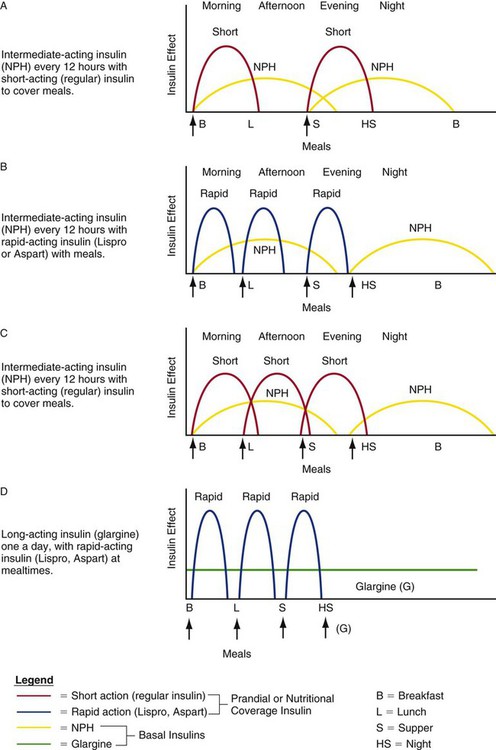
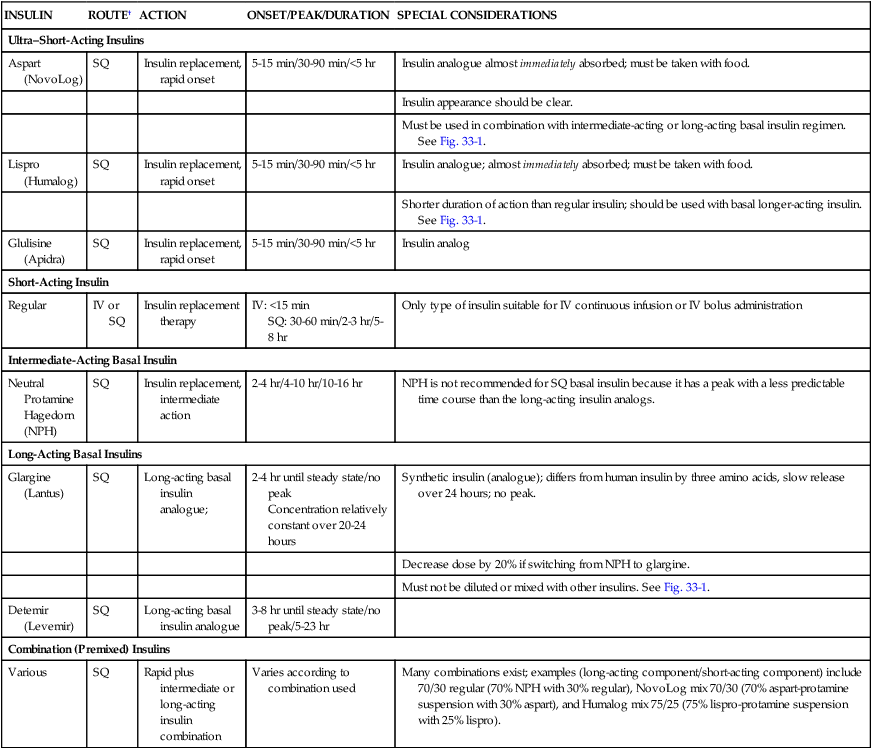
Chapter 33 Major neurologic and endocrine changes occur when an individual is confronted with physiologic stress caused by any critical illness, sepsis,1 trauma, major surgery, or underlying cardiovascular disease.2 The normal “fight or flight” response that is initiated in times of physiologic or psychologic stress is exacerbated in critical illness through activation of the neuroendocrine system, specifically the hypothalamic-pituitary-adrenal axis (HPA).3 All endocrine organs are affected by acute critical illness, as shown in Table 33-1. TABLE 33-1 The fight or flight acute response to physiologic threat is a rapid discharge of the catecholamines norepinephrine and epinephrine into the bloodstream.3 Norepinephrine is released from the nerve endings of the sympathetic nervous system (SNS). Epinephrine (adrenalin) is released from the medulla of the adrenal glands. Epinephrine increases cerebral blood flow and cerebral oxygen consumption and may be the trigger for recruitment of the hypothalamic-pituitary axis.3 The pituitary gland has two parts (anterior and posterior) that function under control of the hypothalamus, as described in Chapter 31. The posterior pituitary gland releases antidiuretic hormone (ADH), also known as vasopressin (pitressin), as a component of the physiologic stress response. This hormone is an antidiuretic with a powerful vasoconstrictive effect on blood vessels.3 The combination of epinephrine and vasopressin quickly raises blood pressure; it also decreases gastric motility.3 Epinephrine increases heart rate, causes ventricular dysrhythmias in susceptible patients, and provides some analgesia or lack of pain awareness during acute physical stress.3 The anterior pituitary gland produces several hormones including corticotropin (also called ACTH), which stimulates release of cortisol from the adrenal cortex.4 Cortisol release is an important protective response to stress. Increased cortisol levels alter carbohydrate, fat, and protein metabolism so that energy is immediately and selectively available to vital organs such as the brain. However, if critical illness is prolonged, the HPA may not be able to respond adequately to prolonged physiologic stress. • Primary hypoadrenalism describes an intrinsic failure of the adrenal gland to produce normal endogenous glucocorticosteroid hormones (e.g., cortisol) and mineralocorticosteroid hormones (e.g., aldosterone). Primary adrenal failure is rare. • Secondary adrenal dysfunction, or Cushing syndrome, occurs as a result of the administration of therapeutic steroids. In response to exogenous glucocorticosteroids, the adrenal glands stop production of intrinsic hormones. Patients who have taken steroids before their admission to the hospital need their dosage increased during illness. • Critical illness—related corticosteroid insufficiency (CIRCI) describes a situation in which the adrenal gland produces glucocorticosteroids but the quantity is insufficient for the disease process. Peripheral cortisol resistance occurs as inflammatory cytokines induce cellular resistance to cortisol.5 Further confirmation of adrenal dysfunction may be obtained by performance of a corticotropin stimulation test (cosyntropin test). Cosyntropin is a medication made from the first 24 amino acids of corticotropin. In the test, 250 mcg cosyntropin is administered by the intravenous (IV) route, and serum blood levels are measured 30 minutes