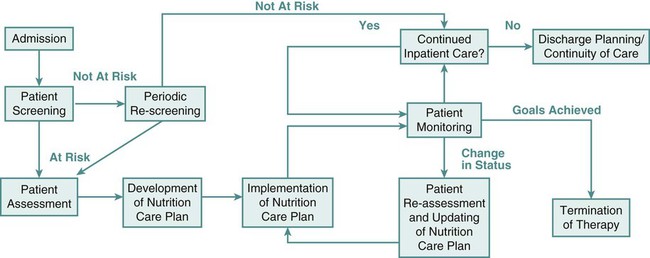
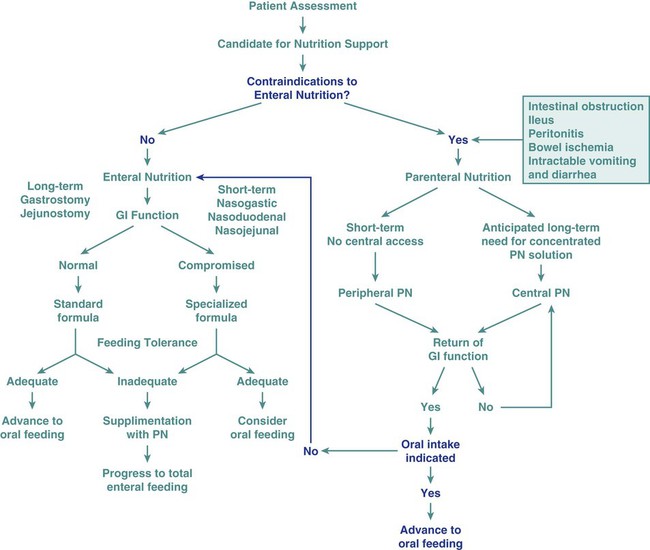
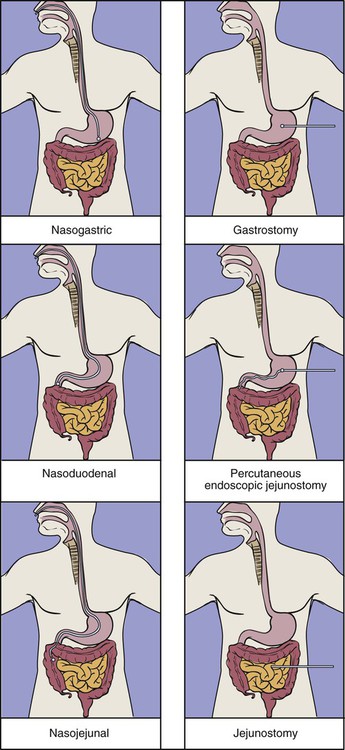
In the United States, The Joint Commission mandates nutrition screening be conducted on every patient within 24 hours of admission to an acute care center.1 A brief questionnaire to be completed by the patient or significant other, the nursing admission form, or the physician’s admission note usually provides enough information to determine whether the patient is at nutritional risk (Box 8-1). Any patient judged to be nutritionally at risk needs a more thorough nutrition assessment. The nutrition assessment process is continuous, with reassessments as a part of the overall nutrition care plan, as shown in Figure 8-1.2 Nutrition assessment involves collection of four types of information: 1) anthropometric measurements; 2) biochemical (laboratory) data; 3) clinical signs (physical examination); and 4) diet and pertinent health history. This information provides a basis for 1) identifying patients who are malnourished or at risk of malnutrition; 2) determining the nutritional needs of individual patients; and 3) selecting the most appropriate methods of nutrition support for patients with or at risk of developing nutritional deficits. Nutrition support is the provision of specially formulated or delivered oral, enteral, or parenteral nutrients to maintain or restore optimal nutrition status.3 The nutrition assessment can be performed by or under the supervision of a registered dietitian or by a nutrition care specialist (e.g., nurse with specialized expertise in nutrition). Figure 8-2 shows the route of administration of specialized nutrition support. Weight is measured in kilograms and height in meters. BMI values are independent of age and gender and are used for assessing health risk. BMI can be classified as shown in Table 8-1.4,5 Evidence that the associations between BMI, percent of body fat, and body fat distribution differ across populations suggests the possible need for developing different BMI cut-off values for different ethnic groups.6 For example, alternate BMI classification cut-off values exist for Asians, who are at risk for obesity-related co-morbidities at lower BMI.6 TABLE 8-1 From World Health Organization. Obesity: preventing and managing the global epidemic. Report of a WHO Consultation. WHO Technical Report Series 894. Geneva: World Health Organization; 2000. It may be impossible to measure the height of some patients accurately. Total height can be estimated from arm span length or knee height.7,8 To measure knee height, bend the knee 90 degrees, and measure from the base of the heel to the anterior surface of the thigh. In addition to height and weight data, other measurements such as arm muscle circumference, skin fold thickness, and body composition (proportion of fat and lean tissue, determined by bioelectric impedance or other methods) are sometimes performed, but these measurements are of limited use in assessing critically ill patients.9 A wide range of laboratory tests can provide information about nutritional status. Those most often used in the clinical setting are described in Table 8-2. No diagnostic tests for evaluation of nutrition are perfect, and care must be taken in interpreting the results of the tests.10 TABLE 8-2 COMMON BLOOD AND URINE TESTS USED IN NUTRITION ASSESSMENT A thorough physical examination is an essential part of nutrition assessment. Box 8-2 lists some of the more common findings that may indicate an altered nutritional state. It is especially important for the nurse to check for signs of muscle wasting, loss of subcutaneous fat, skin or hair changes, and impairment of wound healing. Information about dietary intake and significant variations in weight is a vital part of the history. Dietary intake can be evaluated in several ways, including a diet record, a 24-hour recall, and a diet history. The diet record, a listing of the type and amount of all foods and beverages consumed for some period (usually 3 days), is useful for evaluating the patient’s intake in the critical care setting if the adequacy of intake is questionable. However, such a record reveals little about the patient’s habitual intake before the illness or injury. The 24-hour recall of all food and beverage intake is easily and quickly performed, but it also may not reflect the patient’s usual intake and has limited usefulness. The diet history consists of a detailed interview about the patient’s usual intake, along with social, familial, cultural, economic, educational, and health-related factors that may affect intake. Although the diet history is time consuming to perform and may be too stressful for the acutely ill patient, it does provide a wealth of information about food habits over a prolonged period and a basis for planning individualized nutrition education if changes in eating habits are desirable. Other information to include in a nutrition history is listed in Box 8-3. A variety of methods can be used in clinical practice to estimate caloric requirements. Indirect calorimetry, a method by which energy expenditure is calculated from oxygen consumption (Vo2) and carbon dioxide production (Vco2), is the most accurate method for determining caloric needs.11,12 Indirect calorimetry is useful in those patients suspected to have a high metabolic rate. This test can also analyze substrate use, which can be extrapolated from VO2 and VCO2 during a steady state of respiration. The respiratory quotient (RQ) is equal to the VCO2 divided by the Vo2. Fat, protein, and carbohydrates each have a unique RQ, thus RQ identifies which substrate is being preferentially metabolized and may provide target goals for calorie replacement.12 For example, the RQ for the metabolism of fats is about 0.7 while the RQ for the metabolism of carbohydrates is 1.0. A mixed fuel diet results in an RQ of approximately 0.8.12 The test can be performed on spontaneously breathing patients and on those who require mechanical ventilation. Some ventilators are constructed so that they can perform indirect calorimetry. However, for most patients, indirect calorimetry requires the use of a metabolic cart, which is not available in all institutions. To maintain accuracy and reliability of measurement, several testing criteria must be met.12 Information received from the metabolic cart is limited; measurements are conducted over a relatively brief period (often 20 to 30 minutes) and may not be representative of energy expenditure over the whole day. Calorie and protein needs of patients are often estimated using formulas that provide allowances for increased nutrient use associated with injury and healing. Although indirect calorimetry is considered the most accurate method to determine energy expenditure, estimates using formulas have demonstrated reasonable accuracy.13–15 Commonly used formulas for critically ill patients can be found in Appendix B. Some rules of thumb are available to provide a rough estimate of caloric needs so that nurses and other caregivers can quickly determine if patients are being seriously overfed or underfed (Table 8-3). TABLE 8-3 The goal of nutrition assessment is to obtain the most accurate estimate of nutritional requirements. Underfeeding and overfeeding must be avoided during critical illness. Overfeeding results in excessive production of carbon dioxide, which can be a burden in the person with pulmonary compromise. Overfeeding increases fat stores, which can contribute to insulin resistance and hyperglycemia. Hyperglycemia increases the risk of postoperative infections in diabetic and nondiabetic individuals.16 As many as 40% of hospitalized patients are at risk for malnutrition.17–20 Although illness or injury is the major factor contributing to development of malnutrition, other possible contributing factors are lack of communication among the nurses, physicians, and dietitians responsible for the care of these patients; frequent diagnostic testing and procedures, which lead to interruption in feeding; medications and other therapies that cause anorexia, nausea, or vomiting and thereby interfere with food intake; insufficient monitoring of nutrient intake; and inadequate use of supplements, tube feedings, or parenteral nutrition to maintain the nutritional status of these patients. Nutritional status tends to deteriorate during hospitalization unless appropriate nutrition support is started early and continually reassessed. Malnutrition in hospitalized patients is associated with a wide variety of adverse outcomes. Wound dehiscence, pressure ulcers, sepsis, infections, respiratory failure requiring ventilation, longer hospital stays, and death are more common among malnourished patients.21–23 Decline in nutritional status during hospitalization is associated with higher incidences of complications, increased mortality rates, increased length of stay, and higher hospital costs. Nutrition support is an important aspect of the care of critically ill patients. Maintenance of optimal nutritional status may prevent or reduce the complications associated with critical illness and promote positive clinical outcomes.3 Critical care nurses play a key role in the delivery of nutrition support and must work closely with dietitians and physicians in promoting the best possible outcomes for their patients. Nutrition support is the provision of oral, enteral, or parenteral nutrients. It is an essential adjunct in the prevention and management of malnutrition in critically ill patients.3 The goal of nutrition support therapy is to provide enough support for body requirements, to minimize complications, and to promote rapid recovery. Critical care nurses must have a broad understanding of nutrition support, including the indications, prevention, and management of associated complications. 1. Collaborate with the dietitian to choose appropriate products and allow the patient to participate in the selection process, if possible. Milk shakes and instant breakfast preparations are often more palatable and economical than commercial supplements. However, lactose intolerance is common among adults. Many disease processes (e.g., Crohn’s disease, radiation enteritis, human immunodeficiency virus [HIV] infection, severe gastroenteritis) can cause lactose intolerance. Individuals with this problem require commercial lactose-free supplements or milk treated with lactase enzyme. 2. Offer to serve commercial supplements well chilled or on ice, because this improves flavor. 3. Advise patients to sip formulas slowly, consuming no more than 240 mL over 30 to 45 minutes. These products contain easily digestible carbohydrates. If formulas are consumed too quickly, rapid hydrolysis of the carbohydrate in the duodenum can contribute to dumping syndrome, characterized by abdominal cramping, weakness, tachycardia, and diarrhea. 4. Record all supplement intake separately on the intake-and-output sheet so that it can be differentiated from intake of water and other liquids. Enteral nutrition or tube feedings are used for patients who have at least some digestive and absorptive capability but are unable or unwilling to consume enough by mouth. When possible, the enteral route is the preferred method of feeding over total parenteral nutrition (TPN). The proposed advantages of enteral nutrition over TPN include lower cost, better maintenance of gut integrity, and decreased infection and hospital length of stay.3 A review of the literature comparing enteral nutrition and TPN indicates that enteral nutrition is less expensive than TPN and is associated with a lower risk of infection.3,24 The gastrointestinal (GI) tract plays an important role in maintaining immunologic defenses, which is why nutrition by the enteral route is thought to be more physiologically beneficial than TPN. Some of the barriers to infection in the GI tract include neutrophils; the normal acidic gastric pH; motility, which limits GI tract colonization by pathogenic bacteria; the normal gut microflora, which inhibit growth of or destroy some pathogenic organisms; rapid desquamation and regeneration of intestinal epithelial cells; the layer of mucus secreted by GI tract cells; and bile, which detoxifies endotoxin in the intestine and delivers immunoglobulin A (IgA) to the intestine. A second line of defense against invasion of intestinal bacteria is the gut-associated lymphoid tissue (GALT).25 The systemic immune defenses in the GI tract are stimulated by the presence of food within it. In animal models, resting the GI tract by providing TPN contributes to bacterial translocation, whereby bacteria normally found in the GI tract cross the intestinal