Table 20-1. RISK OF SPECIFIC MEDICAL CONDITIONS THAT MAY BE ASSOCIATED WITH NOT BREASTFEEDING | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
(Castrucci, Hoover, Lim, & Maus, 2007). It is important to note that the higher rate was associated with hospitals that used lactation consultants. Among late preterm infants (34 0/7 to 36 6/7 weeks’ gestation), breastfeeding initiation was reported between 59% and 70%, with a significant reduction in breastfeeding duration and exclusivity (Radtke, 2011).
Table 20-2 HEALTHY PEOPLE BREASTFEEDING GOALS | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||
and newborns in the prenatal and postpartum periods (Display 20-3). Many of those responsibilities are aimed at reducing barriers to breastfeeding.
Apathy
Misinformation
Professional education that lacks information on breastfeeding
Outdated clinical practices
Failing to provide skilled support (i.e., lactation experts)
Routine separation of mother-infant dyad
Delay of first feeding
Routine formula or water supplementation
Use of pacifiers
Lack of staff training
Lack of a breastfeeding policy
Inappropriate interventions (i.e., supplemental feeding, pacifiers, overuse of nipple shields)
Disruptions of breastfeeding
Discharge packs that include formula samples and/or coupons for formula
Lack of discharge policy
Lack of follow-up after discharge
From partner, peers, and family
From workplace
From healthcare professionals
Media portrayal of bottle-feeding as normal
Commercial pressures on mothers to bottle-feed or supplement with formula
Formula club sign-up sheets in obstetric offices and clinics
Prenatal formula starter kits
Coupons for free formula
Formula ads in parent magazines
Formula ads on Internet sites of interest to parents
Discounted formula available through the Internet
Have a written breastfeeding policy that is routinely communicated to all healthcare staff (see Model Breastfeeding Policy [Academy of Breastfeeding Medicine [ABM], 2010b]).
Educate healthcare providers in skills necessary to implement this policy.
Inform all pregnant women about the benefits and management of breastfeeding.
Help mothers initiate breastfeeding within 1 hour of birth.
Show mothers how to breastfeed and how to maintain lactation even when they are separated from their newborns.
Give newborns no food or drink other than breast milk, unless medically indicated.
Practice “rooming-in”; allow mothers and newborns to remain together 24 hours each day.
Encourage unrestricted breastfeeding.
Give no artificial nipples or pacifiers to breastfeeding newborns.
Foster the establishment of breastfeeding support groups and refer mothers to them on discharge from the hospital or clinic.
become prominent and start to secrete a substance that lubricates and protects the nipples and areola. The areola grows in diameter and darkens. Internal changes in the breast also occur and include growth and differentiation of the mammary ducts as well as development of the lobules and alveoli. Sometime in the second trimester, lactogenesis I begins (Lauwers & Swisher, 2011; Lawrence & Lawrence, 2011).
Attain knowledge about the benefits of breastfeeding, including anatomy and physiology of lactation, initiation of lactation, and management of common concerns and problems.
Provide preconception and antenatal counseling on the benefits of breastfeeding.
Provide breastfeeding education to all women during prenatal period, including exploration of concerns, fears, and myths that may inhibit successful breastfeeding.
Work in collaboration with lactation specialists and other healthcare providers to optimize the breastfeeding experience for the mother and infant.
Integrate culturally appropriate and sensitive information into all breastfeeding education.
Ensure that breastfeeding is initiated in the immediate postpartum period whenever possible.
Promote nonseparation of mother and baby during the postpartum period.
Provide information about breastfeeding resources in the community at the time of hospital or birthing center discharge.
Use and conduct research related to breastfeeding.
milk-ejection reflex. Some mothers feel this as a heaviness or tingling sensation in the breast. Other mothers never feel the milk let down but observe milk leaking from the other breast or hear the newborn swallowing milk (Riordan & Wambach, 2010; Walker, 2011).
Infants receiving adequate amounts of breast milk do not need additional water, even in hot climates (Almroth, 1978; Ashraf, Jalil, Aperia, & Lindblad, 1993).
machines, monitoring devices, and alarms (AAFP, 2008). Other sources of stress for NICU mothers often include fear for their infant, separation from their infant, or concerns about the cost of intensive care.
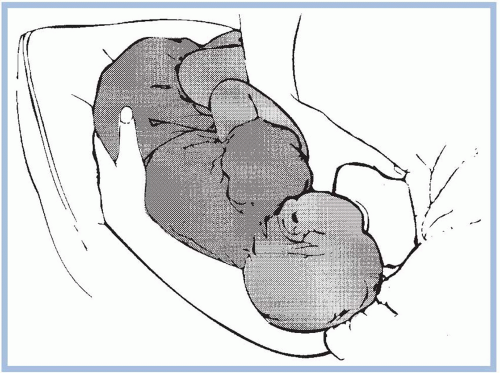
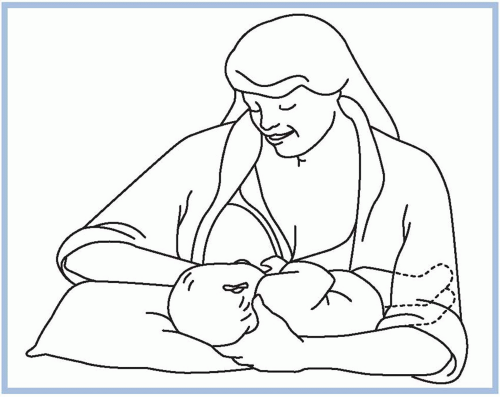
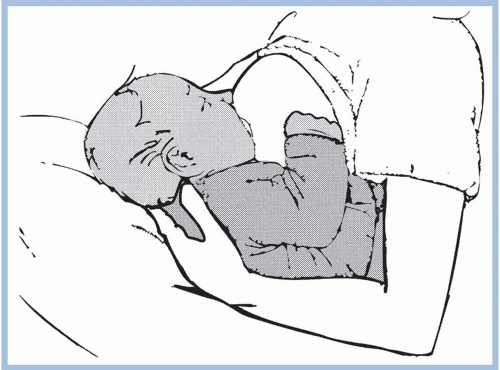
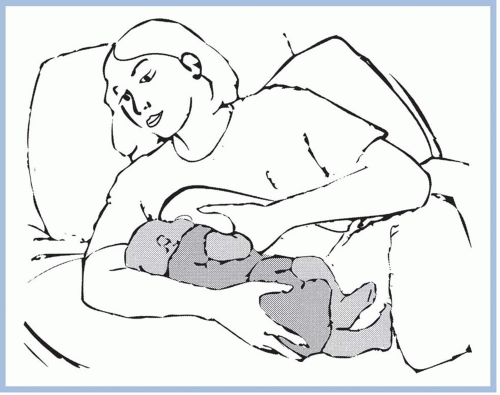
should be taken to avoid pushing the newborn’s head into the breast. Pressure on the occipital region of the head causes extension of the neck. Tilting, squeezing, or distorting the nipple or areola should also be avoided because doing so can cause pain and skin damage. If the mother feels a pinching or biting sensation while nursing, she should be instructed to pull down gently on the newborn’s chin. This causes his mouth to open wider so that more of the areola is drawn into his mouth. If this does not work, have the mother insert her little finger into the side of the newborn’s mouth to release the suction (Riordan & Wambach, 2010; Walker, 2011). She should begin again to achieve a better latch on.
Lips are rolled outward (flared).
Clicking or smacking sounds are absent.
Dimpled cheeks are absent.
Muscles above and in front of the ear move.
Both cheeks are equally close to the breast.
Chin and nose are touching the breast.
All of the nipple and part of the areola is covered by the newborn’s mouth.
More of the areola is visible above the upper lip than below it.
Angle at the corner of the mouth is wide.
When the lower lip is gently pulled away from the breast, the tongue is visible over the lower gum line.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree