Chapter 32 Care of Patients with Noninfectious Lower Respiratory Problems
Safe and Effective Care Environment
1. Ensure safe oxygen delivery.
2. Ensure the proper oxygen flow rate for patients with hypercarbia.
3. Use appropriate infection control methods to protect the patient with cystic fibrosis from respiratory infections.
4. Ensure appropriate functioning of the chest tube drainage system after a thoracotomy.
Health Promotion and Maintenance
5. Use assessment information to identify people at increased genetic risk for a respiratory disease.
6. Encourage everyone to not smoke or to quit smoking.
7. Encourage all people who are exposed to inhalation irritants in the workplace or at home to use appropriate protection.
8. Teach patients with asthma or chronic obstructive pulmonary disease (COPD) how to use a peak flowmeter.
9. Teach patients using any type of inhaler for drug delivery the correct way to use these devices.
10. Teach patients who are using controller (preventive) drug therapy for asthma the importance of taking the prescribed drugs daily, even when asthma symptoms are not present.
11. Teach patients with asthma to have their reliever (rescue) inhalers with them at all times.
12. Encourage the patient and family to express feelings and concerns about a change in breathing status.
13. Teach the patient with activity limitations from respiratory problems how to modify techniques and conserve energy to perform ADLs and desired activities independently.
14. Compare the pathophysiology and clinical manifestations of asthma, bronchitis, and emphysema.
15. Identify risk factors for COPD and lung cancer.
16. Use laboratory data and clinical manifestations to determine the effectiveness of therapy for impaired oxygenation in a patient with breathing problems.
17. Interpret peak expiratory flow (PEF) readings for the need for intervention.
18. Coordinate care for the patient immediately following lung volume reduction surgery.
19. Compare the side effects of radiation treatment for lung cancer with those of chemotherapy for lung cancer.
20. Coordinate nursing interventions for the patient with chest tubes.
http://evolve.elsevier.com/Iggy/
Answer Key for Decision-Making Challenges and NCLEX Examination Challenges
Audio Clip: High- and Low-Pitched Wheezes
Audio Clip: Pleural Friction Rub
Concept Map: Respiratory Acidosis (COPD-Related)
Review Questions for the NCLEX® Examination
Direct gas exchange occurs in the alveoli and the smallest airways. Any problem of these tissues reduces gas exchange and interferes with oxygenation. Many lower airway problems are chronic and progressive, requiring changes in a person’s lifestyle. The older patient with a lower airway problem may need special help before the disorder becomes severe because of age-related changes in breathing effectiveness. Chart 32-1 lists nursing issues for the older patient with a respiratory problem.
Chart 32-1 Nursing Focus on the Older Adult
Chronic Respiratory Disorder
• Provide rest periods between such activities as bathing, meals, and ambulation.
• Place the patient in an upright position for meals to prevent aspiration.
• Encourage nutritional fluid intake after the meal to promote increased calorie intake.
• Schedule drugs around routine activities to increase adherence to drug therapy.
• Arrange chairs in strategic locations to allow the patient with dyspnea to stop and rest while walking.
• Urge the patient to notify the health care provider promptly for any manifestation of infection.
• Encourage the patient to receive the pneumococcal vaccine and to have an annual influenza vaccination.
Chronic Airflow Limitation
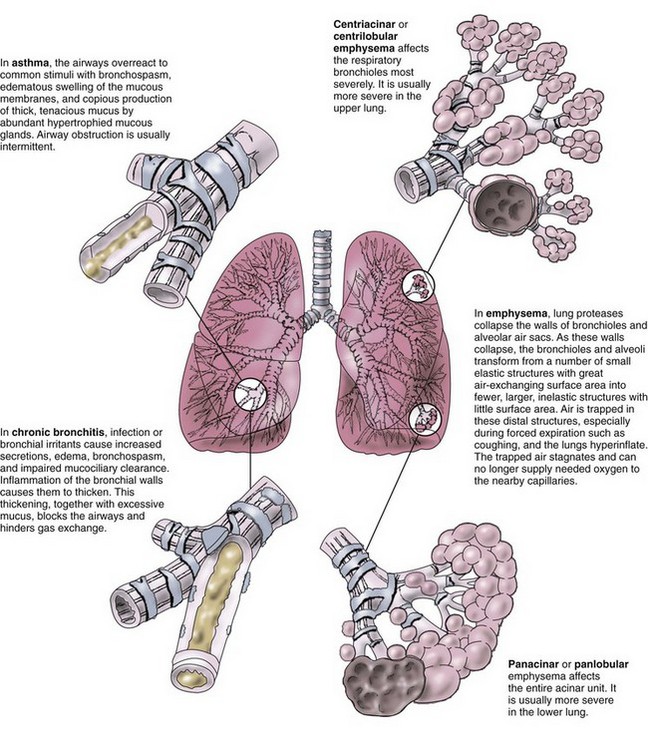
Chronic airflow limitation (CAL) is a group of chronic lung diseases that includes asthma, chronic bronchitis, and pulmonary emphysema. Emphysema and chronic bronchitis, termed chronic obstructive pulmonary disease (COPD), are characterized by bronchospasm and dyspnea (Fig. 32-1). The tissue damage is not reversible and increases in severity over time, eventually leading to respiratory failure. Asthma, unlike COPD, is a chronic disease with intermittent reversible airflow obstruction and wheezing.