barrier, are found in the regional mesenteric lymph nodes, and give rise to generalized sepsis. However, there is insufficient evidence in humans that TPN causes atrophy of the intestinal mucosa or that enteral nutrition prevents bacterial translocation.26,27 Patients who are experiencing severe stress that greatly increases their nutritional needs (caused by major surgery, burns, or trauma) often benefit from tube feedings. Table 8-4 lists different enteral formula types and the nutritional indications for using each one. Individuals who require elemental formulas because of impaired digestion or absorption or the specialized formulas for altered metabolic conditions usually require tube feeding because the unpleasant flavors of the free amino acids, peptides, or protein hydrolysates used in these formulas are very difficult to mask if taken in orally. TABLE 8-4 *These diets may be beneficial for selected patients; costs and benefits must be considered. Immune-enhancing formulas (IEFs) have emerged as a means to protect and stimulate the immune system. Some of the enterally delivered nutrients that may benefit critically ill patients include fiber, the amino acids glutamine and arginine, the omega-3 (n-3) fatty acids, and the nucleotide ribonucleic acid (RNA).28 Fiber is not digested by humans but can be metabolized by gut bacteria to yield short-chain fatty acids, the primary fuel of the colon cells. Glutamine is the major fuel of the small intestinal cells. It is considered a nonessential amino acid, but it becomes conditionally essential in illness. It has been shown to improve mortality and infectious morbidity in critically ill patients.29–31 Arginine is involved in protein synthesis and is a precursor of nitric oxide, a molecule that stimulates vasodilation in the GI tract and heart and mediates hepatic protein synthesis during sepsis.28 The omega-3 fatty acids, derived primarily from fish oils, are involved in synthesis of eicosanoids (molecules with hormone-like activity)—prostaglandins, prostacyclin, and leukotrienes—and may modulate the inflammatory response. There are a variety of commercial enteral feeding products, some of which are designed to meet the specialized needs of the critically ill. Products designed for the stressed patient with trauma or sepsis are usually rich in glutamine, arginine, branched amino acids (a major fuel source, especially for muscle), and antioxidant nutrients, such as selenium and vitamins C, E, and A.32 The antioxidants help to reduce oxidative injury to the tissues (e.g., from reperfusion injury). Despite the fact that IEFs may reduce the incidence of infectious complications, the efficacy and safety of these formulas in critically ill patients have not been clearly demonstrated.33–36 Early enteral nutrition, administered with the first 24 to 48 hours of critical illness, has been advocated as a way to reduce septic complications and improve feeding tolerance in critically ill patients. Although studies37 have shown a lower risk of infection and decreased length of stay with early enteral nutrition, the benefit of early enteral nutrition compared with enteral nutrition delayed a few days remains controversial.3,38 Current guidelines support the initiation of nutrition support in critically ill patients who will be unable to meet their nutrient needs orally for a period of 5 to 10 days.3 To avoid complications associated with intestinal ischemia and infarction, enteral nutrition must be initiated only after fluid resuscitation and adequate perfusion have been achieved.39,40 Critically ill patients may not tolerate early enteral feeding because of impaired gastric motility, ileus, or medications administered in the early phase of illness. This is particularly true for patients receiving gastric enteral feeding.41 The assessment of enteral feeding tolerance is an important aspect of nursing care. Monitoring of gastric residual volume is a method used to assess enteral feeding tolerance. However, evidence suggests that gastric residuals are insensitive and unreliable markers of tolerance to tube feeding.42 There is little evidence to support a correlation between gastric residual volumes and tolerance to feedings, gastric