Diabetes insipidus (DI) is a disorder of urinary concentration caused by a temporary or chronic deficiency of or insensitivity to vasopressin, or antidiuretic hormone (ADH). This condition leaves the kidneys (specifically the renal collecting ducts and tubules) unable to conserve water. The result is excretion of large volumes of dilute or hypotonic urine and excessive thirst (polydipsia) (Fig. 9.1). (Arvanitis & Pasquale, 2005; Wong & Verbalis, 2002)
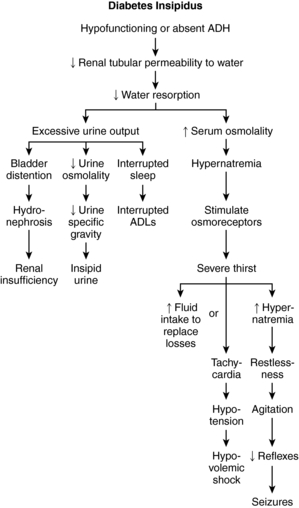 |
| Fig. 9.1Pathophysiology of diabetes.(From Urden L. D., Stacy, K. M., & Lough, M. E. [2006]. Thelan’s critical care nursing: Diagnosis and management. [5th ed.]. St. Louis: Mosby.) |
ADH, which is produced in the supraoptic and paraventricular nuclei of the hypothalamus, controls water balance. The serum water content is measured by serum osmolality (a normal value is 285 to 295 mmol/kg). From the hypothalamus ADH is transported through nerve fibers in neurosecretory granules to the posterior pituitary gland, where it is stored. When released, it travels to the kidneys, where it causes the renal tubules to concentrate the urine and return excess water to the bloodstream. If a person starts to become dehydrated, the anterior pituitary signals the posterior pituitary to release ADH and signals the kidneys to reabsorb free water (Urden et al., 2006;Arvanitis & Pasquale, 2005; Ropper & Brown, 2005; South-Paul et al., 2004; Wong & Verbalis, 2002).
EPIDEMIOLOGY AND ETIOLOGY
DI is an uncommon disorder that affects 3 of 100,000 people in the general population and about 1% of patients suffering from head injury (Arvanitis & Pasquale, 2005). These patients are unable to conserve free water and therefore excrete large volumes of dilute urine (polyuria). They also are excessively thirsty, particularly for ice-cold water (polydipsia), which reflects the body’s attempt to maintain osmolality. Polyuria and polydipsia can lead to dehydration and resulting hypernatremia (Adam, 1997).
DI can be categorized into several types, including central DI (hypothalamic, pituitary, cranial neurogenic, or neurohypophyseal), nephrogenic DI (renal), gestagenic DI (pregnancy), and dipsogenic DI (psychogenic).
Patients who have central DI, the most common type, do not secrete or produce enough ADH in response to osmotic or nonosmotic stimuli, and the result is dilute urine. This usually is caused by damage to the hypothalamus or the posterior pituitary; although a secondary cause may be related to an idiopathic or primary cause (Suarez, 2004; Graham & Lantos, 2002). Primary causes of central DI may include idiopathic or genetic mutations. Secondary causes (most often the cause) may include head trauma, infection, tumors, hypoxic brain tissue injury, and infiltrative systemic disease (see the section, Risk Profile) (Graham & Lantos, 2002; Wong & Verbalis, 2002). Patients affected with central DI generally respond well to medical treatment involving the administration of desmopressin or exogenous ADH and fluid replacement.
Nephrogenic DI (NDI) occurs as a result of renal unresponsiveness to ADH. This impairs the kidneys’ ability to concentrate urine and reabsorb water. Most water reabsorption occurs in the collecting ducts at the V2 receptors; however this process continues with the transfer and transport of sodium chloride in the loop of Henle. In nephrogenic DI, ADH does not increase water permeability or concentrate urine. The cause may be underlying kidney disease or possibly a drug-related reaction. Patients with nephrogenic DI have normal ADH levels and therefore do not respond to desmopressin or exogenous ADH therapy. They are best treated with therapies that target the underlying condition (Greenspan & Gardner, 2004; Wong & Verbalis, 2002).
Gestagenic DI is associated with a pregnancy that is complicated by pre-eclampsia or hepatic dysfunction. It develops as a result of increased levels of vasopressinase, an enzyme that destroys circulating endogenous vasopressin. The enzyme does not affect synthetic vasopressin, therefore these patients respond well to treatment with desmopressin. The condition usually resolves within 10 days after delivery (Fitzgerald, 2006). However, the mother then has a higher risk of developing diabetes.
Dipsogenic DI is a condition seen in the psychiatric population as a result of an excessive intake of water (usually more than 20 L of fluid per day). This type of DI may be due to illness or to side effects (e.g., dry mouth) caused by anticholinergic or antipsychotic medications (Arvanitis & Pasquale, 2005; Adam, 1997).
RISK PROFILE
Oncology: Primary tumors include glioma, hypothalamic hamartoma, hypothalamic tumors, germinoma, meningioma, lymphoma, craniopharyngioma, granular cell tumor (choristoma), large chromophobe adenomas, pinealoma, Rathke’s cleft cyst, massive pituitary tumors that invade the pituitary stalk, and multiple myeloma. Metastatic pituitary lesions and metastatic tumors originating in the lung or breast, leukemic infiltration, and a history of brain irradiation put a patient at risk of developing DI (Ropper & Brown, 2005; Vance, 2003; Wong & Verbalis, 2002). Table 9-1 lists the causes of central and nephrogenic DI.
| Causes of Central DI | Causes of Nephrogenic DI |
|---|---|
Idiopathic Familial causes • Wolfram syndrome, or DIDMOAD (DI + DM + optic atrophy + deafness); rare genetic autosomal recessive mutation of WFS1 gene encoding Trauma • Head injury, especially basilar skull fracture Autoimmune causes Causes in surgical patients • Brain tumors (especially pituitary adenomas and metastatic pituitary lesions) • Intracerebral hemorrhage • Ruptured aneurysm Infection • Meningitis • Tuberculosis • Syphilis • Encephalitis Anoxic encephalopathy Ischemia • Shock • Cardiac arrest • Sheehan’s syndrome • Sickle cell disease Granulomatous causes • Neurosarcoidosis • Langerhans’ cell histiocytosis • Letterer-Siwe disease • Hand-Schüller-Christian disease | Familial causes • X-linked nephrogenic DI (most common in males) Renal disease • Polycystic kidney disease • Chronic pyelonephritis • Medullary cystic disease • Sickle cell nephropathy • Sarcoidosis • Chronic renal failure • Multiple myeloma • Analgesics • Sjögren’s syndrome Medications • Lithium • Demeclocycline • Methoxyflurane anesthesia • Foscavir • Cidofovir • Aminoglycosides • Vinblastine • Rifampin • Valproic acid Metabolic causes • Chronic hypokalemia • Chronic hypocalcemia • Protein starvation |
Genetics: Nearly 90% of patients with congenital NDI are males with X-linked recessive NDI, with mutations of the AVPR2 gene (Bichet, 2006). Other mutations include the AQP2 gene.
PROGNOSIS
Although the outcome depends on the underlying pathologic condition, DI, when treated, does not cause severe problems or reduce life expectancy. Life expectancy largely depends on the underlying cause. Acute DI can be treated in the hospital with medication, fluid and electrolyte replacement, and treatment of the underlying cause, a process that may take days to weeks. Patients with chronic DI are treated on an outpatient basis for control of symptoms and for monitoring of serum sodium levels, other electrolytes, and fluid status (Fitzgerald, 2006).
PROFESSIONAL ASSESSMENT CRITERIA (PAC)
The diagnosis of DI should be considered in patients who have large volumes of urine output, excessive thirst, and nocturia. Excessive thirst and nocturia are found only in responsive patients; therefore serum laboratory values, as well as intake and output, must be closely monitored in the high-risk unresponsive patient. Neurologic signs may include a change in the level of consciousness, restlessness, weakness, confusion and, in severe cases, convulsion caused by sodium and other electrolyte imbalances. Responsive patients may complain of a dry tongue and mouth. Cardiovascular signs are consistent with dehydration and may include hypotension, tachycardia, azotemia, and increased temperature as a result of fluid volume loss and disruption of the hypothalamus. Patients who are dehydrated may have poor skin turgor, sunken eyes, dry mucous membranes, fatigue, lethargy, headache, and irritability. Nocturia and polyuria (urine output greater than 30 mL/kg/day) are classic signs of DI, along with polydipsia (fluid ingestion ranging from 2 to 20 L/day) (Fitzgerald, 2006; Ropper & Brown, 2005; Barkley & Myers, 2001).
Diagnostic findings
1. Urine hyposmolarity (less than 150 to 200 mOsm/L) as a result of dilute urine
2. Serum hyperosmolarity (greater than 300 mOsm/L) as a result of dehydration
3. Low specific gravity (1.001 to 1.005) as a result of decreased urine concentration
4. Sodium level normal or above normal (greater than 145 mEq/L) as a result of dehydration
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


Get Clinical Tree app for offline access