More than 40 million Americans suffer from some form of CAL, and 1 million people between the ages of 40 and 65 years have moderate to severe disability from CAL (Centers for Disease Control and Prevention [CDC], 2010). Although some of the problems are not reversible, good management strategies can help maintain adequate oxygenation and improve overall health.
Asthma
Pathophysiology
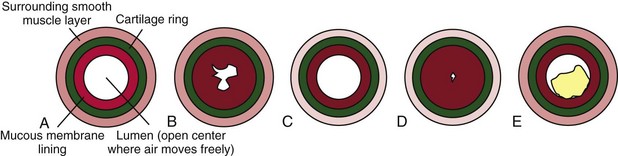
Asthma is usually a chronic condition in which reversible airflow obstruction in the airways occurs intermittently (see Fig. 32-1). Airway obstruction can occur in two ways: (1) inflammation and (2) airway hyperresponsiveness that leads to bronchoconstriction. Inflammation obstructs the lumen (i.e., the inside) of airways (Fig. 32-2). Airway hyperresponsiveness and constriction of bronchial smooth muscle cause a narrowing of the airway from the outside. Airway inflammation can trigger bronchiolar hyperresponsiveness, and many people with asthma have both problems at the same time. Severe airway obstruction can be fatal. More than 3400 deaths from acute asthma occur in the United States each year (CDC, 2010).
Etiology and Genetic Risk
Genetic/Genomic Considerations
Results of genome-wide association studies (GWAS) indicate more than 50 gene variations are associated with asthma from inflammation or hyperresponsive airways, although asthma is a multifactorial disorder with both genetic and environmental input required for expression (Kumar et al., 2010; Online Mendelian Inheritance in Man [OMIM], 2010; Weiss et al., 2009). Some of these variations have greater influence than others, especially within certain racial or ethnic groups. In addition, genetic variation in the gene that controls the synthesis and activity of beta-adrenergic receptors has an impact on drug therapy for asthma. Patients who have a mutation in this gene do not respond as expected to short-acting or long-acting beta agonists and need to have an altered therapy plan. Teaching these patients about why their drug therapies are different from standard recommendations is a nursing responsibility that can assist with therapy adherence.
When asthma is well controlled, the airway changes are temporary and reversible. With poor control, chronic inflammation can lead to damage and hyperplasia of the bronchial epithelial cells and of the bronchial smooth muscle (Global Initiative for Asthma [GINA], 2010). When asthma attacks are frequent, even exposure to low levels of the triggering agent or event may stimulate an attack.
Inflammation triggers asthma for some people when allergens bind to specific antibody molecules (especially immunoglobulin E [IgE]). These molecules are attached to tissue cells called mast cells and white blood cells (WBCs) called basophils. These cells are filled with granules containing chemical mediators that can start local inflammatory responses (see Chapters 19 and 22). Some mediators, such as histamine, start an immediate inflammatory response, which can be blocked by drugs like diphenhydramine (Benadryl). Others, such as leukotriene and eotaxin, are slower and cause later, prolonged inflammatory responses, which can be blocked by drugs like montelukast (Singulair), zafirlukast (Accolate), and zileuton (Zyflo). Mediators also attract more WBCs (eosinophils, macrophages, basophils) to the area, which then release even more inflammation-inducing mediators. Inflammation of airway mucous membranes causes blood vessel dilation and capillary leak, leading to tissue swelling with increased secretions and mucus production (McCance et al., 2010). Inflammation can also occur through general irritation rather than allergic responses.
Gastroesophogeal reflux disease (GERD) is now thought to be a major trigger for asthma in some people, especially in those who have more asthma manifestations at night (GINA, 2010). It does not cause the airway hyperresponsiveness, but the highly acidic stomach contents appear to make the hyperresponsiveness worse when these materials enter the airway.
Incidence/Prevalence
Asthma can occur at any age. About half of adults with asthma also had the disease in childhood. Asthma affects nearly 19 million adults in the United States and Canada (CDC, 2010). It is more common in urban settings than in rural settings.
Considerations for Older Adults
Asthma occurs as a new disorder in about 3% of people older than 55 years. Another 3% of people older than 60 years have asthma as a continuing chronic disorder (CDC, 2010). Lung and airway changes as a part of aging make any breathing problem more serious in the older adult. One problem related to aging is a decrease in the sensitivity of beta-adrenergic receptors. When stimulated, these receptors relax smooth muscle and cause bronchodilation. As these receptors become less sensitive, they no longer respond as quickly or as strongly to agonists (epinephrine, dopamine) and beta-adrenergic drugs, which are often used as reliever (rescue) therapy during an acute asthma attack. Thus teaching older patients how to avoid asthma attacks and to correctly use controller (preventive) drug therapy is a nursing priority.
Patient-Centered Collaborative Care
History
The patient with asthma usually has a pattern of intermittent episodes of dyspnea (shortness of breath), chest tightness, coughing, wheezing, and increased mucus production. Ask whether the manifestations occur continuously, seasonally, in association with specific activities or exposures, or more frequently at night. Some patients have these manifestations for 4 to 8 weeks after a chest cold or other upper respiratory tract infection. The patient with atopic (allergic) asthma often has other allergic problems such as rhinitis, skin rash, or pruritus. Ask whether any family members have asthma or respiratory problems. Ask about current or previous smoking habits. If the patient smokes, use this opportunity to teach him or her about smoking cessation (Chart 32-4). Wheezing in nonsmokers is an important symptom in the diagnosis of asthma.
Chart 32-4 Patient And Family Education
Preparing For Self-Management: Smoking Cessation
• Make a list of the reasons you want to stop smoking (e.g., your health and the health of those around you, saving money, social reasons).
• Set a date to stop smoking, and keep it. Decide whether you are going to begin to cut down on the amount you smoke or are going to stop “cold turkey.” Whatever way you decide to do it, keep this important date!
• Ask for help from those around you. Find someone who wants to quit smoking and “buddy up” for support. Look for assistance in your community, such as formal smoking-cessation programs, counselors, and certified acupuncture specialists or hypnotists.
• Consult your health care provider about nicotine replacement therapy (e.g., patch, gum) or other pharmacologic therapy to assist in smoking cessation.
• Remove ashtrays and lighters from your view.
• Talk to yourself! Remind yourself of all the reasons you want to quit.
• Think of a way to reward yourself with the money you save from not smoking for a year.
• Avoid places that might tempt you to smoke. If you are used to having a cigarette after meals, get up from the table as soon as you are finished eating. Think of new things to do at times when you used to smoke (e.g., taking a walk, exercising, calling a friend).
• Find activities that keep your hands busy: needlework, painting, gardening, even holding a pencil.
• Take five deep breaths of clean, fresh air through your nose and out your mouth if you feel the urge to smoke.
• Keep plenty of healthy, low-calorie snacks, such as fruit and vegetables, on hand to nibble on. Try sugarless gum or mints as a substitute for tobacco.
• Drink at least eight glasses of water each day.
• Begin an exercise program with the approval of your health care provider. Be aware of the positive, healthy changes in your body since you stopped smoking.
• List the many reasons why you are glad that you quit. Keep the list handy as a reminder of the positive things you are doing for yourself.
• If you have a cigarette, think about what the conditions were that caused you to light it. Try and think of a strategy to avoid that (or those) conditions.
• Don’t beat yourself up for backsliding; just face the next day as a new day.
• Think of each day without tobacco as a major accomplishment. It is!!
Physical Assessment/Clinical Manifestations
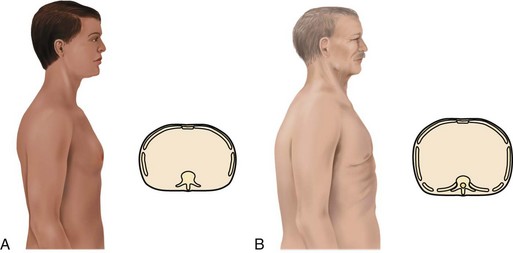
The patient may use accessory muscles to help breathe during an attack. Observe for muscle retraction at the sternum and the suprasternal notch and between the ribs. The patient with long-standing, severe asthma may have a “barrel chest,” caused by air trapping (Fig. 32-3). The anteroposterior (AP) diameter (diameter between the front and the back of the chest) increases with air trapping, giving the chest a rounded rather than an oval shape. The normal chest is about 1.5 times as wide as it is thick. In the patient with severe, chronic asthma, the AP diameter may equal or exceed the lateral diameter. Compare the AP diameter of the chest with the lateral diameter. Chronic air trapping also flattens the diaphragm and increases the space between the ribs.
Laboratory Assessment
Laboratory tests can help determine the type of asthma and the degree of breathing impairment. Arterial blood gas (ABG) levels show how well the patient is obtaining oxygen (see Chapter 14 for discussion of ABGs). The arterial oxygen level (PaO2) may decrease during an asthma attack. Early in the attack, the arterial carbon dioxide level (PaCO2) may be decreased as the patient increases respiratory effort. Later in an asthma episode, PaCO2 rises, indicating carbon dioxide retention. Allergic asthma often occurs with an elevated serum eosinophil count and immunoglobulin E levels. The sputum may contain eosinophils and mucus plugs with shed epithelial cells (Curschmann’s spirals).
Pulmonary Function Tests
• Forced vital capacity (FVC) (volume of air exhaled from full inhalation to full exhalation)
• Forced expiratory volume in the first second (FEV1) (volume of air blown out as hard and fast as possible during the first second of the most forceful exhalation after the greatest full inhalation)
• Peak expiratory flow (PEF) (fastest airflow rate reached at any time during exhalation)
Interventions
The purposes of asthma therapy are to control and prevent episodes, improve airflow, and relieve symptoms. Adult asthma is best managed when the patient is an active partner in the management plan. Priority nursing actions focus on patient education about proper implementation of a personal asthma action plan, which includes drug therapy and lifestyle management strategies to assist the patient in understanding his or her disease and its treatment (GINA, 2010).
Teaching for Self-Management
Asthma often has intermittent overt symptoms. With guided self-care, patients can co-manage this disease, increasing symptom-free periods and decreasing the number and severity of attacks (Ellis, 2008; Pruitt, 2011). Good management decreases the number of hospital admissions and increases participation in patient-chosen work and leisure activities. Self-care requires extensive education for the patient to be able to self-assess respiratory status, self-treat (including adjusting the frequency and dosage of prescribed drugs), and know when to consult the health care provider.
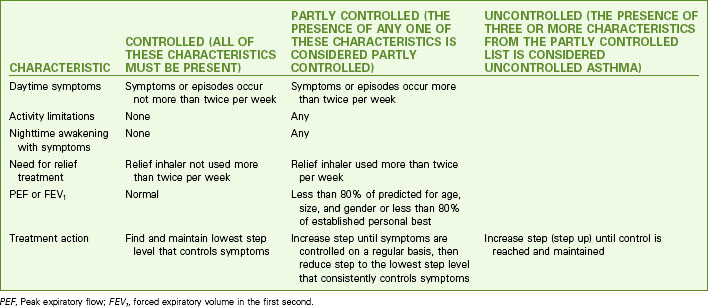
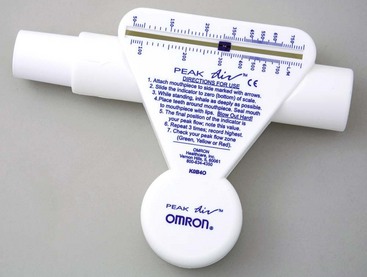
Teach the patient to assess symptom severity at least twice daily with a peak flowmeter (Fig. 32-4) and to adjust drugs according to his or her personal asthma action plan to manage inflammation and bronchospasms to prevent or relieve symptoms. Chart 32-5 describes the correct method to use the meter. Teach the patient to first establish a baseline or “personal best” peak expiratory flow (PEF) by measuring his or her PEF twice daily for 2 to 3 weeks when asthma is well controlled and recording the results. This way, the patient will know when his or her peak flow is reduced to the point that more drugs are needed or that emergency assistance is needed. When the patient has established a “personal best,” all other readings are compared with this value in terms of percent of personal best. Some meters are color-coded to help the patient interpret the results. Green zone readings are at least 80% or above the “personal best.” This is the ideal range for asthma control and indicates that no increases in drug therapy are needed. Yellow is a range between 50% and 80% of personal best. When a patient has a reading in this range, he or she needs to use the prescribed “reliever drug” (formerly called a rescue drug). Within a few minutes after using the reliever drug, another PEF reading should be made to determine whether the reliever drug is working. Frequent readings in the yellow zone or increasing use of reliever drugs is a warning that indicates the need to reassess the asthma plan for a temporary or permanent change in controller (preventive) drugs. Red is a range below 50% of the patient’s personal best and indicates serious respiratory obstruction.
Chart 32-5 Patient And Family Education
Preparing For Self-Management: Using a Peak Flowmeter
• Set the peak flowmeter at zero.
• Use a standing position, without leaning or supporting yourself on anything, if possible.
• Take as deep a breath as you can.
• Place the mouthpiece of the meter in your mouth, taking care to wrap your lips tightly around it.
• Blow your breath out through the mouthpiece as hard and as fast as you are able. (If you cough, sneeze, or have any type of interruption while you exhale, reset the meter and perform the test again.)
• Reset and perform the test two additional times.
• Use the highest reading of the three to determine your current peak flow rate.
• Keep a record or graph of your peak flow rates and examine these for trends.
Action Alert
Teach the patient to keep a symptom and intervention diary to learn specific triggers of asthma symptoms, early cues for impending attacks, and personal response to drugs. Stress the importance of proper use of his or her personal asthma action plan for any severity of asthma (Corbridge & Corbridge, 2010). Chart 32-6 lists areas to emphasize when teaching the patient with asthma.
Chart 32-6 Patient And Family Education
Preparing For Self-Management: Asthma Management
• Avoid potential environmental asthma triggers, such as smoke, fireplaces, dust, mold, and weather changes (especially warm to cold or sudden barometric changes).
• Avoid medications that are triggers for your asthma (e.g., aspirin, NSAIDs, beta blockers).
• Avoid food that has been prepared with monosodium glutamate (MSG) or metabisulfite.
• If you experience symptoms of exercise-induced asthma, use your bronchodilator inhaler 30 minutes before exercise to prevent or reduce bronchospasm.
• Be sure you know the proper technique and correct sequence when you use metered dose inhalers.
• Get adequate rest and sleep.
• Reduce stress and anxiety; learn relaxation techniques; adopt coping mechanisms that have worked for you in the past.
• Wash all bedding with hot water to destroy dust mites.
• Monitor your peak expiratory flow rates with a flowmeter at least twice daily.