emptying, and potential aspiration. Except in selected high-risk patients, there is little evidence to support holding tube feedings in patients with gastric residual volumes less than 400 mL.42 The gastric residual volume should be evaluated within the context of other gastrointestinal symptoms. Prokinetic agents, including metoclopramide and erythromycin, have been used to improve gastric motility and promote early enteral nutrition in critically ill patients.43–45 Achievement of enteral access is the cornerstone of enteral nutrition therapy. Several techniques can be used to facilitate enteral access. These include surgical methods, bedside methods, fluoroscopy, endoscopy, air insufflation, and prokinetic agents.9 Placement of feeding tubes beyond the stomach (postpyloric) eliminates some of the problems associated with gastric feeding intolerance. However, placement of postpyloric feeding tubes is time-consuming and may be costly. Tubes with weights on the proximal end are available; they were originally designed for postpyloric feeding in the belief that that they would be more likely than unweighted tubes to pass spontaneously through the pyloric sphincter. However, randomized trials with the two types of tubes have shown that unweighted tubes are more likely to migrate through the pylorus than weighted tubes.46 The weights sometimes cause discomfort while being inserted through the nares. Unweighted tubes therefore may be preferable. After the tube is placed, correct location must be confirmed before feedings are started and regularly throughout the course of enteral feedings. Radiographs are the most accurate way of assessing tube placement, but repeated radiographs are costly and can expose the patient to excessive radiation. After correct placement has been confirmed, marking the exit site of the tube to check for movement is helpful. Alternative methods for confirming tube placement have been researched that attempt to verify placement in the stomach or small intestine. An inexpensive and relatively accurate alternative method involves assessing the pH of fluid removed from the feeding tube; some tubes are equipped with pH monitoring systems. Assessing the pH and the bilirubin concentration in fluid aspirated from the feeding tube is a newer method for confirming tube placement.47 Decisions regarding enteral access should be determined based on gastrointestinal anatomy, gastric emptying, and aspiration risk.3 Nasal intubation is the simplest and most commonly used route for enteral access. This method allows access to the stomach, duodenum, or jejunum. Tube enterostomy—a gastrostomy or jejunostomy—is used primarily for long-term feedings (6 to 12 weeks or more) and when obstruction makes the nasoenteral route inaccessible. Tube enterostomies may also be used for the patient who is at risk for tube dislodgment because of severe agitation or confusion. A conventional gastrostomy or jejunostomy is often performed at the time of other abdominal surgery. The percutaneous endoscopic gastrostomy (PEG) tube has become extremely popular because it can be inserted at the bedside without the use of general anesthetics. Percutaneous endoscopic jejunostomy (PEJ) tubes are also used. Postpyloric feedings through nasoduodenal, nasojejunal, or jejunostomy tubes are commonly used when there is a high risk of pulmonary aspiration, because the pyloric sphincter theoretically provides a barrier that lessens the risk of regurgitation and aspiration.48 However, some studies have demonstrated that gastric feeding is safe and not associated with an increased risk of aspiration.49–51 Postpyloric feedings have an advantage over intragastric feedings for patients with delayed gastric emptying, such as those with head injury, gastroparesis associated with uremia or diabetes, or postoperative ileus. Delivery of enteral nutrition into the small bowel is associated with improved tolerance,52 higher calorie and protein intake,53 and fewer gastrointestinal complications.41 Small bowel motility returns more quickly than gastric motility after surgery, and it is often possible to deliver transpyloric feedings within a few hours of injury or surgery.48 Figure 8-3 shows the locations of tube feeding sites.