later. A serum cortisol rise from baseline of less than 9 mcg/dL after 30 minutes denotes inability of the adrenal gland to respond to a stress stimulus (nonresponder).5 If the cortisol rise is greater than 9 mcg/dL in response to corticotropin stimulation, the adrenal glands are functioning normally (responder).5 Corticosteroids are given only to nonresponders. The combination of a low baseline cortisol value (less than 10 mcg/dL) with minimal or no rise (less than 9 mcg/dL) after cosyntropin stimulation is evidence of adrenal gland dysfunction with corticosteroid deficiency.5 Clinical practice guidelines5 recommend short-term provision of low-dose hydrocortisone for patients who have a diagnosis of septic shock with refractory vasopressor-dependent hypotension. Hydrocortisone is the recommended replacement because it is the pharmacologic steroid that most resembles endogenous cortisol. The guidelines recommend use of the cosyntropin stimulation test as described previously. However, the test is not recommended as a stand-alone method to identify patients who might receive low-dose steroids. This apparent contradiction is explained by the fact that several clinical trials of low-dose steroid replacement in sepsis have demonstrated a faster resolution of the shock symptoms but no difference in overall mortality compared with placebo.5 High-dose steroid replacement is never recommended in the management of sepsis. Corticosteroids are never discontinued abruptly and must be tapered gradually over several days.5 The liver releases the hormone glucagon to stimulate the liver to pour additional glucose into the bloodstream in response to physiologic stress. Glucagon rapidly raises blood glucose levels. Peripheral tissues may become insulin resistant, meaning the tissues are unable to use the available insulin to transport glucose inside the cells. This further raises blood glucose levels, causing persistent stress-induced hyperglycemia.6 There is a second metabolic system that enables insulin to enter the cell (see Chapter 31). The insulin-independent glucose transporters (GLUT 1, GLUT 2, and GLUT 3) are active during physiologic stress, but may be unable to keep up with the massive increase in glucose production by the liver. Continuous infusion of insulin to return and maintain blood glucose levels within a safe, near-normal range reduces morbidity and mortality.2 Normal fasting blood glucose levels range between 70 and 100 mg/dL in a healthy person. Critically ill patients frequently have much higher blood glucose levels, and several retrospective analyses have reported that hyperglycemic patients have a higher mortality rate than patients with normal blood glucose values. In 2001, a landmark prospective, randomized study showed a significant reduction in morbidity and mortality among critically ill surgical patients whose blood glucose concentration was maintained between 80 and 110 mg/dL with a continuous insulin infusion, compared with those whose blood glucose was only treated if it was greater than 180 mg/dL.7 A study of medical critical care patients by the same group with the same protocol demonstrated a survival benefit after 3 days of tight glucose control with a continuous insulin infusion.8 These initial studies were greeted with tremendous enthusiasm and many critical care units adopted stringent glucose control standards to reduce hyperglycemia-associated morbidity and mortality. However, achievement of such tight glucose control outside of a research