• Seek immediate emergency care if you experience any of these:
Drug Therapy
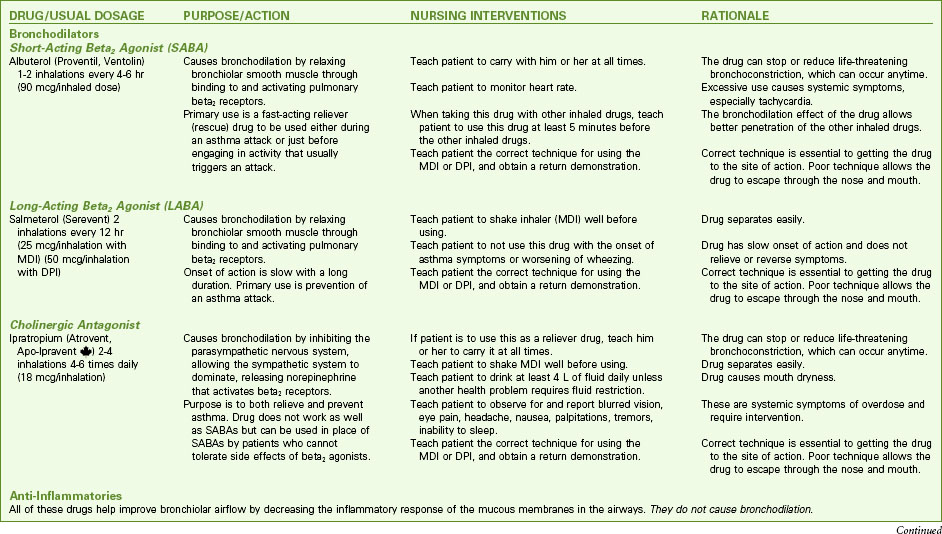
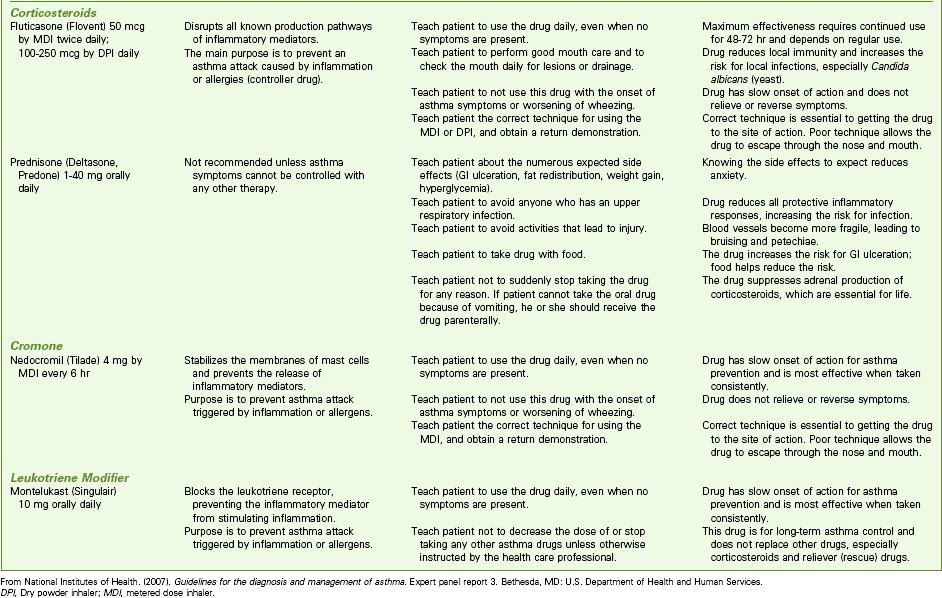
Pharmacologic management of adult patients with asthma is based on the step category for severity and treatment (see Charts 32-2 and 32-3) (GINA, 2010). Control therapy drugs (formerly called preventive drugs) are drugs used to reduce airway responsiveness to prevent asthma attacks from occurring. They are used every day, regardless of symptoms. Reliever drugs are those used to actually stop an attack once it has started. Some patients may need drug therapy only during an asthma episode. For others, daily drugs are needed to keep asthma episodic rather than a more frequent problem. This therapy involves the use of bronchodilators and various drug types to reduce inflammation. Some drugs reduce the asthma response, and other drugs actually prevent the response. Combination drugs are two agents from different classes combined together for better response. Chart 32-7 lists the most common preferred drugs in each class for control (prevention) and relief (rescue) therapy of asthma. The actions, interventions, and rationales for most drugs within a single class are similar although drug dosages may differ. Be sure to consult a pharmacology text or drug handbook for more information on a specific drug.
Bronchodilators
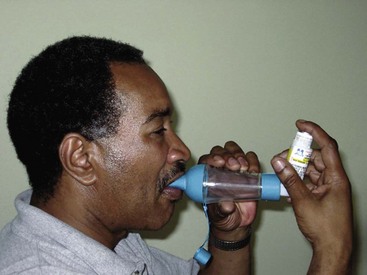
Short-acting beta2 agonists (SABAs) provide rapid but short-term relief. These inhaled drugs are most useful when an attack begins (as relief) or as premedication when the patient is about to begin an activity that is likely to induce an attack (GINA, 2010). Such agents include albuterol (Proventil, Ventolin), bitolterol (Tornalate), levalbuterol (Xopenex), pirbuterol (Maxair), and terbutaline (Brethaire). When inhaled from either a metered dose inhaler (MDI) or a dry powder inhaler (DPI), the drug is delivered directly to the site of action and systemic effects are minimal (unless the agent is overused). Teach the patient the correct technique for using an inhaled drug to achieve the greatest benefit. Chart 32-8 describes the proper way to use an MDI. Fig. 32-5 shows a patient using a “spacer” with an MDI. Spacer use increases the amount of drug delivered to the lungs. Chart 32-9 describes the proper care and use of a DPI. Fig. 32-6 shows a patient using a DPI.