Nutrition Alterations and Management
Assessing Nutritional Status
Anthropometric Measurements
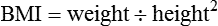
CLASSIFICATION
BMI (kg/m2)
Underweight
<18.5
Normal
18.5-24.99
Overweight:
≥25
Pre-obese
25-29.99
Obese class I
30-34.99
Obese class II
35-39.99
Obese class III
≥40
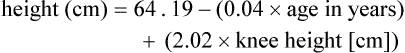
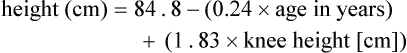
Biochemical Data
TEST
COMMENTS AND LIMITATIONS
Serum Proteins
Albumin or prealbumin
Levels decrease with protein deficiency and in liver failure. Albumin levels are slow to change in response to malnutrition and repletion. Prealbumin levels fall in response to trauma and infection.
Hematologic Values
Anemia
Normocytic (normal MCV, MCHC)
Common with protein deficiency
Microcytic (decreased MCV, MCH, MCHC)
Indicative of iron deficiency (can be from blood loss)
Macrocytic (increased MCV)
Common in folate and vitamin B12 deficiency
Lymphocytopenia
Common in protein deficiency
Clinical or Physical Manifestations
Diet and Health History
Determining Nutritional Needs
CATEGORY
DESCRIPTION
CALORIES/kg
CALORIES/lb
Obese
More than 40% over ideal body weight or BMI >30
21
9.5
Sedentary
Relatively inactive individual without regular aerobic exercise; hospitalized patient without severe injury or sepsis
25-30
11-13.5
Moderate activity or injury
Individual obtaining regular aerobic exercise plus routine activities; patient with trauma or sepsis
30-35
13.5-16
Very active or severe injury
Manual laborer or athlete in very active training; patient with major burns or trauma
40
18

Implications of Undernutrition for the Sick or Stressed Patient
Nutrition Support
Nursing Management of Nutrition Support
Oral Supplementation
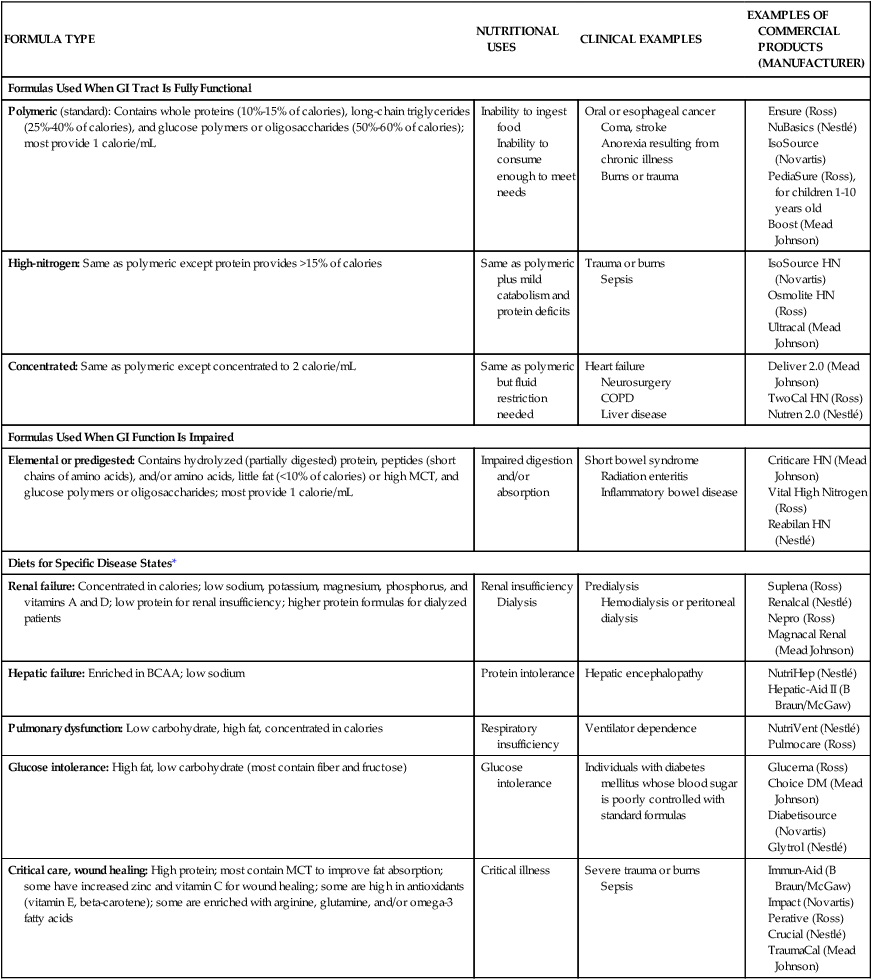
Enteral Nutrition
FORMULA TYPE
NUTRITIONAL USES
CLINICAL EXAMPLES
EXAMPLES OF COMMERCIAL PRODUCTS (MANUFACTURER)
Formulas Used When GI Tract Is Fully Functional
Polymeric (standard): Contains whole proteins (10%-15% of calories), long-chain triglycerides (25%-40% of calories), and glucose polymers or oligosaccharides (50%-60% of calories); most provide 1 calorie/mL
Inability to ingest food
Inability to consume enough to meet needs
Oral or esophageal cancer
Coma, stroke
Anorexia resulting from chronic illness
Burns or trauma
High-nitrogen: Same as polymeric except protein provides >15% of calories
Same as polymeric plus mild catabolism and protein deficits
Trauma or burns
Sepsis
Concentrated: Same as polymeric except concentrated to 2 calorie/mL
Same as polymeric but fluid restriction needed
Heart failure
Neurosurgery
COPD
Liver disease
Formulas Used When GI Function Is Impaired
Elemental or predigested: Contains hydrolyzed (partially digested) protein, peptides (short chains of amino acids), and/or amino acids, little fat (<10% of calories) or high MCT, and glucose polymers or oligosaccharides; most provide 1 calorie/mL
Impaired digestion and/or absorption
Short bowel syndrome
Radiation enteritis
Inflammatory bowel disease
Diets for Specific Disease States*
Renal failure: Concentrated in calories; low sodium, potassium, magnesium, phosphorus, and vitamins A and D; low protein for renal insufficiency; higher protein formulas for dialyzed patients
Renal insufficiency
Dialysis
Predialysis
Hemodialysis or peritoneal dialysis
Hepatic failure: Enriched in BCAA; low sodium
Protein intolerance
Hepatic encephalopathy
Pulmonary dysfunction: Low carbohydrate, high fat, concentrated in calories
Respiratory insufficiency
Ventilator dependence
Glucose intolerance: High fat, low carbohydrate (most contain fiber and fructose)
Glucose intolerance
Individuals with diabetes mellitus whose blood sugar is poorly controlled with standard formulas
Critical care, wound healing: High protein; most contain MCT to improve fat absorption; some have increased zinc and vitamin C for wound healing; some are high in antioxidants (vitamin E, beta-carotene); some are enriched with arginine, glutamine, and/or omega-3 fatty acids
Critical illness
Severe trauma or burns
Sepsis
Enteral Feeding Access.
Location and Type of Feeding Tube.
![]()
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Nutrition Alterations and Management
Get Clinical Tree app for offline access

 NH2). Amino acids are the protein components that can be used by cells.
NH2). Amino acids are the protein components that can be used by cells.