trial can be challenging as shown by the results of more recent clinical trials. The NICE-SUGAR trial was a prospective randomized trial of 6014 critically ill patients. It compared continuous insulin infusion to achieve tight glucose control (target 81 to 108 mg/dL) with a conventional glucose control range (target below 180 mg/dL).9 In the tight glucose control group 6.8% had episodes of severe hypoglycemia (below 40 mg/dL); in the conventional control group only 0.5% experienced severe hypoglycemia.9 There was a 2.6% higher risk of death in the intensive glucose control group (27.5% died) compared with the conventional control group (24.9% died).9 As a result of the studies just described, clinical practice guidelines were developed by the American Association of Clinical Endocrinologists (AACE) and the American Diabetes Association (ADA) that recommend the use of continuous insulin infusions to maintain blood glucose in critical care patients between 140 and 180 mg/dL, with frequent monitoring of blood glucose.2 The 140 to 180 mg/dL level was selected to minimize the risk of hypoglycemia. Other glucose-control guidelines relevant to critical illness have also been published. The Society of Critical Care Medicine (SCCM) recommends initiating glycemic control when the blood glucose rises above 150 mg/dL.1,10 Insulin management must be initiated if the blood glucose level is above 180 mg/dL.2,10 More liberal glucose control represents the current trend of targeted values. As a result of the research that has highlighted the deleterious effects of hyperglycemia in critical illness, most hospitals have developed an institution-specific glucose–insulin algorithm to lower blood glucose into the targeted range.11 The vigilance of the critical care nurse is pivotal to the success of any intervention to lower blood glucose using a continuous insulin infusion. As discussed earlier, many glucose control protocols are using less restrictive ranges due to concerns about iatrogenic hypoglycemia. Monitoring the blood glucose with a point-of-care glucometer is the basis of targeted glucose control. As part of the comprehensive initial assessment, the blood sugar is measured by a standard laboratory sample or by a finger-stick capillary blood sample. In many institutions, if the blood sugar is greater than 180 mg/dL, the patient is started on a continuous IV insulin infusion. While the glucose is elevated, blood sample measurements are usually obtained hourly, to allow titration of the insulin drip to lower blood glucose.11 After the patient is stable, blood glucose measurements can be spaced approximately every 2 hours, based on individual hospital protocols. Many hospitals use insulin infusion protocols for management of stress-induced hyperglycemia that are implemented by the critical care nurse.2,11 Effective glucose protocols gauge the insulin infusion rate based on two parameters: 1) the immediate blood glucose result and 2) the rate of change in the blood glucose level since the last hourly measurement. The following three examples illustrate this concept: • Patient A receives 3 units of continuous IV regular insulin per hour and has a blood glucose measurement of 110 mg/dL, but 1 hour ago it was 190 mg/dL; the insulin rate must be decreased to avoid sudden hypoglycemia. • Patient B receives 3 units of continuous IV regular insulin per hour and has a blood glucose measurement of 110 mg/dL, but 1 hour ago it was 112 mg/dL; in this situation, no change is made in the insulin infusion rate. • Patient C receives 3 units of continuous IV regular insulin per hour and has a blood glucose measurement of 190 mg/dL, and 1 hour ago it was 197 mg/dL; the insulin rate must be increased to more rapidly move the patient’s blood sugar toward the targeted glucose range (i.e., 140 to 180 mg/dL, although this range will vary by individual hospital protocol). The important point to emphasize is that the rate of change of the blood glucose is as important as the most recent blood glucose measurement.11 Each of the patients described in the examples may have the same insulin infusion rate, depending on their catabolic state, but individualization among patients with different diagnoses can be safely achieved as long as the rate of change is also considered. The transition from a continuous insulin infusion to intermittent insulin coverage must be handled with care to avoid large fluctuations in blood glucose levels. Before the conversion, the regular insulin infusion should be at a stable and preferably low rate, and the patient’s blood glucose level should be maintained consistently within the target range. The transition from IV to subcutaneous administration depends on numerous factors, including whether the patient is able to eat a consistent amount of dietary carbohydrate.12 Clinicians use various methods to calculate the quantity of insulin to prescribe during the transition from IV to subcutaneous insulin to maintain stable blood glucose levels. Figure 33-1 depicts hypothetical examples of how a combination of basal and bolus insulin regimens (prandial insulin) can work in clinical practice. The following paragraphs describe the application of one calculation method for a 67-year-old patient, Alice Smith, who is recovering from critical illness and has recently been weaned from the ventilator and extubated. 1. Ms. Smith is in stable condition on a regular insulin drip at 1 unit per hour. She is ready to be transitioned to subcutaneous insulin and will be taking food and liquids by mouth. Her total insulin requirement over the previous 24 hours was 32 units. Ms. Smith will now require basal coverage (provided by subcutaneous intermediate or long-acting insulin) and prandial coverage for mealtimes (provided by short-acting subcutaneous insulin). 2. The 30 units of insulin infused during the previous 24 hours is Ms. Smith’s required daily insulin dose. To transition to subcutaneous insulin, a proportion of this amount (i.e., 75% to 80%) will be divided between basal and prandial components.2 In this situation, 75% of the 32 units = 24 units. Half of this amount (12 units) will be administered subcutaneously as intermediate or long-acting insulin; the other half will be administered as short-acting insulin to coincide with meals (i.e., 4 units with each of three meals). 3. The options for insulin administration for Ms. Smith are as follows: Basal insulin: 12 units of glargine once daily, or 6 units twice daily of Neutral Protamine Hagedorn (NPH) administered subcutaneously Prandial insulin: 4 units regular insulin given subcutaneously before each meal (short-acting), or 4 units Lispro or Aspart given subcutaneously with each meal (ultra–short-acting insulin); verify current blood glucose level. Supplemental corrective insulin: A supplemental correction scale can be used to cover any hyperglycemia above the target range, and administration can be combined with scheduled blood glucose measurements; verify current blood glucose level. Table 