Chart 32-8 Patient And Family Education: Preparing For Self-Management: How to Use an Inhaler Correctly*
With a Spacer (Preferred Technique)
1. Before each use, remove the caps from the inhaler and the spacer.
2. Insert the mouthpiece of the inhaler into the non-mouthpiece end of the spacer.
3. Shake the whole unit vigorously three or four times.
4. Place the mouthpiece into your mouth, over your tongue, and seal your lips tightly around it.
5. Press down firmly on the canister of the inhaler to release one dose of medication into the spacer.
6. Breathe in slowly and deeply. If the spacer makes a whistling sound, you are breathing in too rapidly.
7. Remove the mouthpiece from your mouth, and, keeping your lips closed, hold your breath for at least 10 seconds and then breathe out slowly.
8. Wait at least 1 minute between puffs.
9. Replace the caps on the inhaler and the spacer.
10. At least once a day, clean the plastic case and cap of the inhaler by thoroughly rinsing in warm, running tap water; at least once a week, clean the spacer in the same manner.
Without a Spacer (Primary Method)
1. Before each use, remove the cap and shake the inhaler according to the instructions in the package insert.
2. Tilt your head back slightly, and breathe out fully.
3. Open your mouth, and place the mouthpiece 1 to 2 inches away.
4. As you begin to breathe in deeply through your mouth, press down firmly on the canister of the inhaler to release one dose of medication.
5. Continue to breathe in slowly and deeply (usually over 3-5 sec).
6. Hold your breath for at least 10 seconds to allow the medication to reach deep into the lungs, and then breathe out slowly.
7. Wait at least 1 minute between puffs.
8. Replace the cap on the inhaler.
9. At least once a day, remove the canister and clean the plastic case and cap of the inhaler by thoroughly rinsing in warm, running tap water.
Chart 32-9 Patient And Family Education
Preparing For Self-Management: How to Use a Dry Powder Inhaler (DPI)
After Loading the Drug and for Inhalers That Do Not Require Drug Loading
• Read your doctor’s instructions for how fast you should breathe for your particular inhaler.
• Place your lips over the mouthpiece, and breathe in forcefully (there is no propellant in the inhaler; only your breath pulls the drug in).
• Remove the inhaler from your mouth as soon as you have breathed in.
• Never exhale (breathe out) into your inhaler. Your breath will moisten the powder, causing it to clump and not be delivered accurately.
• Never wash or place the inhaler in water.
• Keep your inhaler in a dry place at room temperature.
• If the inhaler is preloaded, discard the inhaler after it is empty.
• Because the drug is a dry powder and there is no propellant, you may not feel, smell, or taste it as you inhale.
Action Alert
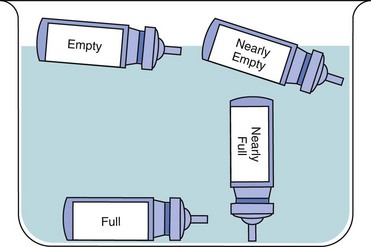
Dry powder inhalers indicate the amount of remaining drug; however, aerosol inhalers (MDIs) do not. Demonstrate how to check approximate aerosol inhaler drug levels by placing the inhaler in water (Fig. 32-7). Full inhalers sink to the bottom. An empty inhaler floats on its side. This technique is controversial because it is only a general approximation. The more recommended technique is for the patient to count the number of doses as they are used; however, many patients have difficulty keeping the dose count accurate.
Long-acting beta2 agonists (LABAs) are also delivered by inhaler directly to the site of action—the bronchioles. Proper use of the long-acting agonists can decrease the need to use reliever (rescue) drugs as often. Unlike short-acting agonists, long-acting drugs need time to build up an effect but the effects are longer lasting. Thus these drugs are useful in preventing an asthma attack but have no value during an acute attack. Therefore teach patients not to use LABAs alone to relieve (rescue) them during an attack or when wheezing is getting worse but, instead, to use a SABA. Relying on LABAs during an attack can lead to worsening of symptoms and death (Aschenbrenner, 2010). Examples of LABAs include formoterol (Foradil) and salmeterol (Serevent). These drugs are never prescribed as the only drug therapy for asthma. Teach the patient to use these control drugs daily as prescribed, even when no symptoms are present.
