Postpartum Complications
Objectives
2. Summarize major causes of postpartum hemorrhage.
3. Identify nursing interventions in the care of the woman with postpartum hemorrhage.
4. Describe the dangers that deep vein thrombosis presents.
5. Explain the nursing care of a woman who has a thromboembolism.
5. List four common sites for puerperal infection.
6. Describe predisposing factors for infections of the reproductive system.
7. Discuss the nursing care of a woman who has an infected episiotomy.
Key Terms
endometritis (e˘n-dō-me˘ -TRĪ-tĭs, p. 350)
mastitis (măs-TĪ-tĭs, p. 351)
parametritis (păr-ăme˘ -TRĪ-tĭs, p. 350)
placenta accreta (plăSĔ N-tă ăKRĒ-tă, p. 345)
postpartum hemorrhage (HĔ M-ŏr-ĭj, p. 343)
puerperal fever (pū-Ĕ R-pĕ r-ăl, p. 350)
pulmonary embolism (PŬL-mō-nărē Ĕ M-bō-lĭz-ŭm, p. 349)
REEDA (p. 350)
subinvolution (sŭb-ĭn-vō-LŪ-shŭn, p. 345)
thrombophlebitis (thrŏm-bō-fle˘ BĪ-tŭs, p. 349)
uterine atony (Ū-te˘ r-ĭn ĂT-ŏnē, p. 344)
 http://evolve.elsevier.com/Leifer/maternity
http://evolve.elsevier.com/Leifer/maternity
Shortened inpatient (postpartum) stays are common in maternity care. Women are often discharged after childbirth before clinical signs of puerperal infection and other postpartum disorders are evident. Consequently, hospital-based nurses are challenged to perform a risk assessment and attempt to recognize subtle signs of complications that may require a delay in discharge. Before discharge, the nurse teaches preventive measures to avoid common postpartum complications.
Many problems can occur during the postpartum period, but most problems fall into the following five categories:
Postpartum Hemorrhage: Overview
Postpartum hemorrhage is the most common cause of excessive bleeding during the childbearing cycle. Postpartum hemorrhage is traditionally defined as loss of more than 500 mL of blood after an uncomplicated vaginal birth or 1000 mL after a cesarean birth. Excessive blood loss after a complicated birth, such as placenta previa (see Chapter 13) or accreta placenta, is discussed later in this chapter. Because most women have 1 to 2 L of increased blood volume during pregnancy, they can tolerate this amount of blood loss. Postpartum hemorrhage can occur early (in the first 24 hours) or late (between 24 hours and 6 weeks after birth). The greatest danger, however, is in the first 24 hours because of the large venous area exposed after placental separation from the uterine wall (Box 17-1).
The most common causes of early postpartum hemorrhage are uterine atony and laceration. Late postpartum hemorrhage (secondary postpartum hemorrhage) is caused by retained placental fragments or subinvolution. Coagulation defects and infection can also result in postpartum hemorrhage (Rice-Simpson, 2010). Women at greatest risk for postpartum hemorrhage include those who have labor induction or augmentation, multiple fetuses (twins, etc.), macrosomia, preeclampsia, operative deliveries, and chorioamnionitis.
The body responds to hypovolemia (reduced blood volume) with increased heart and respiratory rates. These reactions increase the oxygen content of circulating erythrocytes (red blood cells). A decrease in blood volume causes the woman’s skin and mucous membranes to become pale, cold, and moist (clammy). As the blood loss continues, blood flow to the brain decreases and the woman becomes restless, confused, anxious, and lethargic. A collaborative effort by the health care team is necessary to provide prompt care.
The management for hypovolemic shock (reduced blood volume) resulting from postpartum hemorrhage includes:
• Recognizing the specific cause (where the blood is coming from)
• Starting intravenous fluids to maintain circulating volume
• Inserting an indwelling (Foley) catheter to assess kidney function and urinary output
Early Postpartum Hemorrhage
Uterine Atony
Uterine atony (hypotonic uterus) is the inability of the myometrium muscle (middle muscle, which has interlacing “figure eight” fibers) of the uterus to contract and stay contracted around the open blood vessels. Without this contraction, the vessels at the placental implantation site cannot close and begin to heal. Uterine atony is the most common cause of early postpartum hemorrhage and occurs during the first hours after birth.
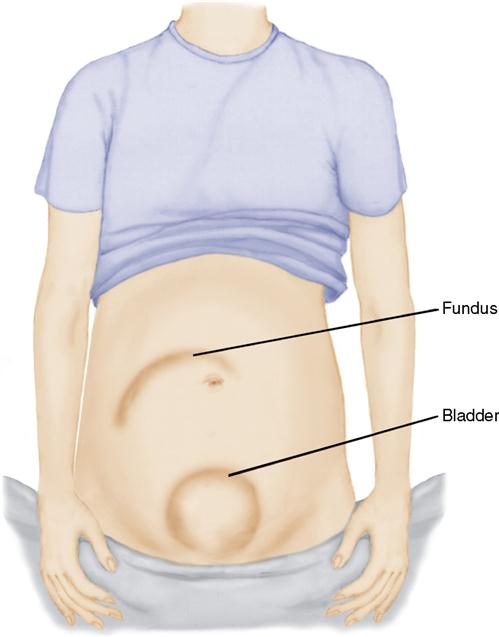
Mechanical factors that contribute to the inability of muscles to contract include retained placental fragments or large blood clots. Extreme uterine distention (e.g., multifetal gestations, hydramnios) can cause uterine atony. Overstretching may cause a lack of efficiency of the smooth muscle cells to contract. A full bladder can also prevent the uterus from contracting (see Figure 17-1).
Metabolic factors may contribute to uterine atony. Muscle exhaustion can occur from lactic acid buildup (derived from prolonged muscle activity and the breakdown of glycogen). Because calcium is an important regulator of smooth muscle tone, hypocalcemia may be implicated in some cases of uterine atony.
Drugs have important effects on postpartum uterine tone. Magnesium sulfate administered to prevent seizures or as a tocolytic agent may result in uterine atony by impairing calcium-mediated properties within the cells. In addition, calcium channel blockers, such as nifedipine, used in preterm uterine contractions may also inhibit postpartum uterine contractions.
Trauma and Lacerations
Trauma includes vaginal, cervical, or perineal lacerations. Postpartum hemorrhage can occur as a result of extension of the perineal incisions (and incisions during a cesarean birth). All lacerations should be promptly sutured. Large perineal lacerations can occur in difficult or precipitate deliveries, in primigravidas with large newborns (more than 4.1 kg [9 lbs]), and in deliveries requiring instrument assistance such as forceps or a vacuum extractor.
Lacerations are suspected when excessive bleeding occurs and the uterine fundus is firmly contracted. Bleeding from the vagina is usually bright red, compared with lochia, which is dark red and not profuse or continuous. Signs of shock may occur.
Retained Placenta
Hemorrhage may occur if pieces of the placenta remain in the uterus. This prevents the uterus from contracting effectively and can result in either early or late hemorrhage. Careful examination of the placenta after delivery is essential. Oxytocics are administered to expel the fragments of the placenta but may not be sufficient to expel all of the fragments, so dilation and evacuation (D&E) may be necessary. Placenta accreta is the term used for a placenta that adheres to the uterine wall. Profuse bleeding may result, and a hysterectomy may be required.
Hematoma
Hematoma (collection of blood within the tissues) may result from injury to blood vessels in the perineum or in the vagina. Soft tissues in other areas may be involved, which are typically seen as a bulging, bluish mass. Hematomas containing 250 to 500 mL of blood may develop rapidly. A hematoma may form in the upper portion of the vagina or may occur upward into the broad ligament, which can result in massive hemorrhage.
Perineal pain, rather than noticeable bleeding, is a distinguishing characteristic of a hematoma, and the uterus remains firm but the blood pressure decreases. The woman may not be able to void because of pressure on the urethra, or she may feel the urge to defecate because of pressure on the rectum. A hematoma can cause severe pain and may require a surgical incision to remove the clot. Small vulvar hematomas may be treated with the application of ice packs or alternate hot and cold applications.
Late Postpartum Hemorrhage
Late postpartum hemorrhage can occur 1 to 2 weeks after delivery and is typically due to subinvolution (failure of the uterus to return to pre-pregnant size). The site of placental implantation is the last to heal and regenerate after delivery. A vascular area, retained placental fragments, or infection may be the cause of late postpartum hemorrhage. The clinic nurse should be alert to symptoms that include a fundal height higher than expected postpartum (after 10 days it should be difficult to palpate because it should be well into the pelvis) and persistent lochia rubra. Infection may be suspected if a foul odor to the lochia is noted. Treatment may include administration of methylergonovine (Methergine), oxytocin, or prostaglandins to contract the uterus, antimicrobials if infection is present, or D&E if retained placental fragments are suspected.
Subinvolution
Subinvolution of the uterus occurs when the uterus fails to return to its nonpregnant size; instead, it remains enlarged and still has a lochial discharge. Subinvolution may result from a small retained placental fragment or mild endometritis. Ultrasound can be used to identify retained placental fragments. Excessively vigorous massage of the uterus may contribute to this problem. Long-term loss of blood from subinvolution results in anemia, lack of energy, and exhaustion. Conservative treatment consists of the administration of methylergonovine orally or intramuscularly. Methylergonovine may be contraindicated in hypertensive women. D&E may be needed to remove retained placental fragments.
Coagulation Pathology
Disseminated Intravascular Coagulation
Disseminated intravascular coagulation (DIC) is a condition in which clotting and anticoagulation stimulation occur at the same time. The release of thromboplastin uses up available fibrinogen and platelets, which results in profuse bleeding and intravascular clotting. The key to successful management of DIC is treatment of the causative event. It often is a secondary condition associated with abruptio placentae, gestational hypertension, missed abortion, or fetal death in utero. DIC is discussed in more detail in Chapter 13.
DIC is suspected when the usual measures to stimulate uterine contractions fail to stop vaginal bleeding. Signs of DIC include oozing from an intravenous insertion site, petechiae, ecchymosis, oliguria, and restlessness. In pregnancy and the early postpartum period, shock is considered a late sign of DIC because the increased blood volume during pregnancy delays evidence of the serious blood loss. However, a decreasing pulse pressure (the difference between systolic and diastolic blood pressure) with continued bleeding may indicate a serious problem and should be promptly reported to the health care provider. Recombinant activated factor VIIa given intravenously can reverse symptoms of DIC (Karrie, 2006).
Von Willebrand’s Disease
Von Willebrand’s disease is an inherited disorder characterized by a decrease in plasma factor VIII, which is essential for proper platelet function. The woman may have a history of easy bruising, frequent nosebleeds, and heavy menses. There is typically a normal increase in plasma factor VIII during pregnancy, so symptoms can be masked. Hemorrhage from von Willebrand’s disease is treated with the administration of cryoprecipitate to raise factor VIII levels.
Anaphylactoid Syndrome of Pregnancy (Amniotic Fluid Embolism)
Amniotic fluid embolism, currently known as anaphylactoid syndrome of pregnancy, is due to the unanticipated entrance of amniotic fluid into maternal circulation, which triggers the release of mediators (such as bradykinin, cytokines, prostaglandins, leukotrienes, thromboxane, and others) that causes pulmonary artery vasospasm and hypoxia. The hypoxia results in myocardial (heart muscle) damage that can cause maternal left-sided heart failure.
Nursing interventions include recognizing symptoms such as acute dyspnea, hypotension, and possibly seizures; providing oxygen for the woman; preparing blood work for laboratory coagulation studies; providing emotional support for the family; and assisting in the transfer of the patient to the intensive care unit (ICU). This condition appears without warning, and the morbidity and mortality rate is high.
Interventions for Hemorrhage
Prevention of Hemorrhage
Postpartum hemorrhage caused by uterine atony after a vaginal birth can be greatly reduced by prophylactic administration of uterotonic drugs (oxytocin) after the delivery of the placenta. An intravenous solution of oxytocin may be started to contract the uterus. Early clamping of the umbilical cord and assisted delivery of the placenta may also prevent uterine atony and postpartum hemorrhage. The placenta should be carefully examined to determine that it is intact. Massage of the uterine fundus can aid in uterine muscle contraction. Observation and prevention of bladder distention is an important postpartum nursing responsibility (seeFigure 17-1).
Nursing Assessment and Management of Postpartum Hemorrhage
Prompt assessment and management can minimize blood loss. Essential nursing interventions are shown in Box 17-2 and Nursing Care Plan 17-1. In the first 24 hours after delivery, the nurse should be alert to the signs and symptoms of uterine atony and postpartum hemorrhage, which include a soft, boggy uterine fundus, a fundus that quickly loses firmness after massage, a fundus that is not midline or above the level of the umbilicus, excessive lochia, or excessive clots. The nurse should report signs of shock, including tachycardia and low blood pressure.
The first nursing action with uterine atony should be to massage the uterus until firm and to express clots that may have accumulated in the uterus. One hand should be placed above the symphysis pubis to support the lower uterine segment while the fundus is gently but firmly massaged in a circular motion (Skill 17-1). It is critical not to try to express clots until the uterus is firmly contracted. Pushing on an uncontracted uterus can invert the uterus and cause a massive hemorrhage. If the uterus does not stay contracted, the health care provider may order an intravenous infusion of dilute oxytocin, which usually will increase muscle tone and control bleeding. Methylergonovine may be given intramuscularly if bleeding continues. This drug has side effects of elevating blood pressure; therefore it should not be given to a woman who is hypertensive. If uterine massage and administration of oxytocin and other medications are not effective, it may be necessary to return the woman to a birthing room to stop the bleeding. A physician may attempt bimanual compression by placing one hand in the vagina with the other pushing against the fundus through the abdominal wall or surgical intervention may be needed.
An accurate assessment of blood loss is important. Blood loss can be assessed by weighing the perineal pad (1 g = 1 mL, subtracting weight of dry pad from saturated pad). If possible, a gram scale should be kept in the postpartum unit and used to measure blood loss. Vital signs should be assessed at least every 15 minutes until stabilized, and the woman should be assessed for signs of hypovolemic shock. Accurate assessment of intake and output is necessary. Urinary output should be at least 30 mL/hour.
Other measures include observing for a full bladder. A full bladder pushes on the uterus and can keep the uterus from contracting; catheterization may be


 Nursing Care Plan 17-1
Nursing Care Plan 17-1