33-2 describes the various types of insulin available for use. These include ultra–short-acting, short-acting, intermediate-acting, long-acting, and combination insulin replacement options.13 Even after the transition to subcutaneous insulin is completed, blood glucose is monitored frequently to maintain blood glucose within the target range and detect hyperglycemia or hypoglycemia.14 TABLE 33-2 PHARMACOLOGIC MANAGEMENT IV, Intravenous; SQ, subcutaneous. *Dosages are individualized according to patient’s age and size. A patient may be prescribed supplemental or corrective doses of insulin in addition to the basal/prandial insulin combination. The use of the trio of basal, prandial, and corrective insulin is designed to eliminate the use of the traditional sliding scale. Criticisms of the sliding scale method are that the dosages are rarely re-evaluated or adjusted once established and that the scales treat hyperglycemia only after it has occurred; they are not proactive in the manner of the basal/bolus/corrective insulin method.11 It is important to have a protocol for the management of hypoglycemia. The major drawback to use of intensive insulin protocols, as described earlier, is the potential for hypoglycemia.14 Whenever hypoglycemia is detected, it is important to stop any continuous infusion of insulin. An example of one protocol to reverse hypoglycemia follows: • Blood glucose level lower than 40 mg/dL (severe hypoglycemia): administer 50 mL dextrose (50%) in water (D50W) as an IV bolus. • Blood glucose level between 40 and 70 mg/dL (hypoglycemia): administer 25 mL of D50W as an IV bolus. Nursing management of the patient with neuroendocrine stress resulting from critical illness incorporates a variety of nursing diagnoses (Box 33-1). The goals of nursing management are to monitor the hyperglycemic side effects of vasopressor therapy; administer prescribed corticosteroids; monitor blood glucose and insulin effectiveness; avoid hypoglycemia; provide nutrition; and provide education to the patient’s family and supportive others (Box 33-2).
Endocrine Disorders and Therapeutic Management
Neuroendocrinology of Stress and Critical Illness
GLAND OR ORGAN
HORMONE
RESPONSE OR PHYSICAL EXAMINATION
Adrenal cortex
Cortisol
↑ Insulin resistance → ↑ glycogenolysis → ↑ glucose circulation
↑ Hepatic gluconeogenesis → ↑ glucose available
↑ Lipolysis
↑ Protein catabolism
↑ Sodium → ↑ water retention to maintain plasma osmolality by movement of extravascular fluid into the intravascular space
↓ Connective tissue fibroblasts → poor wound healing
Glucocorticoid
↓ Histamine release → suppression of immune system
↓ Lymphocytes, monocytes, eosinophils, basophils
↑ Polymorphonuclear leukocytes → ↑ infection risk
↑ Glucose
↓ Gastric acid secretion
Mineralocorticoids
↑ Aldosterone → ↓ sodium excretion → ↓ water excretion → ↑ intravascular volume
↑ Potassium excretion → hypokalemia
↑ Hydrogen ion excretion → metabolic acidosis
Adrenal medulla
Epinephrine
↑ Endorphins → ↓ pain
Norepinephrine, epinephrine
↑ Metabolic rate to accommodate stress response
↑ Live glycogenolysis → ↑ glucose
↑ Insulin (cells are insulin resistant)
↑ Cardiac contractility
↑ Cardiac output
↑ Dilation of coronary arteries
↑ Blood pressure
↑ Heart rate
↑ Bronchodilation → ↑ respirations
↑ Perfusion to heart, brain, lungs, liver, and muscle
↓ Perfusion to periphery of body
↓ Peristalsis
Norepinephrine
↑ Peripheral vasoconstriction
↑ Blood pressure
↑ Sodium retention
↑ Potassium excretion
Pituitary
All hormones
↑ Endogenous opioids → ↓ pain
Anterior pituitary
Corticotropin
↑ Aldosterone → ↓ sodium excretion → ↓ water excretion → ↑ intravascular volume
↑ Cortisol → ↑ blood volume
Growth hormones
↑ Protein anabolism of amino acids to protein
↑ Lipolysis → ↑ gluconeogenesis
Posterior pituitary
Antidiuretic hormone
↑ Vasoconstriction
↑ Water retention → restoration of circulating blood volume
↓ Urine output
↑ Hypo-osmolality
Pancreas
Insulin
↑ Insulin resistance → hyperglycemia
Glucagon
↑ Glycolysis (directly opposes action of insulin)
↑ Glucose for fuel
↑ Glycogenolysis
↑ Gluconeogenesis
↑ Lipolysis
Thyroid
Thyroxine
↓ Routine metabolic demands during stress
Gonads
Sex hormones
Energy and oxygen supply diverted to brain, heart, muscles, and liver
Acute Neuroendocrine Response to Critical Illness
Hypothalamic-Pituitary-Adrenal Axis in Critical Illness
Cosyntropin Stimulation Test
Corticosteroid Replacement
Liver and Pancreas in Critical Illness
Hyperglycemia in Critical Illness
Clinical Practice Guidelines Related to Blood Glucose Management in Critically Ill Patients
Insulin Management in the Critically Ill
Frequent Blood Glucose Monitoring
Continuous Insulin Infusion
Transition from Continuous to Intermittent Insulin Coverage
Insulin*
INSULIN
ROUTE†
ACTION
ONSET/PEAK/DURATION
SPECIAL CONSIDERATIONS
Ultra–Short-Acting Insulins
Aspart (NovoLog)
SQ
Insulin replacement, rapid onset
5-15 min/30-90 min/<5 hr
Insulin analogue almost immediately absorbed; must be taken with food.
Insulin appearance should be clear.
Must be used in combination with intermediate-acting or long-acting basal insulin regimen. See Fig. 33-1.
Lispro (Humalog)
SQ
Insulin replacement, rapid onset
5-15 min/30-90 min/<5 hr
Insulin analogue; almost immediately absorbed; must be taken with food.
Shorter duration of action than regular insulin; should be used with basal longer-acting insulin. See Fig. 33-1.
Glulisine (Apidra)
SQ
Insulin replacement, rapid onset
5-15 min/30-90 min/<5 hr
Insulin analog
Short-Acting Insulin
Regular
IV or SQ
Insulin replacement therapy
IV: <15 min
SQ: 30-60 min/2-3 hr/5-8 hr
Only type of insulin suitable for IV continuous infusion or IV bolus administration
Intermediate-Acting Basal Insulin
Neutral Protamine
Hagedorn
(NPH)
SQ
Insulin replacement, intermediate action
2-4 hr/4-10 hr/10-16 hr
NPH is not recommended for SQ basal insulin because it has a peak with a less predictable time course than the long-acting insulin analogs.
Long-Acting Basal Insulins
Glargine (Lantus)
SQ
Long-acting basal insulin analogue;
2-4 hr until steady state/no peak
Concentration relatively constant over 20-24 hours
Synthetic insulin (analogue); differs from human insulin by three amino acids, slow release over 24 hours; no peak.
Decrease dose by 20% if switching from NPH to glargine.
Must not be diluted or mixed with other insulins. See Fig. 33-1.
Detemir (Levemir)
SQ
Long-acting basal insulin analogue
3-8 hr until steady state/no peak/5-23 hr
Combination (Premixed) Insulins
Various
SQ
Rapid plus intermediate or long-acting insulin combination
Varies according to combination used
Many combinations exist; examples (long-acting component/short-acting component) include 70/30 regular (70% NPH with 30% regular), NovoLog mix 70/30 (70% aspart-protamine suspension with 30% aspart), and Humalog mix 75/25 (75% lispro-protamine suspension with 25% lispro).
Corrective Insulin Coverage
Hypoglycemia Management
Nursing Management
Endocrine Disorders and Therapeutic Management
Get Clinical Tree app for offline access