Health Promotion and Maintenance
A. Instruct the client to use the albuterol (Proventil) inhaler instead.
B. Assist the client to use oxygen for three breaths between the two puffs of the inhaled drug.
C. Instruct the client to attach the spacer to the inhaler before using it and inhale as rapidly as possible.
D. Remind the client to take a deep breath, hold it for 15 seconds, and then exhale before using the inhaler.
Exercise/Activity
Regular exercise is a recommended part of asthma therapy (Holcomb, 2008). Aerobic exercise assists in maintaining cardiac health, enhancing skeletal muscle strength, and promoting ventilation and perfusion. Teach patients with asthma to examine the conditions that trigger an attack and adjust the exercise routine as needed. Some may need to premedicate with inhaled SABAs before beginning activity. For others, adjusting the environment may be needed (e.g., changing from outdoor ice-skating in cold, dry air to indoor ice-skating).
Chronic Obstructive Pulmonary Disease
Pathophysiology
Chronic obstructive pulmonary diseases (COPD) include emphysema and chronic bronchitis. Although these are separate disorders with different pathologic processes, many patients with emphysema also have chronic bronchitis at the same time (Fig. 32-8).
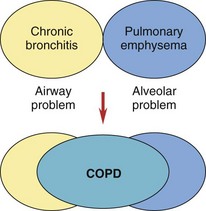
FIG. 32-8 The interaction of chronic bronchitis and emphysema in chronic obstructive pulmonary disease (COPD).
Emphysema
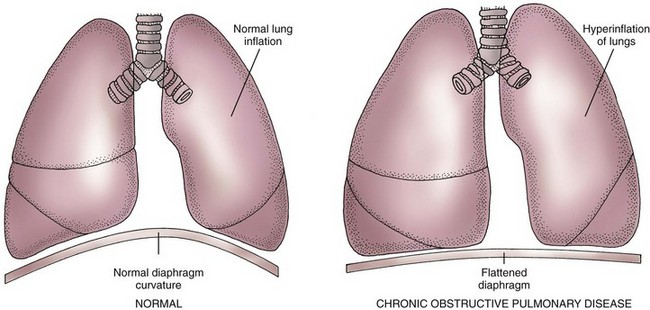
The two major changes that occur with emphysema are loss of lung elasticity and hyperinflation of the lung (see Fig. 32-1). These changes result in dyspnea and the need for an increased respiratory rate.
An increased amount of air is trapped in the lungs. Causes of air trapping are loss of elastic recoil in the alveolar walls, overstretching and enlargement of the alveoli into air-filled spaces called bullae, and collapse of small airways (bronchioles). These changes greatly increase the work of breathing. The hyperinflated lung flattens the diaphragm (Fig. 32-9), weakening the effect of this muscle. As a result, the patient with emphysema needs to use additional muscles (accessory muscles) in the neck, chest wall, and abdomen to inhale and exhale. This increased effort increases the need for oxygen, making the patient work harder and have an “air hunger” sensation. Often, inhalation starts before exhalation is completed, resulting in an uncoordinated pattern of breathing.
Oxygenation is affected by the increased work of breathing and the loss of alveolar tissue. Although some alveoli enlarge, the curves of alveolar walls decrease and less surface area is available for gas exchange. Often the patient adjusts by increasing the respiratory rate, so arterial blood gas (ABG) values may not show gas exchange problems until the patient has advanced disease. Then carbon dioxide is produced faster than it can be eliminated, resulting in carbon dioxide retention and chronic respiratory acidosis (see Chapter 14). The patient with late-stage emphysema also has a low arterial oxygen (PaO2) level, because it is difficult for oxygen to move from diseased alveoli into the blood.
Emphysema is classified as panlobular, centrilobular, or paraseptal depending on the pattern of destruction and dilation of the gas-exchanging units (acini) (see Fig. 32-1). Each type can occur alone or in combination in the same lung. Most are associated with smoking or chronic exposure to other inhalation irritants.
Etiology and Genetic Risk
About 100,000 Americans have severe AAT deficiency, and many more have mild to moderate deficiencies (Nussbaum et al., 2007). Although an AAT deficiency also can cause problems in the skin and liver, lung diseases are more common.
Genetic/Genomic Considerations
The gene for AAT has many known mutations, some of which increase the risk for emphysema. Variation of mutations (polymorphisms) results in different levels of AAT deficiency. This variation is one reason why the disease is more severe for some people than for others. The most serious mutation for an increased risk for emphysema is the Z mutation, although others also increase the risk but to a lesser degree. Table 32-1 shows the most common AAT mutations increasing the risk for emphysema.
TABLE 32-1 CHARACTERISTICS ASSOCIATED WITH THE MOST COMMON ALPHA1-ANTITRYPSIN GENE MUTATIONS
| MUTATION GENOTYPE | LEVEL OF SERUM ALPHA1-ANTITRYPSIN (% OF NORMAL) | DISEASE SEVERITY |
|---|---|---|
| M/S | 80% | No detectable disease |
| S/S | 50%-60% | Minimal to no disease expression |
| M/Z | 50%-55% | Minimal to no disease expression |
| S/Z | 30%-35% | Pulmonary disease, early age |
| Z/Z | 10%-15% | Severe COPD, extra-pulmonary involvement |
COPD, Chronic obstructive pulmonary disease.
From Workman, M.L., & Winkelman, C. (2008). Genetic influences in common respiratory disorders. Critical Care Nursing Clinics of North America, 20(2), 171-189.
