The initiation of feedings assists in the stabilization of the newborn’s blood glucose levels. Colostrum contains high amounts of glucose thus also assisting in the stabilization of blood glucose levels in breastfed neonates (colostrum is higher in protein, but lower in glucose, than mature milk) (see Evidence-Based Practice box in Chapter 17). Jaundice is the manifestation of the pigment bilirubin in the tissues of the body. Jaundice does not usually appear until the bilirubin level reaches 5 mg/dl. Any visible jaundice within the first 24 hours of life or persistence of jaundice beyond 7 to 10 days requires further investigation into the cause as this represents an underlying pathologic process (Fig. 16-3). See Chapter 17 for a further discussion of bilirubin metabolism and hyperbilirubinemia. The healthy term newborn has a plump appearance because of large amounts of subcutaneous tissue and extracellular water content. Fine lanugo hair may be noted over the face, shoulders, and back. Edema of the face and ecchymosis (bruising) or petechiae may be present as a result of face presentation, forceps-assisted birth, or vacuum extraction (see Fig. 17-6). Caput succedaneum is a generalized, easily identifiable edematous area of the scalp, most commonly found on the occiput (Fig. 16-4, A). The sustained pressure of the presenting vertex against the cervix results in compression of local vessels, thereby slowing venous return. The slower venous return causes an increase in tissue fluids within the skin of the scalp, and an edematous swelling develops. This edematous swelling, present at birth, extends across suture lines of the skull and disappears spontaneously within 3 to 4 days. Infants who are born with the assistance of vacuum extraction usually have a caput in the area where the cup was applied. Subgaleal hemorrhage is bleeding into the subgaleal compartment (Fig. 16-4, C). The subgaleal compartment is a potential space that contains loosely arranged connective tissue; it is located beneath the galea aponeurosis, the tendinous sheath that connects the frontal and occipital muscles and forms the inner surface of the scalp. The injury occurs as a result of forces that compress and then drag the head through the pelvic outlet (Paige & Moe, 2006). Researchers have reported concern regarding the increased use of the vacuum extractor at birth and an association with cases of subgaleal hemorrhage, neonatal morbidity, and deaths (Boo, Foong, Mahdy, Yong, & Jaafar, 2005: Uchil & Arulkumaran, 2003). The bleeding extends beyond bone, often posteriorly into the neck, and continues after birth, with the potential for serious complications such as anemia or hypovolemic shock. Early detection of the hemorrhage is vital; serial head circumference measurements and inspection of the back of the neck for increasing edema and a firm mass are essential. A boggy scalp, pallor, tachycardia, and increasing head circumference may also be early signs of a subgaleal hemorrhage (Doumouchtsis & Arulkumaran, 2006). Computed tomography or magnetic resonance imaging is useful in confirming the diagnosis. Replacement of lost blood and clotting factors is required in acute cases of hemorrhage. Another possible early sign of subgaleal hemorrhage is a forward and lateral positioning of the infant’s ears because the hematoma extends posteriorly. Monitoring the infant for changes in level of consciousness and a decrease in the hematocrit are also key to early recognition and management. An increase in serum bilirubin levels may be seen as a result of the breakdown of blood cells within the hematoma. Sweat glands are present at birth but do not respond to increases in ambient or body temperature. Some fetal sebaceous gland hyperplasia and secretion of sebum result from the hormonal influences of pregnancy. Vernix caseosa is a product of the sebaceous glands. Removal of the vernix is followed by desquamation of the epidermis in most infants. Vernix has been shown to be an epidermal barrier with positive benefits for neonatal skin such as decreasing the skin pH, decreased skin erythema, and improved skin hydration (Visscher et al., 2005). Distended, small, white sebaceous glands (milia) may be noticeable on the newborn face. Mongolian spots, bluish-black areas of pigmentation, may appear over any part of the exterior surface of the body, including the extremities. They are more commonly noted on the back and buttocks (Fig. 16-5). These pigmented areas are most frequently noted in newborns whose ethnic origins are in the Mediterranean area, Latin America, Asia, or Africa. They are more common in dark-skinned individuals but may occur in 5% to 13% of Caucasians as well (Blackburn, 2007). They fade gradually over months or years. Telangiectatic nevi, known as “stork bites,” are pink and easily blanched (Fig. 16-6, A). They appear on the upper eyelids, nose, upper lip, lower occipital area, and nape of the neck. They have no clinical significance and fade by the second year of life.
Physiologic and Behavioral Adaptations of the Newborn
Web Resources
![]()
Physiologic Adaptations

Respiratory System
CHARACTERISTIC
EFFECT ON FUNCTION
Decreased lung elastic tissue and recoil
Decreased lung compliance requiring higher pressures and more work to expand; increased risk of atelectasis
Reduced diaphragm movement and maximal force potential
Less effective respiratory movement; difficulty generating negative intrathoracic pressures; risk of atelectasis
Tendency to nose breathe; altered position of larynx and epiglottis
Enhanced ability to synchronize swallowing and breathing; risk of airway obstruction; possibly more difficult to intubate
Small compliant airway passages with higher airway resistance; immature reflexes
Risk of airway obstruction and apnea
Increased pulmonary vascular resistance with sensitive pulmonary arterioles
Risk of ductal shunting and hypoxemia with events such as hypoxia, acidosis, hypothermia, hypoglycemia, and hypercarbia
Increased oxygen consumption
Increased respiratory rate and work of breathing; risk of hypoxia
Increased intrapulmonary right-left shunting
Increased risk of atelectasis with wasted ventilation; lower PCO2
Immaturity of pulmonary surfactant system in immature infants
Increased risk of atelectasis and respiratory distress syndrome; increased work of breathing
Immature respiratory control
Irregular respirations with periodic breathing; risk of apnea; inability to rapidly alter depth of respirations
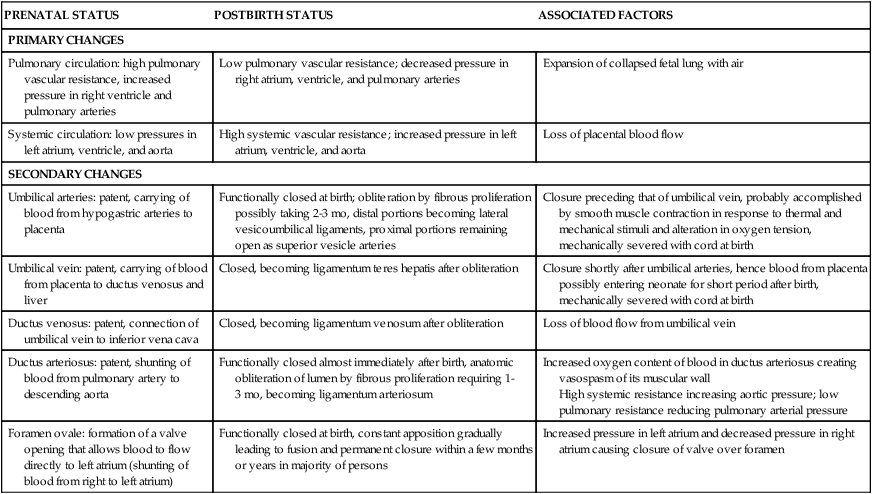
Cardiovascular System
PRENATAL STATUS
POSTBIRTH STATUS
ASSOCIATED FACTORS
PRIMARY CHANGES
Pulmonary circulation: high pulmonary vascular resistance, increased pressure in right ventricle and pulmonary arteries
Low pulmonary vascular resistance; decreased pressure in right atrium, ventricle, and pulmonary arteries
Expansion of collapsed fetal lung with air
Systemic circulation: low pressures in left atrium, ventricle, and aorta
High systemic vascular resistance; increased pressure in left atrium, ventricle, and aorta
Loss of placental blood flow
SECONDARY CHANGES
Umbilical arteries: patent, carrying of blood from hypogastric arteries to placenta
Functionally closed at birth; obliteration by fibrous proliferation possibly taking 2-3 mo, distal portions becoming lateral vesicoumbilical ligaments, proximal portions remaining open as superior vesicle arteries
Closure preceding that of umbilical vein, probably accomplished by smooth muscle contraction in response to thermal and mechanical stimuli and alteration in oxygen tension, mechanically severed with cord at birth
Umbilical vein: patent, carrying of blood from placenta to ductus venosus and liver
Closed, becoming ligamentum teres hepatis after obliteration
Closure shortly after umbilical arteries, hence blood from placenta possibly entering neonate for short period after birth, mechanically severed with cord at birth
Ductus venosus: patent, connection of umbilical vein to inferior vena cava
Closed, becoming ligamentum venosum after obliteration
Loss of blood flow from umbilical vein
Ductus arteriosus: patent, shunting of blood from pulmonary artery to descending aorta
Functionally closed almost immediately after birth, anatomic obliteration of lumen by fibrous proliferation requiring 1-3 mo, becoming ligamentum arteriosum
Increased oxygen content of blood in ductus arteriosus creating vasospasm of its muscular wall
High systemic resistance increasing aortic pressure; low pulmonary resistance reducing pulmonary arterial pressure
Foramen ovale: formation of a valve opening that allows blood to flow directly to left atrium (shunting of blood from right to left atrium)
Functionally closed at birth, constant apposition gradually leading to fusion and permanent closure within a few months or years in majority of persons
Increased pressure in left atrium and decreased pressure in right atrium causing closure of valve over foramen
Blood pressure
Blood volume
Thermogenic System
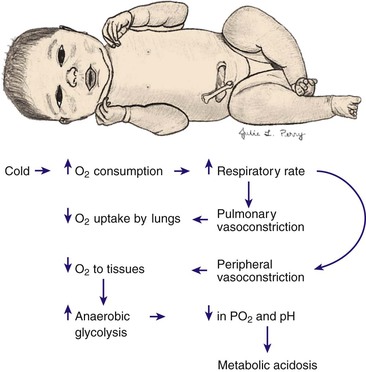
Heat loss
Temperature regulation
Gastrointestinal System
Stools
Hepatic System
Carbohydrate metabolism
Jaundice
Integumentary System
Caput succedaneum
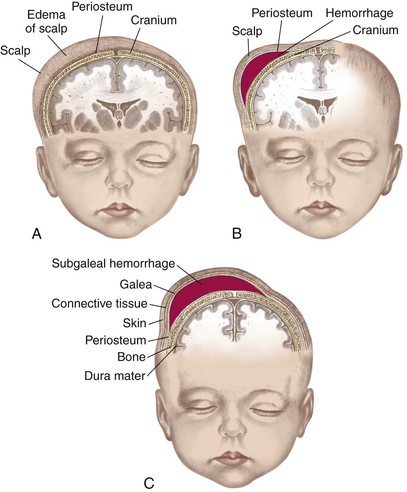
Subgaleal hemorrhage
Sweat glands
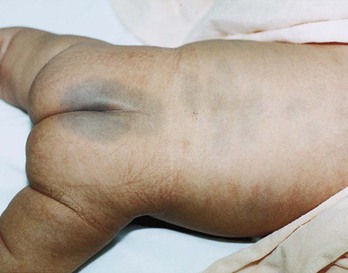
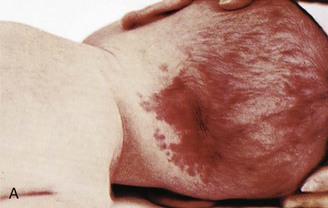
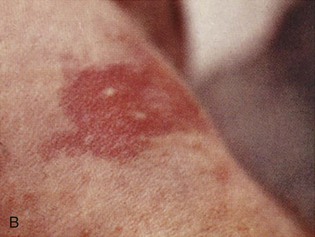
Mongolian spots
Nevi
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Physiologic and Behavioral Adaptations of the Newborn
Get Clinical Tree app for offline access