Incidence/Prevalence
The prevalence of chronic bronchitis and emphysema in the United States has been estimated at about 13.5 million (for chronic bronchitis) and 2 million (for emphysema). Chronic obstructive pulmonary disease (COPD) is the fourth leading cause of morbidity and mortality in the United States (Global Initiative for Chronic Obstructive Lung Disease [GOLD], 2009).
Complications
Cardiac failure, especially cor pulmonale (right-sided heart failure caused by pulmonary disease), occurs with bronchitis or emphysema. Air trapping, airway collapse, and stiff alveolar walls increase the lung tissue pressure, making blood flow through lung vessels more difficult. The increased pressure creates a heavy workload on the right side of the heart, which pumps blood into the lungs. As the disease progresses, the amount of oxygen in the blood decreases, causing major blood vessels in the lung to constrict. To pump blood through these narrowed vessels, the right side of the heart generates high pressures. In response to this heavy workload, the right chambers of the heart enlarge and thicken, causing right-sided heart failure with backup of blood into the general venous system. Chart 32-10 lists key features of cor pulmonale.
Cor Pulmonale
Health Promotion and Maintenance
The incidence and severity of COPD would be greatly reduced by smoking cessation. COPD is rare among people who have never smoked cigarettes. Disease progression can be slowed by smoking cessation. Urge all people who smoke to quit smoking. Chart 32-4 provides tips to teach people about smoking cessation.
Patient-Centered Collaborative Care
The Concept Map on p. 615 addresses assessment and nursing care issues related to COPD.
Assessment
Physical Assessment/Clinical Manifestations
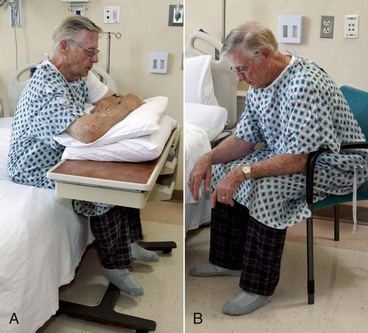
General appearance can provide clues about the patient’s respiratory status and energy level. Observe weight in proportion to height, posture, mobility, muscle mass, and overall hygiene. The patient with increasingly severe COPD is thin, with loss of muscle mass in the extremities, although the neck muscles may be enlarged. He or she tends to be slow moving and slightly stooped. Usually the person sits with a forward-bending posture, sometimes with the arms held forward, a position known as the orthopneic or tripod position (Fig. 32-10). When dyspnea becomes severe, activity intolerance may be so great that bathing and general grooming are neglected.
Assess the degree of dyspnea using an assessment tool called a Visual Analog Dyspnea Scale (VADS). The VADS is a straight line with verbal anchors at the beginning and end of a 100-mm line. Ask the patient to place a mark on the line to indicate his or her perceived breathing difficulty (Fig. 32-11). Document the response, and use this scale to assess dyspnea, determine the therapy effectiveness, and pace the patient’s activities.
Examine the patient’s chest for the presence of a “barrel chest” (see Fig. 32-3). With a barrel chest, the ratio between the anteroposterior (AP) diameter of the chest and its lateral diameter is 2 : 2 rather than the normal ratio of 1 : 1.5. This shape change results from lung overinflation and diaphragm flattening.
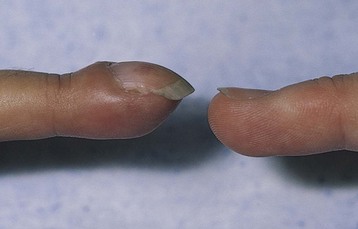
The patient with chronic bronchitis often has a cyanotic, or blue-tinged, dusky appearance and has excessive sputum production. Assess for cyanosis, delayed capillary refill, and finger clubbing (Fig. 32-12), which indicate chronically decreased arterial oxygen levels.
Laboratory Assessment
Arterial blood gas (ABG) values identify abnormal oxygenation, ventilation, and acid-base status. Compare serial or repeated ABG values to assess changes in the patient’s respiratory status. Once baseline ABG values are obtained, pulse oximetry can gauge the response to treatment. As COPD worsens, the amount of oxygen in the blood decreases (hypoxemia) and the amount of carbon dioxide in the blood increases (hypercarbia). Chronic respiratory acidosis (increased arterial carbon dioxide [PaCO2]) then results; metabolic alkalosis (increased arterial bicarbonate) occurs as compensation by kidney retention of bicarbonate. This change is seen on ABGs as an elevation of 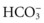 , although pH remains lower than normal. Not all patients with COPD are carbon dioxide retainers, even when hypoxemia is present. Carbon dioxide diffuses more easily across lung membranes than does oxygen. Hypercarbia is a problem in advanced emphysema (because the alveoli are affected) rather than in bronchitis (wherein the airways are affected). For more detailed information about acidosis, see Chapter 14.
, although pH remains lower than normal. Not all patients with COPD are carbon dioxide retainers, even when hypoxemia is present. Carbon dioxide diffuses more easily across lung membranes than does oxygen. Hypercarbia is a problem in advanced emphysema (because the alveoli are affected) rather than in bronchitis (wherein the airways are affected). For more detailed information about acidosis, see Chapter 14.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree